1. Introduction
A large number of high-latitude and plateau areas in China remain in a low-temperature environment in winter for a long time. The hydration reaction of concretes slows down in a low-temperature environment and stops when the temperature reaches below 5 °C.1,2) A low-temperature environment prolongs the initial and final setting times of concretes, impeding the improvement of the early strength of concretes. Moreover, it is difficult for concretes to reach the designed demolding strength in a short time, causing early curing difficulties.3,4) In order to make concretes reach the designed demolding trength in a short time, hot air curing and steam curing are most frequently used;5-7) however, these methods require special equipment, consume a lot of energy, lead to the phenomena of late strength decline and cracking, decrease the strength and quality of concretes, and even cause structural damage.8,9) Early strength agents can be divided into three categories: inorganic, organic, and mineral. Generally, chlorate, sulfate, nitrate, and organic amine early strength agents are most commonly used. The use of 0.5 ~ 2.0 % of chlorate early strength agents (CaCl2, KCl) can effectively increase the strength of concretes by 50 % ~ 100% in three days and by 20%~ 40% in seven days.10,11) Ca(OH)2 reacts with CaCl2 generated in a hydration reaction to produce CaCl2·3Ca(OH)2·H2O, which is hardly soluble in water, decreases the content of Ca(OH)2,12) and increases the concentration of Ca+ ions to accelerate the production of C3S. However, as Cl− ions can corrode steel bars in a concrete structure,13-15) C2H2O4Ca and CaBr2 are used to improve the early strength of concretes. Nitrate can improve the pore structure of early concrete hydration products and make them denser. However, the improvement of the early strength of concretes by nitrate-based early strength agents does not depend on the mixing amount.16-18)
Calcium formate (C2H2O4Ca), anhydrous sodium sulfate (Na2SO4), triethanolamine (C6H15O3N), calcium bromide (CaBr2), and triisopropanolamine (C9H21NO3) were used as early strength agents in the present work. The effects of single-doped early strength agents on the early strength of concretes were analyzed, and the best mixing ratio of early strength agents was determined. The aim was to improve the early strength of concrete in low temperature environment and improve the efficiency of the template in construction in low temperature in winter, ensuring the quality of concrete, protecting the environment, and improving construction efficiency. The reaction mechanism of early strength agents was analyzed from the perspectives of strength, porosity, and hydration products.
2. Materials and Experiments
2.1 Raw Materials
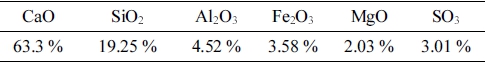
Cement: P·O 42.5 cement produced by Anhui Tongling Conch Cement Co.(main ingredients in Table 1).
Fly ash: grade II, fineness 2.6 %, and loss on ignition 6.9 %.
Sand: medium sand produced in Lu'an area, fineness modulus 2.81, and apparent density 2.62 Kg/cm3.
gravel: medium gravel from Wuhu area, continuous gradation 5 ~ 25 mm, apparent density 2.73 g/cm3, and mud content 0.59 %.
Water reducing agent: polycarboxylic acid water reducing agent made by Nanjing Subot New Material Co., water reducing ratio: 20 %, with a recommended the optimum mix amount of 0.7 %.
Early strength component: C2H2O4Ca white powder, Na2SO4 white powder, TEA(C6H15O3N) colorless viscous liquid, CaBr2 white crystals and TIPA(C9H21NO3) white powder produced by Shanghai Macleans Biochemical Co., Ltd.
2.2 Experimental Methods
2.2.1 Single-doped Experiment of Early Strength Agents
Strength experiment: Calcium formate (C2H2O4Ca), anhydrous sodium sulfate (Na2SO4), triethanolamine (C6H15O3N), calcium bromide (CaBr2), and triisopropanolamine (C9H21NO3) were used as early strength agents in different proportions to perform single-doped experiments. According to the “Concrete Strength Inspection and Evaluation Standard” (GBJ107-87), these early strength agents were mixed in a concrete mixer based on the C40 concrete mixing ratio. The mixed concrete was then placed in a 100 mm × 100 mm × 100 mm cube mold and kept on a vibration table for mechanical vibration molding. The compressive strengths of the 18 h concrete specimens at 5 °C, 10 °C, and 20 °C were measured, and the average of three test blocks in each group was considered as the final value. This step was the basis for the analysis and determination of the type and proportion of double-doped early strength agents.
2.2.2 Double-doped Experiment of Early Strength Agents
Strength experiment: According to the “Concrete Strength Test and Evaluation Criteria” (GBJ107-87), the compressive strength of the double-doped group was evaluated based on the C40 concrete mixing ratio. The method of test block preparation was similar to that of the single-doped strength experiment, and the test block specimen was 100 mm × 100 mm × 100 mm. Six specimens in each group were cured at 5 °C and 10 °C for 18 h, 1 d, 3 d, 7 d. One of them immediately stopped hydration, and three of the other five specimens were selected for the experiment.
Fluidity and setting time experiments: According to the “Method for Determining Cement Setting Time” (GB1346- 2011-T) and the “Test Method for Net Cement Paste Fluidity” (GB/T8077-2000), the effects of additives on concrete cement components were determined by comparing the initial and final setting times of the cement paste and the changes in the liquidity of the early strength agents and the cement paste in the control group. The recommended water/cement ratios of 0.3 and 0.4 were used for the determination of the cement setting time and the flowability test, respectively.
