1. Introduction
Aluminum nitride (AlN) exhibits high thermal conductivity, which is comparable to that of BeO and silicon carbide (SiC) and is more than five times greater than that of Al2O3. AlN also possesses excellent high-temperature strength, a low thermal expansion coefficient similar to that of Si, and strong chemical resistance to fluorine- and chlorine-based gases, making it an attractive material for use in semiconductor manufacturing processes.1,2,3,4) AlN is primarily used in dry etching equipment, chemical vapor deposition (CVD) systems, and other semiconductor process components. Owing to its superior plasma resistance, the application of AlN has been extended to high-power optoelectronic devices. In particular, AlN has been increasingly employed in heater components for photolithography and development equipment, where high thermal conductivity, chemical durability, and minimal light reflectance are required.5,6) To address the requirement for minimal light reflectance, recent studies have focused on achieving black surface coloration using industrially scalable fabrication methods to suppress reflectance while maintaining high density and thermal conductivity.7,8,9) Traditionally, heater materials such as SiC and molybdenum disilicide (MoSi2) have been used. However, AlN has recently attracted intense research attention owing to the growing demand for materials that offer both high thermal conductivity and electrical insulation. AlN shows excellent thermal conductivity (~200 W/m・K) and insulating properties; however, it suffers from low surface emissivity, which can lead to thermal losses and decreased heater efficiency. To address this problem, black AlN has been developed by doping with carbon or other impurities to enhance the AlN surface emissivity.10,11,12,13)
To improve the electrical insulation of AlN ceramics, Lee et al. introduced TiO2 into AlN.14) Consequently, TiO2 partially reacted with AlN during sintering to form TiN phases dispersed along grain boundaries. An addition of only 0.2 wt% TiO2 led to a 1,000-fold increase in electrical resistivity, which was attributed to the suppression of aluminum vacancy migration and the resulting reduction in the ionic conductivity. To enhance the thermal conductivity of AlN, Y2O3 is commonly used as a sintering aid. In the study by Na et al.15) and Chen et al.,16) AlN powder (average particle size: 1.13 µm) and Y2O3 (0.8 µm) were mixed by ball milling and then subjected to uniaxial pressing at 30 MPa and cold isostatic pressing at 200 MPa. After binder removal at 600 °C, the samples were sintered at 1900 °C in a nitrogen atmosphere for 2-10 h. During sintering, Y2O3 reacted with Al2O3, an oxidation product on the surface of AlN, to form secondary phases such as YAG (Y3Al5O12), and the volatilization of these phases during annealing contributed to the improvement in the thermal conductivity.
Building upon the aforementioned prior studies, the current work aims to fabricate a black AlN ceramic heater material with thermal conductivity exceeding 160 W/m・K and sintered density greater than 3.30 g/cm3. To this end, we investigated the effects of TiO2 as a blackening additive, as well as the influence of the granulation, forming, and sintering processes, to establish the optimal processing conditions for achieving high thermal performance.
2. Experimental Procedure
AlN powder (TOYO, JCGA grade, Japan) was used as the starting material, and 3 wt% yttrium oxide (Y2O3) powder (H.C. Starck, C-grade, China) was added as a sintering aid. To achieve black color, various amounts of titanium dioxide (TiO2) powder with a rutile crystal structure (Nanomaterials, Korea) were added with the resulting TiO2 content ranging from 0.05 to 1.0 wt%.
For granule fabrication, the solid powder mixture was dispersed in isopropyl alcohol (IPA) to achieve a solid loading of 20 vol%. Polyvinyl butyral (PVB) was added as a binder to enhance granule strength, and polyethylene glycol (PEG #400) was used to improve formability and lubricity. Uniform slurry mixing was performed using zirconia balls (3Y-TZP) in a ball-milling process performed at 200 rpm for 24 h. The resulting slurry was dried using a spray dryer (Spray Dryer, Atomizer type, Model FS-EP2.5D, Fine Tech Co., Korea) equipped with a disk-rotation nozzle atomizer. During spray drying, the inlet and outlet temperatures were maintained at 80 and 70 °C, respectively, owing to the use of an organic solvent. The nozzle rotation speed was set to 9,000 rpm, and the slurry feed pump was operated at 150 rpm to form the granules. Dried granules were sieved using 80-270 mesh screens to control the particle size distribution.
The granulated powders were packed into a metal mold by tapping and then were uniaxially pressed at 500 kg/cm2. The green compacts were debinded in a vacuum furnace under flowing N2 gas at 600 °C for 5 h. For pressureless sintering, the samples were further sintered in a high-temperature vacuum furnace at 1,850 °C for 5 h in nitrogen atmosphere, using a Boron Nitride (BN) crucible.
