1. Introduction
2. Experimental Procedure
2.1. Chemicals
2.2. Anchoring monoatomic Pd to BaTiO3
2.3. Characterization and piezoelectric activity test
3. Results and Discussion
3.1. XRD characterization
3.2. TEM and HAADF-STEM observations
3.3. XPS characterization
3.4. Piezoelectric properties of the samples
3.5. Degradation of tetracycline
4. Conclusion
1. Introduction
Owing to the rapid industrialization in recent years, the seriousness of environmental water pollution has increased, along with the discharge of toxic and harmful organic pollutants. The contemporary world is being affected by environmental contamination owing to increasing global industrialization, which has resulted in deterioration of the quality of water, air, and soil from the accumulated toxic organic and inorganic compounds.1,2) Antibiotics are chemotherapeutic agents used to treat and prevent bacterial infections. They are widely used by humans owing to their remarkable ability to resist or treat bacterial infections. Tetracycline is a purely natural antibiotic that quickly inhibits the growth of bacteria and interferes with the synthesis of bacterial proteins, thereby killing bacteria at very high concentrations. Frequent and massive use and abuse of antibiotics has led to a sharp increase in the amount of antibiotics in the environment.3,4)
Therefore, further explorations of green environmental protection approaches are required to solve the problems caused by dye wastewater. The application of nanostructured piezoelectric materials in energy conversion is considered a new and cutting-edge technology.5) In 2010, Hong et al.4) first reported the direct conversion of mechanical energy to chemical energy via water splitting by vibrating piezoelectric microfibers. In recent years, piezoelectric catalysis has attracted increasing interest from researchers for environmental remediation; it was found that piezoelectric catalysis takes advantage of the mechanical vibration energy, which is an abundant resource common in daily life instances,6) such as walking7) and river flow.8) Piezoelectric materials9,10) can collect vibrational energy and release large amounts of free electrical charge that react with water, thereby achieving dye decomposition; this process is called piezoelectric catalysis. The potential field resulting from piezoelectric materials can effectively transform mechanical vibration energy to electrical energy.11,12,13,14) It is important to note that the free charges generated during the piezoelectric catalytic process can be easily segregated on the catalyst surface under the influence of a potential field.15,16) Hence, piezoelectric catalysis is a potential technology for improving the separation of photogenerated charges, which offers better catalytic efficiency than photocatalysis.