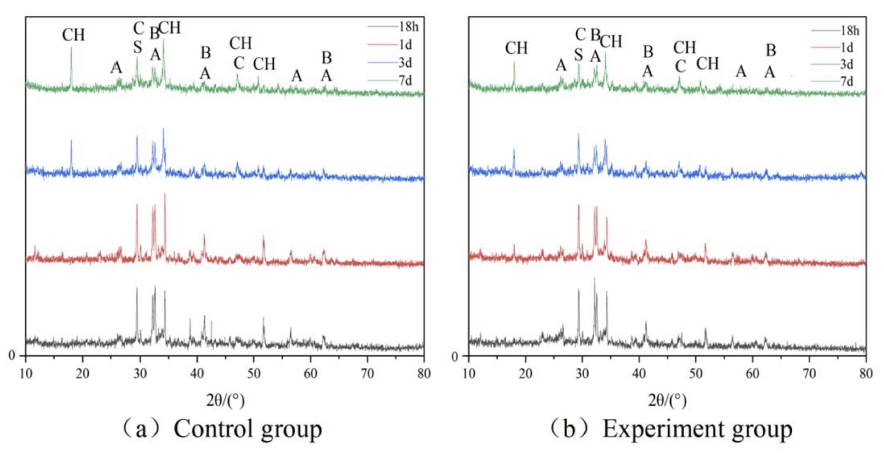
XRD nalysis: Hydration products were analyzed by an X-ray diffractometer equipped with a Cu target (Empyrean, Panaco, The Netherlands) at a voltage of 3 kW in a scan range 10o ~ 80o with a step of 0.02o and a scan rate of 10o/min. Cement paste samples cured to different ages were crushed and sampled at different locations, immediately immersed in absolute ethanol to stop hydration, dried in a vacuum oven at 60 ºC, ground, and sieved out powder particles of size < 0.075 mm.
TG/DSC analysis: TG experiments were performed on a differential thermal analyzer (STA 7300, HITACHI, Japan) in a temperature range of 28 °C ~ 1,000 °C under a nitrogen environment at a heating rate of 10 °C/min. Cement paste samples cured to different ages were crushed and sampled at different locations, immediately immersed in absolute ethanol to stop hydration, dried in a vacuum oven at 60 ºC, ground, and sieved out powder particles pf size < 0.075 mm.
SEM analysis: The morphology of hydration products was observed by a scanning electron microscope (ZEISS GeminiSEM 450, Germany). Samples were taken from different locations of broken concrete specimens, immediately immersed in anhydrous ethanol to stop hydration, dried in a vacuum oven at 60 °C. New cross-sectional sheet samples were vacuum-sprayed with platinum, and their micro-morphologies were observed.
Porosity analysis: Concrete porosity tests were performed on a mercury porosimeter (AutoPore Iv 9510, Mack Inc., USA). Concrete specimens were crushed and sampled at various locations, immediately immersed in anhydrous ethanol to stop hydration, dried in a vacuum oven at 60 °C, and collected powder particles of diameter <10 mm for the MIP est.
3. Experimental Results and Discussion
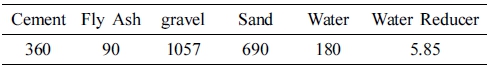
The C40 concrete mixing proportion provided by the Dongkai New Building Material Company was adopted in this experiment, and the five early strength agents were mixed in a gradient. The mixing ratio of the control group is presented in Table 2.
3.1 Results and Analysis of the Early Strength Agent Single-doped Group
Calcium formate (C2H2O4Ca), anhydrous sodium sulfate (Na2SO4), triethanolamine (C6H15O3N), calcium bromide (CaBr2), and triisopropanolamine (C9H21NO3) were used in different proportions separately to perform singledoped experiments. The experimental ratio was determined according to the recommended mixing amount and trial data. The mixing amount of Na2SO4, C2H2O4Ca, and CaBr2 ranged from 1 % to 2.5 %, and the corresponding value for TIPA (C9H21NO3) and TIPA (C9H21NO3) ranged from 0.06 % to 0.09 %. These early strength agents were cured for 18 h at 5 °C, 10 °C, and 20 °C. The measured compressive strengths are presented in Fig. 1.