The sintered density was measured using the Archimedes’ method. The powders and fracture surfaces were examined by field-emission scanning electron microscopy (FE-SEM; JSM-7100F, JEOL, Japan) to investigate the microstructure. Phase analysis was conducted with X-ray diffraction (XRD, Rigaku DMAX 2200) measurements carried out at room temperature using CuKα radiation under conditions of 40 kV and 30 mA. The thermal conductivity was measured with the laser flash method using a thermal diffusivity analyzer (LFA-447) following the KSL 1604:2007 standard.
3. Results and Discussion
The microstructures of the AlN, Y2O3, and TiO2 starting powders are shown in Fig. 1. Fine AlN powder particles with sizes ranging from 0.3 to 1.0 µm were used in this study to improve densification during sintering. The particles exhibit agglomerated morphologies owing to their fine sizes. The Y2O3 particles which were used as a liquid-phase sintering aid had an average size of approximately 1.0 µm and also showed aggregation; however, the aggregates are expected to be partially broken and dispersed during the ball-milling process. The TiO2 added in small quantities to induce black coloration in AlN was composed of ultrafine spherical particles with an average size of approximately 300 nm to maximize the dispersion of TiO2 in the AlN powder.
To enhance the densification of the AlN green bodies, the granulation and forming processes were optimized to achieve a high packing density. During granulation, the binder and plasticizer additives in the nonaqueous AlN slurry influenced the packing behavior. Table 1 presents the various amounts of the PVB binder and PEG plasticizer used in the slurry. The PVB content was fixed at 1.0 wt%, whereas the PEG content was varied. The AlN-granulated powders were then prepared by spray drying. The morphologies of the resulting granules are shown in Fig. 2. The granules were mostly spherical, with many exhibiting a donut-like shape and a hollow center. Although the particle size distribution was not uniform, this morphology was favorable for improving powder packing in the mold, thereby enhancing the green density.17,18)
Table 1.
Variation of PVB and PEG contents in the AlN slurry used for the spray drying process.
| Sample | Additive composition (wt%) |
| #1 | 1.00 % PVB |
| #2 | 1.00 % PVB + 0.15 % PEG |
| #3 | 1.00 % PVB + 0.30 % PEG |
| #4 | 1.00 % PVB + 0.45 % PEG |
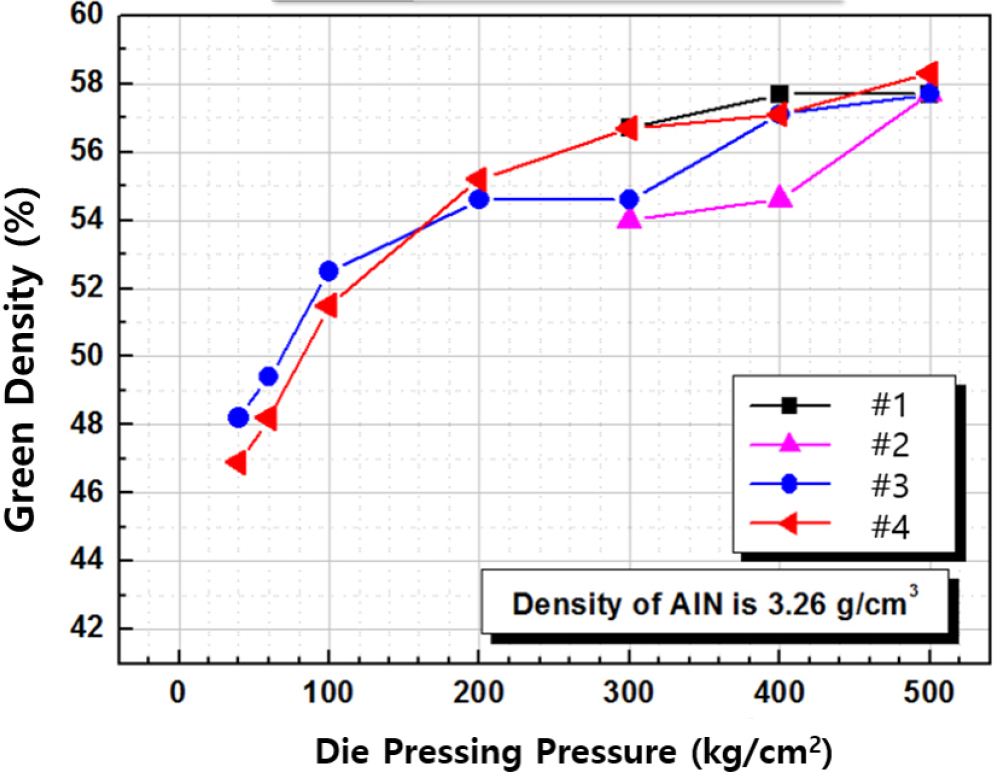
Fig. 3 shows the relative density of green body of the AlN granules as a function of the organic additive content and forming pressure. When only PVB was added, the green density gradually increased with increasing uniaxial pressure, reaching a maximum of approximately 58 % at 400 kg/cm2. This demonstrates the importance of the binders, because typical ceramic powders without binders rarely exceed a packing factor of 55 %. With the addition of PEG, the green density increased rapidly at lower pressures (100-200 kg/cm2) and then gradually increased at pressures above 200 kg/cm2. At higher pressures (above 400 kg/cm2), the increase became more significant, and at 500 kg/cm2, the green density values for samples with higher PEG content exceeded 58 %. This suggests that PEG acted as a lubricant during the initial stage of forming but enhanced the density at higher pressures by improving the binder mobility.19) In the case of samples #1 and #2, the lack of plasticity at low forming pressure led to edge fracturing in the formed bodies.