Barium titanate (BaTiO3 or BTO) is the first ferroelectric ceramic discovered in recent times, which has excellent ferroelectric, dielectric, piezoelectric, and thermoelectric properties; it has been used in a wide range of industrial applications.17,18) Therefore, BaTiO3-based coatings have attracted more attention from researchers in recent years.
Recently, single-atom catalysis has become a popular topic of research because the dispersion promotion of a noble metal via downsizing a nanoparticle to the atomic level can maximally enhance its utilization. Li et al.18) used thermal treatment to promote Ag nanoparticle migration along an external surface and insertion into the tunnels of hollandite-type MnO2 nanorods to effectively catalyze oxidation of HCHO by the high dispersion of the sub-nanosized Ag and even single atoms. However, the decoration of catalysts with highly dispersed noble metals remains a tough task because Ag or Au nanoparticles with high surface energies can easily agglomerate into large particles via Ostwald ripening. Moreover, most noble-metal atoms are lost in the bulk during synthesis through conventional methods, such as coprecipitation or photodeposition, where they are no longer effective as active sites. Some studies have also indicated that tighter contacts can be achieved between the atomic metal and MnO2 than an Ag cluster and MnO2, thereby enhancing the charge transfer of the catalyst for the catalytic reaction.19,20)
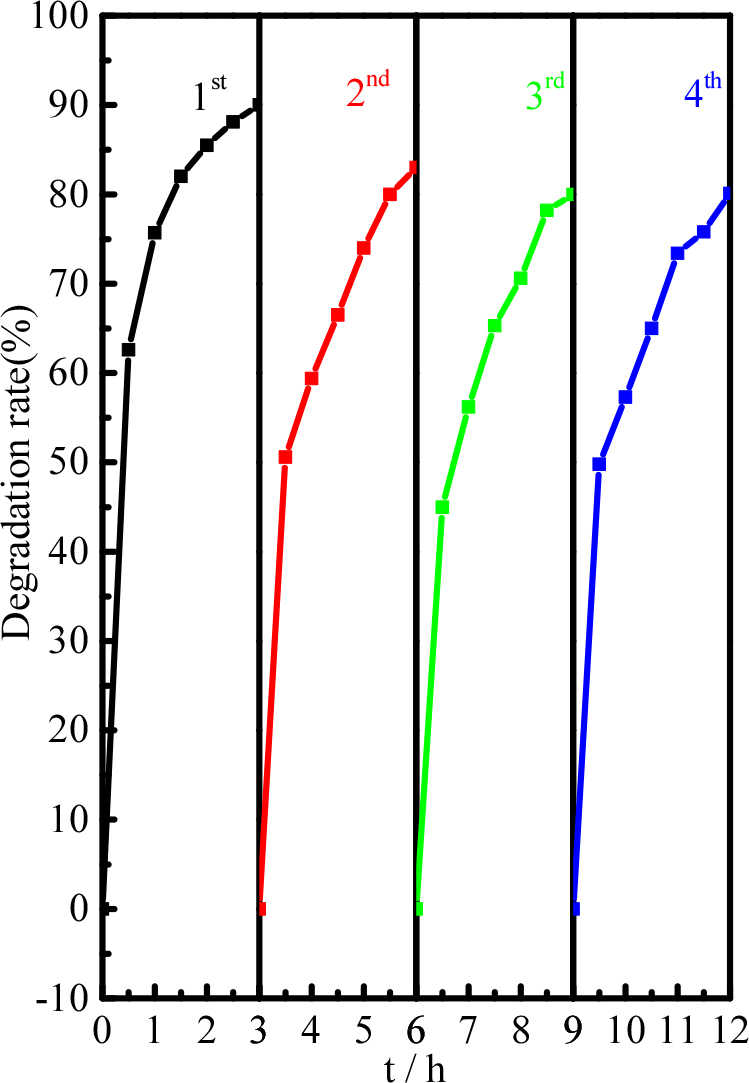
In this study, we designed and fabricated a piezoelectric catalytic system to synthesize a Pd-BaTiO3 composite structure with synergistic catalytic effects using hydrothermal and ultrasonic methods. Under ultrasonic vibrations, this system may be used for catalytic degradation of pharmaceutical pollutants. For a larger piezoelectric potential on the surface of the catalyst, the conduction and valence bands move, which assists in easier and faster migrations of electrons and holes when reacting with dissolved oxygen. The Pd-BaTiO3 nanomaterials show excellent piezoelectric catalytic activities and stabilities during four consecutive piezoelectric catalytic processes.
2. Experimental Procedure
2.1. Chemicals
Barium chloride dehydrate (BaCl2 ∙ 2H2O), tetrabutyl titanate [(C4H9O)4 ∙ Ti], and sodium hydroxide (NaOH) were purchased from Shanghai Silian Chemical Plant, Runjie Chemical Reagent, and Jiangsu Qiangsheng Functional Chemical Co., Ltd., respectively. The oxygen used in the experiments was purchased from Suzhou Jinhong Gas Co., Ltd. The cubic-phase barium titanate (BaTiO3) nanopowder was prepared with the above substances as the raw material. PdCl2 was purchased from Shanghai Zhenxing Reagent Factory, ammonia (NH3 ∙ H2O) was obtained from Wuxi Zhanwang Chemical Reagent Co., Ltd., and tetracycline was purchased from Aladdin. Except for tetrabutyl titanate [(C4H9O)4 ∙ Ti], which was chemically pure, the other reagents used were of analytical grade, and all reagents were used as received. The solvent used in all preparation solutions was 18.2 MΩ ∙ cm deionized (DI) milli-Q water.