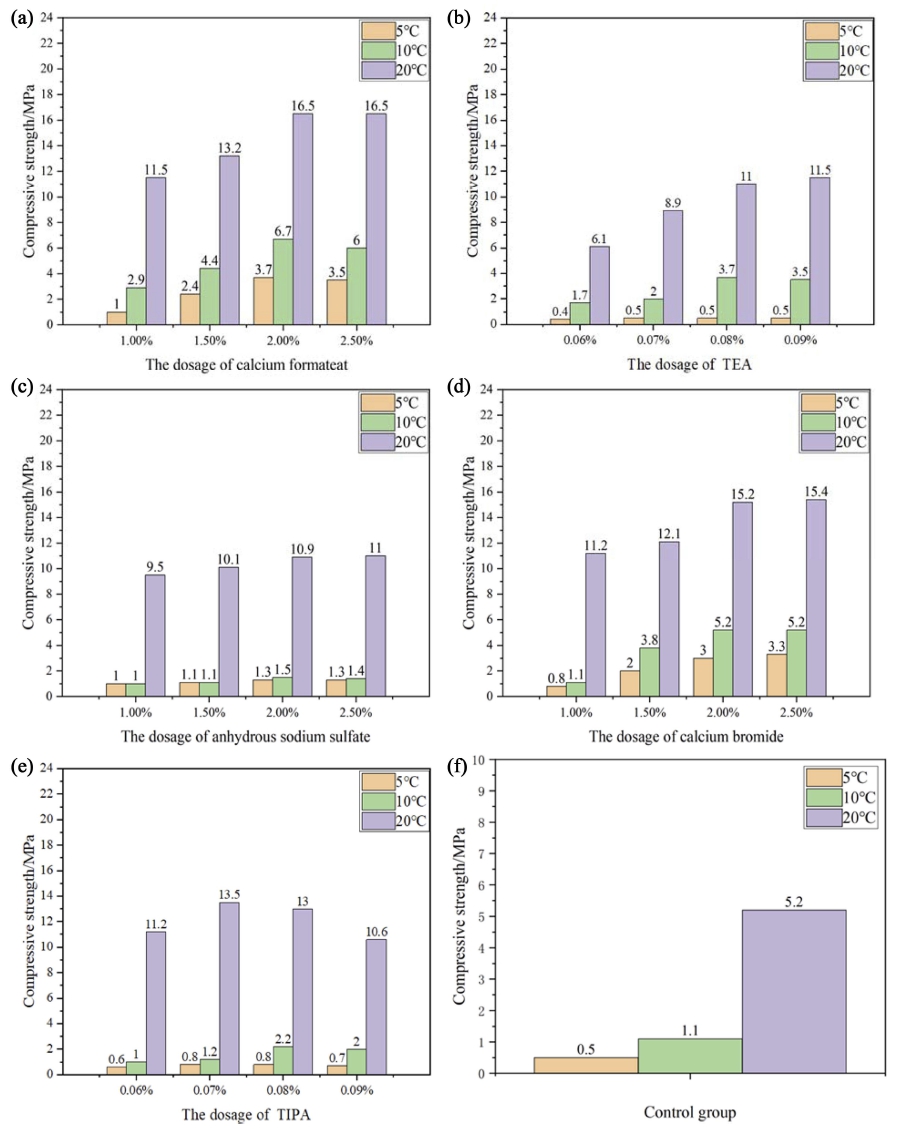
Fig. 1
18 h compressive strength of concrete (a: Calcium formateat group, b: TEA group, c: Anhydrous sodium sulfate group, d: Calcium bromide group, e:TIPA group, f:Control group).
The early strength of the concrete decreased at low temperatures.19) The lower the temperature during the same age, the lower the early strength of the concrete. The compressive strength of the experimental groups mixed with early strength agents at each temperature increased as compared to that of the control group. As the percentage of early strength agents increased, the concrete strength manifested an increasing trend. After reaching the optimum mixing amount, the early strength of the concrete started to stabilize or slightly decrease with the increasing early strength agent content. The optimum mixing amount of C2H2O4Ca and Na2SO4 was 2 %, and that for CaBr2 was 2.5 %. Na2SO4 had a greater influence on improving the early strength of the concrete at 20 °C and room temperature. When the mixing amount was 2 %, the concrete strength increased by 5.7 MPa. The effect of ameliorating the early strength of the concrete was not obvious at 5 °C and 10 °C. C2H2O4Ca and CaBr2 enhanced the early strength of the concrete at low, medium, and high temperatures. When the mixing amount was 2 %, the strength of the C2H2O4Ca group increased by 3.2 MPa, 5.6 MPa, and 11.3 MPa, respectively, at low, medium, and high temperatures. The strength of the CaBr2 group with a mixing amount of 2.5 % increased by 2.8 MPa, 4.1 MPa, and 10.2 MPa, respectively, at low, medium, and high temperatures. TEA (C6H15O3N) and TIPA (C9H21NO3) had almost no effect on concrete strength improvement at 5 °C; however, the early strength of the concrete increased noticeably at 10 °C and 20 °C by these two agents. TEA (C6H15O3N) had the best performance when the mixing amount was 0.08 %, and the early strength of the concrete increased by 2.6 MPa and 5.8 MPa at 10 °C and 20 °C, respectively. The early strength improvement performance of TIPA (C9H21NO3) was relatively good when the mixing content was 0.08 %, and the early strength of the concrete increased by 1.1 MPa and 7.8 MPa at 5 °C and 10 °C, respectively. TIPA (C9H21NO3) had a great influence on the workability of the concrete. With the increase of the mixing amount, the amount of collapsed slump declined significantly. With the increasing mixing amount, the phenomenon of flash set easily occurred because TIPA (C9H21NO3) had a relatively quick setting effect.20) When the prolonged mixing time, small pieces easily appeared in the concrete, affecting the internal structure of the concrete. It is not conducive to the later strength and stability of concretes; thus, the amount of TIPA (C9H21NO3) must be limited according to the actual type of concrete used in a construction project.