Fig. 4 shows the fracture surface microstructures of the black AlN compacts formed under different pressures. Sample #4, formed with granules of 20-50 µm, was prepared at the pressures of both 100 and 530 kg/cm2. With increasing forming pressure, better particle contact, and reduced porosity were observed, indicating an improvement in the green density. At lower pressures, some intergranular pores remained owing to the incomplete breakdown of the granules, whereas densification was improved at the pressure of 530 kg/cm2.
The effect of TiO2 addition on the coloration of the sintered AlN was investigated, and the results are shown in Fig. 5. As the TiO2 content increased, the AlN samples gradually darkened, and a distinct black coloration was achieved with 1.0 wt% TiO2. High-temperature heat treatment under nonoxidizing conditions leads to the reduction of TiO2 and the formation of TiN phases, as well as the formation of a solid solution of Ti ions within the AlN lattice. These effects contribute to the black coloration. The substitution of Ti into Al sites reduces ionic conductivity by suppressing Al vacancies, and lattice distortion caused by size mismatch increases visible light absorption, resulting in a darker color.14)
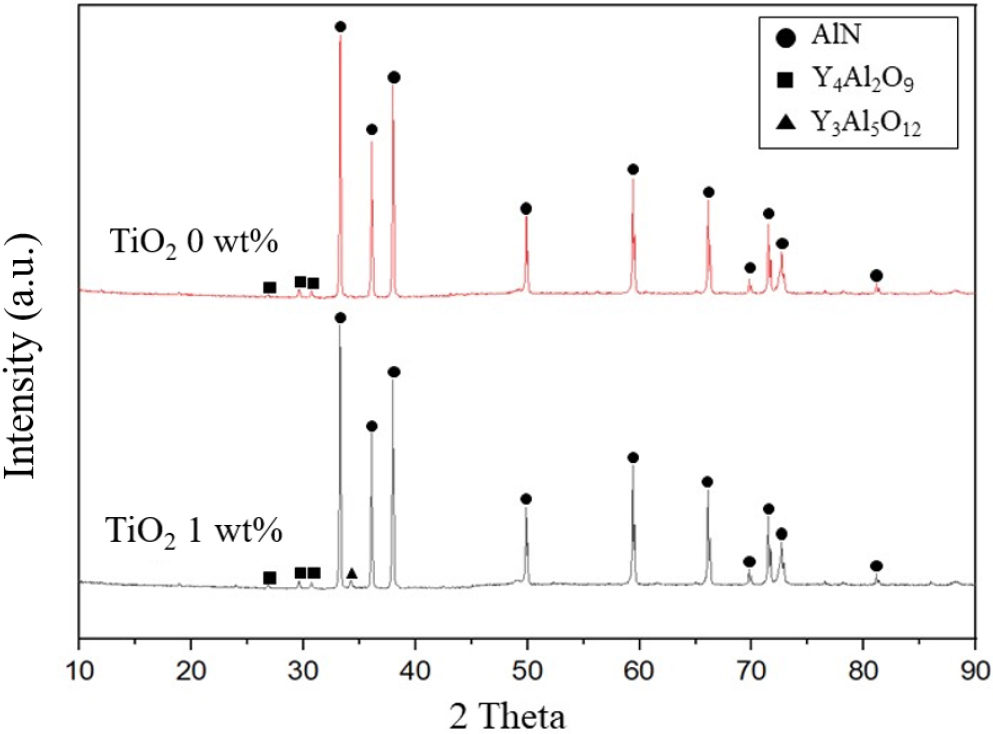
Fig. 6 shows the XRD patterns of AlN sintered at 1,850 °C for 5 h in nitrogen atmosphere according to TiO2 content. In addition to the main peak of AlN crystalline phase, YAG (Y3Al5O12) phase was observed. In the black AlN with 1 wt% TiO2 addition, no TiO2 crystalline phase was observed, while YAM (Y4Al2O9) phase was additionally detected with the YAG crystalline phase. Generally, when Y2O3 is added as a sintering aid in AlN sintering, intermediate crystalline phases exist in addition to the pure YAG crystalline phase, which are influenced by reducing atmosphere, other additives, etc.15,20) In this study, it is considered that the results were caused by the difference in reducing atmosphere due to TiO2 addition.