2.2. Anchoring monoatomic Pd to BaTiO3
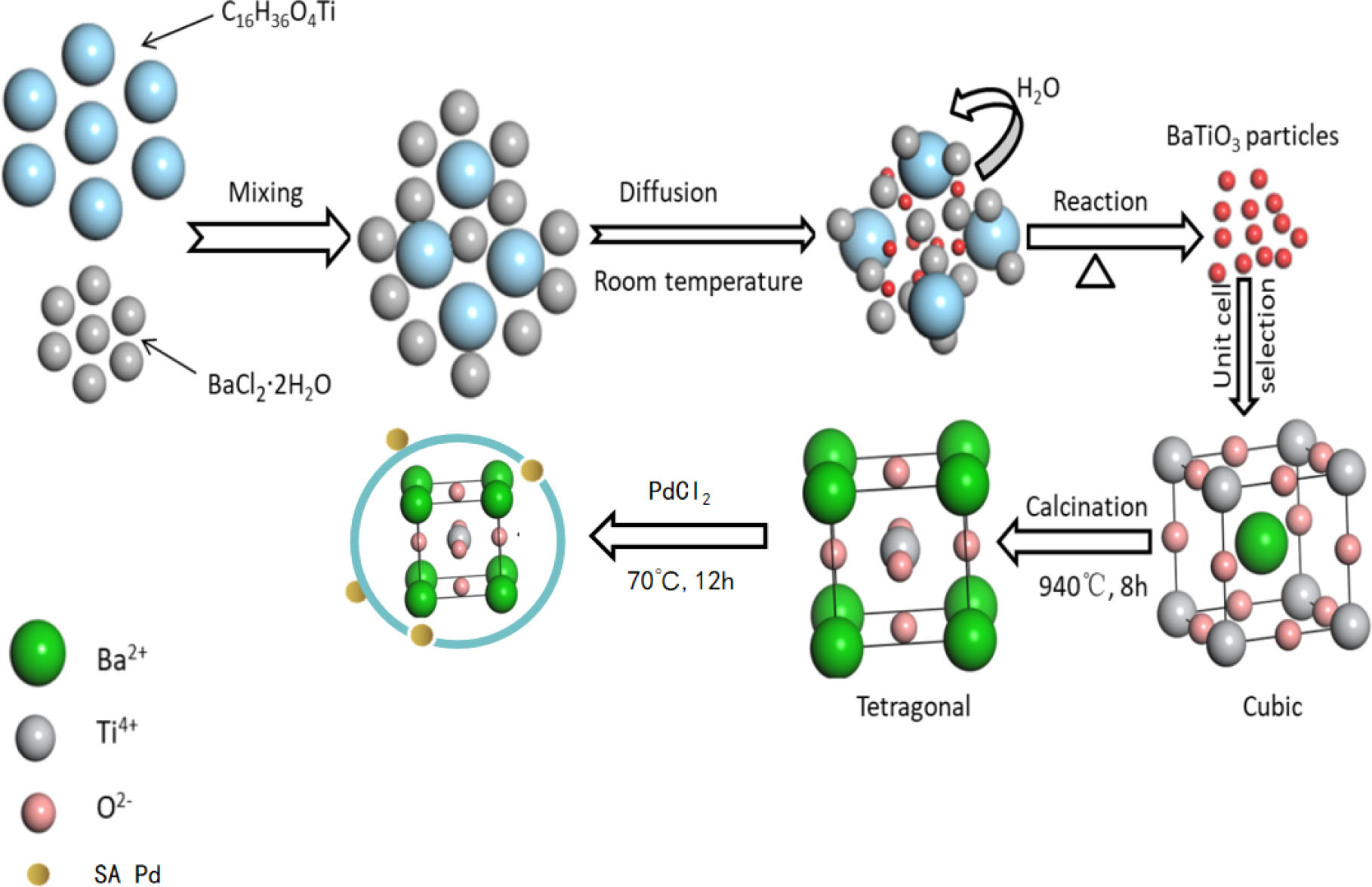
The sample preparation process is shown in Fig. 1. Single-atom Pd-BaTiO3 was prepared as follows. First, about 7.5 mmol BaCl2 ∙ 2H2O was weighed in a 100 mL beaker to which 30 mL DI water was added for ultrasonic dissolution; then, about 2 mL of (C4H9O)4 ∙ Ti was measured using a graduated cylinder and added dropwise to the barium chloride solution under magnetic stirring for 30 min for adequate mixing. Then, about 75 mmol of NaOH was ultrasonically dispersed in 30 mL of DI water, and after dissolution, it was slowly added dropwise to the mixed solution under magnetic stirring; the stirring was continued for 1 h to ensure full reaction. Next, the mixed solution was transferred to a 100 mL hydrothermal autoclave and placed in an oven at 180 °C to react for 16 h. When the reaction was complete and product cooled to room temperature, the reaction solution was vacuum filtered, the sample was washed 2~3 times with DI water and absolute ethanol, and the product was placed in an oven at 70 °C for vacuum drying over 12 h to obtain the cubic BaTiO3 sample. The obtained sample was then placed in a 50 mL crucible, which was placed in a muffle furnace, before heating to 940 °C at the rate of 5 °C/min followed by maintaining at a constant temperature for 8 h. After the product cooled to room temperature, the crucible containing the tetragonal BaTiO3 sample was removed.