Moreover, C2H2O4Ca and CaBr2 yielded a better comprehensive effect. TEA (C6H15O3N) also had a better early strength effect at 10 °C and 20 °C. C2H2O4Ca increased the concentration of Ca2+ ions in the liquid phase; thus, it could dissolve quickly in C3S, accelerate the crystallization of Ca(OH)2,21,22) and promote an early hydration reaction. HCOO- in C2H2O4Ca could form substances similar to AFt and AFm to shorten the setting time, thereby enhancing the early strength of the concrete. HCOO− diffused faster in cement than Ca2+, and penetrated into C2S and C3S hydration layers to accelerate the precipitation of Ca(OH)2 crystals, the decomposition of calcium silicate, promote the formation of C-S-H gel;23) thus, enhancing the early strength of the concrete. CaBr2 had the same early strength mechanism as C2H2O4Ca. CaBr2 ameliorated the early hydration reaction by increasing the Ca2+ concentration and generated substances similar to AFt and AFm to increase the early strength by improving the early density of the concrete microstructure. C2H2O4Ca had a greater impact on the improvement of the early strength of the concrete. TEA (C6H15O3N) accelerated the hydration reaction of C3A and C4AF through the complexation of Ca2+ and Al3+ in the process of concrete hydration. TEA (C6H15O3N) also experienced emulsification; thus, its molecules were adsorbed. On the surface of cement particles, the surface colloid film produced by cement hydration was decomposed, preventing the agglomeration of cement particles and increasing the contact area between cement particles and water.24,25) The addition of TEA (C6H15O3N) reduced the tension of the solution to a certain extent and ensured the penetration of water in cement particles, strengthening the solid volume expansion of cement, making the internal structure of the concrete denser, and enhancing the later strength of the concrete.26,27)
3.2 Results and Analysis of the Early Strength Agent Double-doped Group
C2H2O4Ca and TEA (C6H15O3N) were used as doubledoped compound early strength agents. The optimum mixing amount of C2H2O4Ca and TEA (C6H15O3N) was obtained according to the single-doped experiments. Doubledoped orthogonal experiments with different proportions were carried out, and the optimum mixing proportion was determined as 2 % of C2H2O4Ca + 0.08 % of TEA (C6H15O3N) at low and medium temperatures.
3.2.1 Strength Test
The concrete group with the compound early strength agent (2 % of C2H2O4Ca + 0.08 % of TEA) was denoted as the experimental group, and the group without the compound early strength agent was termed as the control group. The concrete mixing proportion was similar to that of the single-doped experiments. Both the control and experimental groups were kept at 5 °C and 10 °C for 18 h, 1 d, 3 d, and 7 d, and the corresponding strength are listed in Table 3.
Table 3
Compressive strength of control group and experimental group at different ages under low temperature condition.
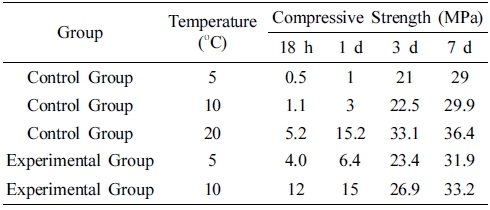 |
It is clear that the early strength of the concrete was directly proportional to the curing temperature. The lower the curing temperature, the lower the early strength of the concrete. With the prolonged aging time, the degree of strength reduction had a decreasing trend. The strength of the experimental group was higher than that of the control group at the same temperature. With the increasing aging time, the strength difference between the control and experimental groups decreased gradually. When the concrete was cured at 5 °C for 18 h, 1 d, 3 d, and 7 d, the strength of the experimental group increased by 420 %, 540 %, 11.4 %, and 10 %, respectively, whereas at 10 °C, the corresponding values were 991 %, 400 %, 19.6 %, and 11 %, respectively. The concrete strength of each age is close to the 20 °C standard curing strength. As compared to all single early strength agents, the compound early strength agent had a better effect on the improvement of the concrete strength in the lowtemperature environment, and it can be attributed to the synergistic effect of C2H2O4Ca and TEA (C6H15O3N) on the early strength of the concrete. C2H2O4Ca promoted the rate of the first-stage hydration reaction of cement to improve the early strength of the concrete. TEA (C6H15O3N) had an emulsifying effect and prevented the agglomeration of particles during cement hydration to improve the early strength of the concrete.28) Hence, cement particles could extend the contact area with water and increase the reaction rate, making the cement hydration reaction deeper and more comprehensive.
3.2.2 Fluidity
According to the “Net Cement Paste Fluidity Test Method” (GB/T8077−2000), the fluidity of the control and experimental groups with a water/cement ratio of 0.4 at room temperature (20 °C) after 0 min, 30 min, and 60 min was measured, and the corresponding results are presented in Table 4.
The fluidity of the control group after 0 min, 30 min, and 60 min was measured as 255 mm, 225 mm, and 186 mm, respectively, and the corresponding values for the experimental group were 236 mm, 213 mm, and 174 mm, respectively. The admixture made the fluidity of the cement paste drop significantly at the initial moment. With the increase of time, the decrease rate of fluidity was reduced slightly. Hence, the compound admixture had little influence on the fluidity of cement components of the concrete. It happened because TEA (C6H15O3N) hindered the agglomeration of cement particles,29) dispersed the slurry, and reduced the influence of the early strength agent on the initial fluidity of cement. The low-temperature early-strength agent reduces the initial fluidity of the pure slurry. With the extension of time, the fluidity decreases gradually. Therefore, the low-temperature early-strength agent should be used together with a small amount of water reducing agent.