Table 2 summarizes the changes in the thermal conductivity, thermal diffusivity, and sintered density of AlN specimens with varying TiO2 contents that were processed under the conditions used for Sample #4. As the TiO2 content increased, both thermal conductivity and thermal diffusivity increased, accompanied by a slight increase in the sintered density. Although the AlN lattice disorder caused by Ti substitution in the solid solution can negatively affect the thermal conductivity, the formation of TiN and TiC phases appears to enhance the thermal conductivity, compensating for the effect of disorder. Furthermore, the atomic mobility promoted by the Ti solid solution likely contributes to the increase in the sintered density.21) While liquid-phase sintering via YAG is the primary densification mechanism, the observed increase in the density with higher TiO2 addition suggests that TiO2 also played a role in sintering promotion.
Table 2.
Thermal conductivity and sintered density of AlN samples according to TiO2 content.
Fig. 7 shows a comparison of the color differences between black AlN samples sintered using different crucible materials. Both BN and graphite crucibles were used with lids of the same material to prevent external exposure. It is observed that samples sintered in graphite crucibles showed a light pink surface coloration, whereas those sintered in BN crucibles exhibited a black hue with the darkness intensity proportional to the TiO2 content. In graphite crucibles, the yttrium in the residual YAG phases reacted with graphite at high temperatures (1,850 °C), possibly reducing Y3+ to Y2+. This change in the electron configuration can account for the absorption of specific visible-light wavelengths, leading to the observed pinkish-red color.10) Additionally, carbon decomposition in the graphite crucible at high temperatures may have altered the surface color of the sample.22)
Fig. 8 shows the final black AlN heater specimen for semiconductor photolithography and its fracture-surface microstructure. The heater had a uniform black appearance, and SEM images of its fracture surface revealed a dense microstructure with primarily intergranular fracture behavior. YAG phases, formed by the reaction between Y2O3 and the oxidized (Al2O3) layer on the AlN particles, were mainly observed at triple junctions. During high-temperature sintering, these phases are mostly volatilized, which contributes to the high thermal conductivity of the final material.15)
4. Conclusion
Densified black AlN ceramics were successfully fabricated by adding 1.0 wt% TiO2 to AlN powder containing 3 wt% Y2O3 as a sintering aid. The resulting black AlN exhibited a sintered density of 3.33 g/cm3, thermal conductivity of 162.7 W/m・K, and thermal diffusivity of 64.22 mm2/s. To maximize green density and improve sinterability, the relative amounts of the PVB binder and the PEG plasticizer were optimized during granule preparation. The highest green body relative density of 58% was achieved through uniaxial pressing under the condition of 1.00 wt% PVB and 0.45 wt% PEG with die pressing at 530 kg/cm2.
As the TiO2 content increased, the black coloration of the AlN became more pronounced, with complete blackening observed at approximately 1 wt% TiO2. Thermal conductivity and diffusivity remained stable beyond 0.45 wt% TiO2, indicating saturation in functional improvement. During heat treatment in a nitrogen atmosphere in a vacuum furnace, the use of graphite crucibles interfered with the black coloration owing to the reduction of yttrium in the YAG phase and carbon contamination. By contrast, BN crucibles provide a stable atmosphere conducive to consistent color development.
Fracture surface observations of the sintered samples revealed dense microstructures and predominantly intergranular fracture behavior. Residual secondary phases, particularly YAG, located at the triple-grain junctions, were observed, which are beneficial for achieving enhanced thermal conductivity.