3.2.3 Setting Time
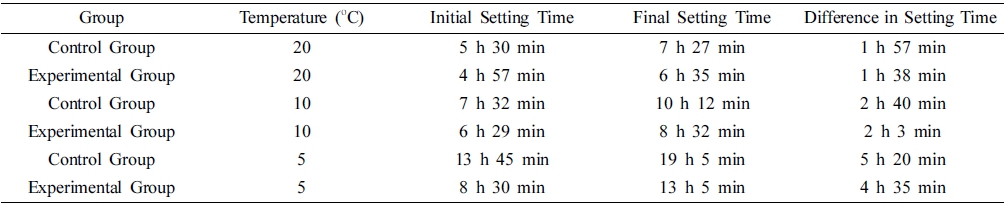
According to the “Method for Measuring Cement Setting Time” (GB1346-2011-T) the initial and final setting times of the control and experimental groups with a water/cement ratio of 0.3 at 5 °C, 10 °C, and 20 °C were measured, and the corresponding results are presented in Table 5.
The initial and final setting times of the experimental group at different temperatures are earlier than those of the control group, and the difference between the initial and final setting times of the experimental group was shorter than that of the control group. This phenomenon became more apparent as the temperature decreased. The initial and final setting times of the control group at room temperature were 5 h 30 min and 7 h 27 min, respectively, and the corresponding values at 5 °C were 13 h 45 min and 19 h 5 min, respectively. The difference between the initial and final setting times changed from 1 h 38 min to 5 h 20 min. After the addition of the early strength agent, the setting time of cement got effectively shortened. The initial setting time was reduced by 10 % at room temperature, the final setting time was shortened by 11.6 %, and the difference between the initial and final setting times decreased by 16.2 %. At 5 °C, the corresponding values were 30.7 %, 31.4 %, and 14 %, respectively, whereas at 10 °C, the values were 13.3 %, 16.3 %, and 23.1 %, respectively. Therefore, the addition of the compound early strength agent significantly shortened the setting time of the cement slurry. The initial setting time and final setting time of the experimental group with the addition of early-strength agent at low temperature lags behind that of the control group at 20 °C.
4. Microscopic Analysis of Hydration Products
4.1 Analysis of Hydration Products
4.1.1 XRD Analysis
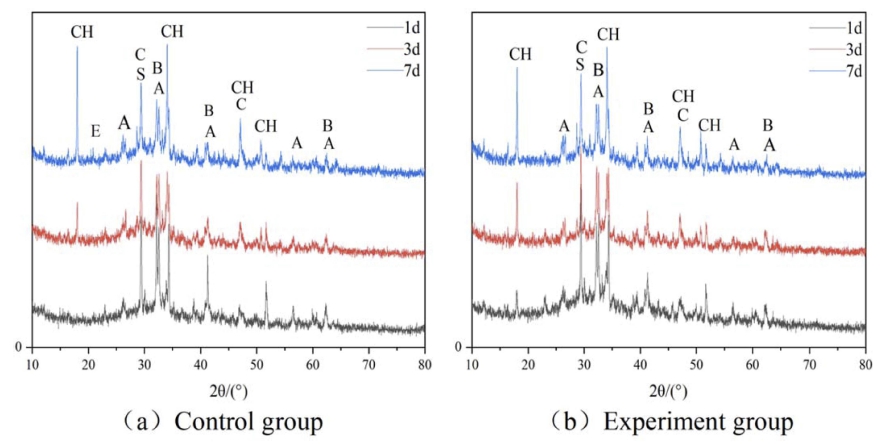
Figs. 2 and 3 display the XRD spectra of cement paste hydration products at 5 °C and 10 °C, respectively. The diffraction peaks of Ca(OH)2 appeared around 18°, 34°, 47°, and 50°, and those of C3S were detected near 26°, 32°, 41°, 56°, and 62°. The diffraction peaks of C2S appeared near 32°, 41°, and 62°, and those of CaCO3 were detected near 29° and 47°. The diffraction peak intensity of Ca(OH)2 was enhanced with the prolonged aging time, whereas those of C2S and C3S had a decreasing trend. Ca(OH)2 remained stable in the later stage of the hydration process because CAF absorbed a part of Ca(OH)2. The diffraction peak intensity of each hydration product of the early experimental group was higher than that of the control group at the same age. The experimental group had a prominent Ca(OH)2 diffraction peak around 18o at each temperature after 1 d of curing, whereas the control group had an insignificant Ca(OH)2 diffraction peak at 18o. The compound early strength agent significantly promoted cement hydration at low temperatures, improved the crystallinity of Ca(OH)2, and generated a large amount of Ca(OH)2 after 1 d of curing. It happened because the diffusion rate of HCOO- in C2H2O4Ca was faster than that of Ca+. HCOO- penetrated the Ca(OH)2 layer covering C2S and decreased the concentration of hydration products around it, thereby promoting the slower hydration of C2S to generate more C-S-H and Ca(OH)2. The difference in the product diffraction peak intensities of the experimental and control groups after 3 d of curing was significantly reduced. Hydration products of the experimental and control groups after 7 d of curing were the same. The compound early strength agent promoted the early hydration reaction of cement at low temperatures; however, it could not significantly improve middle and late hydration reactions.
4.1.2 TG Analysis
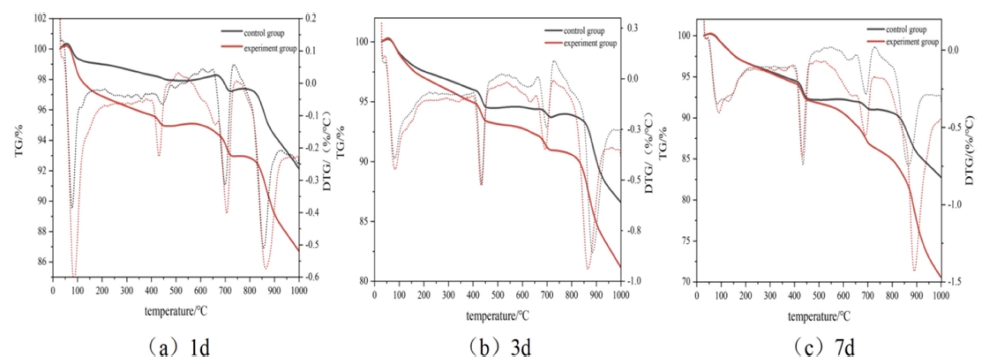
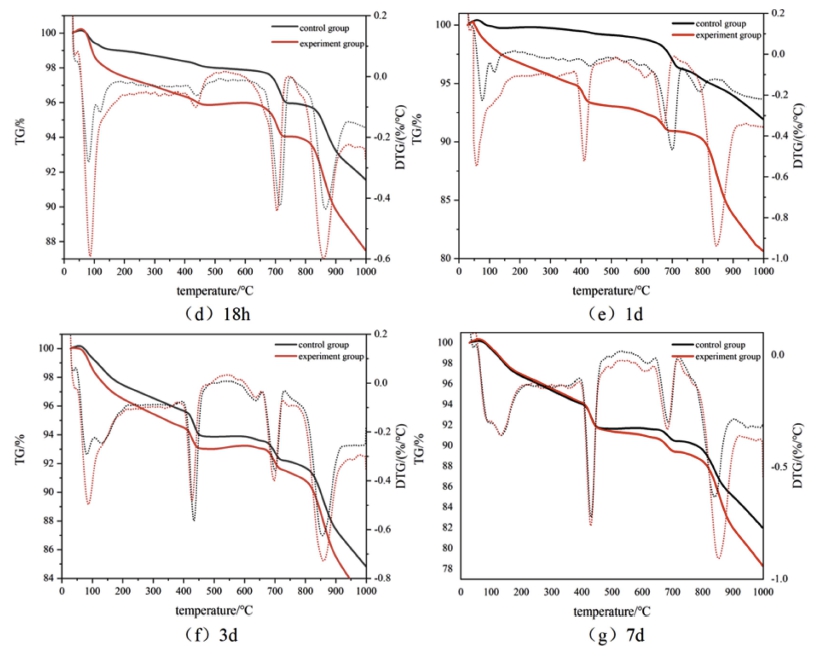
The blank sample of cement paste and the sample mixed with the compound early strength agent were denoted as the control group and the experimental group, respectively. The TG curves after 1 d, 3 d, and 7 d of curing at 5 °C are displayed in Fig. 3. The mass loss at 50 °C ~ 100 °C can be attributed to the adsorption of water in C-S-H gel, and that at 100 °C ~ 200°C can be ascribed to the bound water in C-S-H gel and the endothermic dehydration of ettringite. The mass loss at 390 °C ~ 460°C occurred due to the thermal decomposition of Ca(OH)2, whereas that at 650 °C ~ 750 °C corresponds to the decomposition of CaCO3.
Fig. 4 reveals that the quality loss of the experimental group at each stage was greater than that of the control group in the same age period, especially after 1 d and 3 d of curing. At the same age, the mass loss of the experimental group at 50°C ~ 200°C was significantly greater than that of the control group, indicating that the amount of C-S-H gel and ettringite produced at this stage was higher than that of the control group. The mass loss of the experimental group at 50 °C ~ 200 °C was greater than that at 350 °C ~ 450 °C. The amount of Ca(OH)2 generated in the control group at this stage was higher than that of the experimental group. The compound early strength agent promoted the speed of hydration reactions and increased the amount of hydration products. The remaining mass of the final sample decreased with the increasing curing age. The hydration speed of the experimental group in the early stage was significantly higher than that of the control group. The difference in the quality loss caused by hydration products between the two groups after 7 d of curing was very small, implying that the compound early strength agent had a better impact on the early hydration stage. The improvement effect on cement hydration gradually decreased with the prolonged curing age; thus, the influence was very small in the middle and late stages.
Fig. 5 exhibits the TG curves after 18 h, 1 d, 3 d, and 7 d of curing at 10 °C. The total mass losses of the experimental group after 18 h and 1 d were greater than those of the control group, revealing that the experimental group generated more hydration products. A distinct loss of quality was noticed at 100 °C ~ 200 °C due to the evaporation of the absorbed water in C-S-H gel and the endothermic dehydration of ettringite, indicating that the compound early strength agent increased the amount of hydration products in this stage. C2H2O4Ca promoted the formation of C3A·Ca(HCOO)·10H2O and C3A· 3Ca(HCOO)2·30H2O. The mass loss at 350 °C ~ 450 °C occurred due to the decomposition of Ca(OH)2. The mass loss of Ca(OH)2 in the experimental group was higher than that in the control group, indicating that the compound early strength agent generated more Ca(OH)2 after 18 h and 1 d of curing due to the early hydration reaction and accelerated precipitation of Ca(OH)2 crystals by C2H2O4Ca. The total mass loss gradually increased with the prolonged curing time. The increase of the curing age made the hydration more complete. At 10 °C, the compound early strength agent affected the cement in the early stage. The promotion effect of hydration was obvious, and the degrees of hydration of the experimental and control groups were almost the same after 3 d and 7 d of curing.
Therefore, the compound early strength agent had a significant enhancement effect on early hydration at both 5 °C and 10 °C.
Use this formula:
where LOI(CH) is the dehydration of Ca(OH)2 in the range of 390 ~ 460 °C, and LOI(CC) is the loss of CO2 in the range of 650 ~ 750 °C.
where WL(CH) is the weight loss due to the dehydration of CH, MW(CH) and MW(H) are the corresponding molecular weights of the CH (74 g/mol) and water (18 g/ mol).
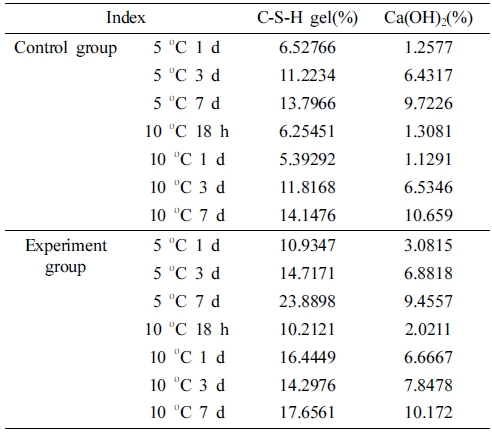
The amount of C-S-H gel and Ca(OH)2 can be calculated. It can be concluded from Table 6 that the amount of CS- H gel produced in the 1d experimental group is 4.41 % higher than that of the control group, and the amount of Ca(OH)2 produces 1.82 % higher than that of the control group under 5 °C. The amount of CSH gel produced in the 3d experiment group is 3.49 % higher than that of the control group, and the amount of Ca(OH)2 produced is 0.45 % higher than that of the control group. The amount of CSH gel produced in the experimental group at 10 °C is 11.05 % higher than that of the control group, and the amount of Ca(OH)2 produced is 5.54 % higher than that of the control group. The amount of C-S-H gel produced in the 3 d experimental group is 2.48 % higher than that of the control group, and the amount of Ca(OH)2 produced is 1.31 % higher than the control group. Data shows that the compound early strength agent can effectively increase the amount of early hydration products of cement in a low-temperature environment, thereby increasing the early strength of concrete.
4.2 Microstructural Analysis
4.2.1 SEM Analysis
Fig. 6 displays the SEM images of concrete hydration products at 5 °C. The surface of the experimental group was covered with hydration products after 18 h of aging. A large number of needle-shaped ettringite particles appeared in the morphology, and numerous flocculent CS- H gels were attached to these ettringite particles, forming a scaffold. At this time, flaky Ca(OH)2 crystals appeared in hydration products. Hydration products were piled up on each other, resulting in a compact structure. In the SEM image of hydration products of the 1 d control group, only a few needle-shaped ettringite particles and sparse CSH gels were observed. Cement surface hydration products were sparsely distributed, the structure was very loose, and some cement surfaces were not hydrated. Hydration products had a thin layer, and the surface contour of the sample was observed. The surface of the 1 d experimental group was covered with hydration products, and a large number of flaky Ca(OH)2 crystals and columnar ettringite particles appeared in the morphology. The increment of the volume and quantity of Ca(OH)2 crystals and ettringite particles was obvious. Pores were filled with columnar ettringite particles and C-S-H gel clusters, and hydration products were piled up to make the structure compact.
The hydration rate at 5 °C was slow, generating very few hydration products at the same aging time. The amount of hydration products in the experimental group after 18 h of aging was significantly higher than that of the control group after 1 d of aging. It implies that the compound early strength agent accelerated the hydration rate of the early cement, effectively improved the density of the concrete structure, and increased the early strength of the concrete. In comparison to the control group after 1 d of aging, the hydration process of the experimental group was greatly accelerated, producing a large amount of hydration products.
Fig. 8 presents the SEM images of concrete hydration products at 10 °C. The 18 h experimental group possessed a large number of hydration products, and numerous flocculent C-S-H gels covered the cement surface. Hydration products were mixed with a large amount of flaky Ca(OH)2 crystals, and the overall structure was loose and porous. Hydration products were poorly attached on the surface of the 1 d control group, and only a few loose C-S-H gels and flaky Ca(OH)2 crystals appeared in the morphology. The structure was not completely developed yet, and the exposed part of the sample surface was large. The 1 d experimental group contained numerous columnar ettringite particles with a compact structure. These ettringite particles became attached with Ca(OH)2 crystals and formed dense clusters and columnar hydrated product crystals to fill structural gaps.
Therefore, the hydration reaction rate of the concrete was reduced at 10 °C. The amount of hydration products produced by the experimental group after 18 h of curing was greater than that after 1 d of aging. The hydration product structure of the experimental group after 1 d of aging was more compact than that of the control group, reflecting that the compound early strength agent accelerated the cement hydration reaction at low temperatures and deepened the hydration.
The type of hydration products was not dependent on temperature; however, the amount of hydration products decreased significantly at low temperatures after the same curing period,30,31) leading to a loose concrete structure with low strength.32,33) The EDS analysis reveals that the same type of hydration product elements (Ca, Si, Al, O) was noticed in the experimental and control groups at 5 °C and 10 °C. The compound early strength agent promoted the hydration process at 5 °C and 10 °C. Early hydration products in the experimental group at 10 °C were denser, leading to a higher concrete strength than the experimental group at 5 °C. C2H2O4Ca in the compound early strength agent effectively accelerated the initial hydration reaction of cement at low temperatures and increased the precipitation of Ca(OH)2 crystals. TEA (C6H15O3N) prevented cement particles from coalescing during hydration and increased the contact area of cement particles with water, thereby increasing the amount of hydration products in the early cement.34-36)
4.2.2 MIP Analysis
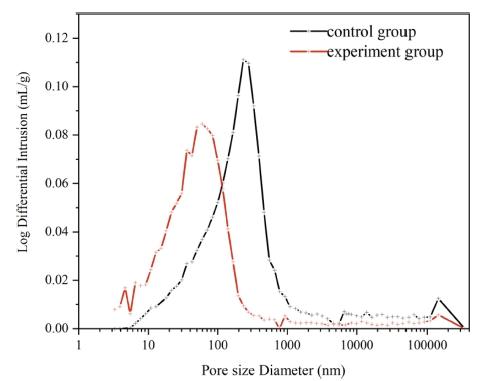
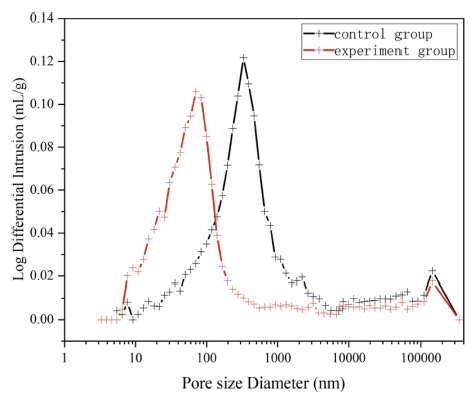
Fig. 10 exhibits the pore size distribution of the 1 d concrete at 10 °C. The pore size distribution of the control group was concentrated in the range of 10 ~ 100 nm (micro-pores) and 100 ~ 1000 nm (large pores). Moreover, a small amount of non-capillary pores (> 1,000 nm) also existed in the sample. These larger pores accounted for a relatively large and loose structure. The pore size distribution of the experimental group was concentrated in the range of 1 ~ 100 nm and 100 ~ 1000 nm. The proportion of ultramicropores and microcapillary pores was relatively large, and the structure was more compact. The control group had a median pore diameter of 203.9 nm, an average pore diameter of 31.6 nm, and a porosity of 24.5832%, and the corresponding values for the experimental group were 58.8 nm, 30.7 nm, and 18.6385 %, respectively. The pore size distribution of the 1 d concrete at 5 °C is displayed in Fig. 11, and it is similar to that at 10 °C. The pore size distribution of the experimental group was concentrated in the range of 1 ~ 100 nm belonging to ultramicropores and microcapillary pores. The pore size distribution of the control group was concentrated in the range of 100–1,000 nm belonging to large pores. The control group had a median pore diameter of 411.3 nm, an average pore diameter of 158.6 nm, and a porosity of 21.6001 %, and the corresponding values for the experimental group were 76.8 nm, 46.9 nm, and 20.7196 %, respectively. Under the same conditions, the concrete with a lower curing temperature had higher porosity and larger mesopore diameter and average pore diameter, and it happened because low curing temperatures slowed down the early hydration reaction.
At low curing temperatures, the early hydration reaction of cement was slow, the precipitation speed of hydration products was slow, and the diffusion speed of ions in the solution was faster than the precipitation speed of hydration products.37) Hence, the C-S-H gel was not evenly and densely distributed, resulting in a loose concrete internal structure and an increased pore size.38-40)
The porosity and pore distribution of the experimental group were significantly smaller than those of the control group at the same aging period. The proportion of fine pores was large. At the same temperature and aging period, the compound early strength agent affected the pore distribution and the total porosity. The early strength agent reduced the pore size of the concrete structure, reduced the porosity, and caused concrete pore refinement, leading to a denser internal structure and an improved early strength of the concrete.
5. Conclusion
(1) In the experiment of single-doped early strength agent, the early strength effect of calcium formate (C2H2O4Ca) and TEA(C6H15O3N)is more prominent in the low temperature environment. When the early strength agent is used in combination, it is not that the higher the amount of the early strength agent, the better the early strength effect. The type, the mechanism of action and the amount of the early strength agent should be considered. In the double-doped experiment of early strength agent, the 2 % calcium formate (C2H2O4Ca) + 0.08 % TEA(C6H15O3N) group has the best early strength effect under low temperature environment, and the strength is increased by 700 % and 540 % at 5 °C for 18 h and 1 d respectively. The strength of 18 h and 1 d under 10 °C environment increases by 991 % and 350 % respectively.
(2) The incorporation of compound early strength agent makes the initial setting time, final setting time and difference of setting time of cement at various temperatures slightly shortened, and the shortening of the time becomes more obvious with the decrease of temperature. Compound early strength agent makes the fluidity of cement paste has decreased, and the impact on the fluidity decreases with the increase of time.
(3) From a microscopic point of view, compound early strength agent under low temperature environment increases the type and quantity of early cement hydration products [C-S-H gel, Ca(OH)2, ettringite, etc.]. Especially in samples aged 18 h and 1 d, compound early strength agent promotes the early hydration reaction of concrete and makes the concrete microstructure denser, thereby improving the early strength of concrete and refining the internal pore size of the concrete. So that the large pores are significantly decreased and the total porosity is reduced.