In the second set of procedures, the single atom of Pd was anchored on the tetragonal-phase BTO by the thermal equilibrium method. Next, 50 mL PdCl2 was added to ammonia solutions of various concentrations (0.006, 0.008, 0.01, 0.012, and 0.014 mol/L), resulting in blue turbidity; then, the solution was add continuously until the precipitate dissolved completely. Subsequently, a certain amount of the calcined BaTiO3 was added to the solution with stirring for 1 h, and the mixture was poured into a 100 mL autoclave. After the reaction cooled to room temperature, the sample was washed by filtration and placed in a vacuum drying oven at 70 °C for 12 h. The sample was then placed in a crucible and calcined in a muffle furnace at 200 °C for 2 h. The resulting single-atom Pd-anchored BTO sample was subjected to electro-Fenton catalytic degradation and characterization.
2.3. Characterization and piezoelectric activity test
High-angle annular dark field-scanning transmission electron microscopy (HAADF-STEM) was performed on THEMIS Z (FEI) equipped with double aberration correctors and a cold field emission gun, which was operated at 80 kV. Energy dispersive X-ray (EDX) mapping was operated at 200 kV. X-ray diffraction (XRD) was performed with an X’Pert-Pro MPD X-ray diffractometer (Panalytical, Netherlands). The X-ray source emitted Cu-Kα radiation with a wavelength of 0.154 nm at a tube voltage of 40 kV and a tube current of 40 mA. The monochromatic Al-Ka source (mono Al-Ka) was used as the X-ray source to analyze the sample via X-ray photoelectron spectroscopy (XPS) to study the chemical composition of the catalyst surface. The microstructures of the BTO samples anchored with single-atom Pd were studied by high-resolution transmission electron microscopy (HRTEM).
To test the degradation efficiency of the piezoelectric catalyst, tetracycline was therefore used as the target pollutant. In a typical measurement, 0.1 g of the Pd-BaTiO3 sample was dispersed in 50 mL of 8 mg/L tetracycline solution. Oxygen was passed through the solution for 20 min to combine with the oxygen in the solution. The solution was then stirred for 5 min so that the catalyst could reach the adsorption equilibrium. Ultrasonic vibration was used to maintain continuous oxygen flow during the degradation process. The ultrasonic vibration frequency was 40 kHz, and the power of the ultrasonic cleaning machine was 100 W. To study the degradation kinetics of tetracycline, 3 mL of the mixed solution was collected regularly and centrifuged to obtain the supernatant using a Beijing Puxi TU-1901 spectrophotometer and the UV-visible absorption spectrum to determine the concentration of tetracycline in the centrifugal fluid. In addition, to evaluate the cyclic stability of the catalyst for the degradation of tetracycline, the catalyst (Pd-BaTiO3) used in the experiment was recovered and subjected again to catalytic degradation experiments.
3. Results and Discussion
3.1. XRD characterization
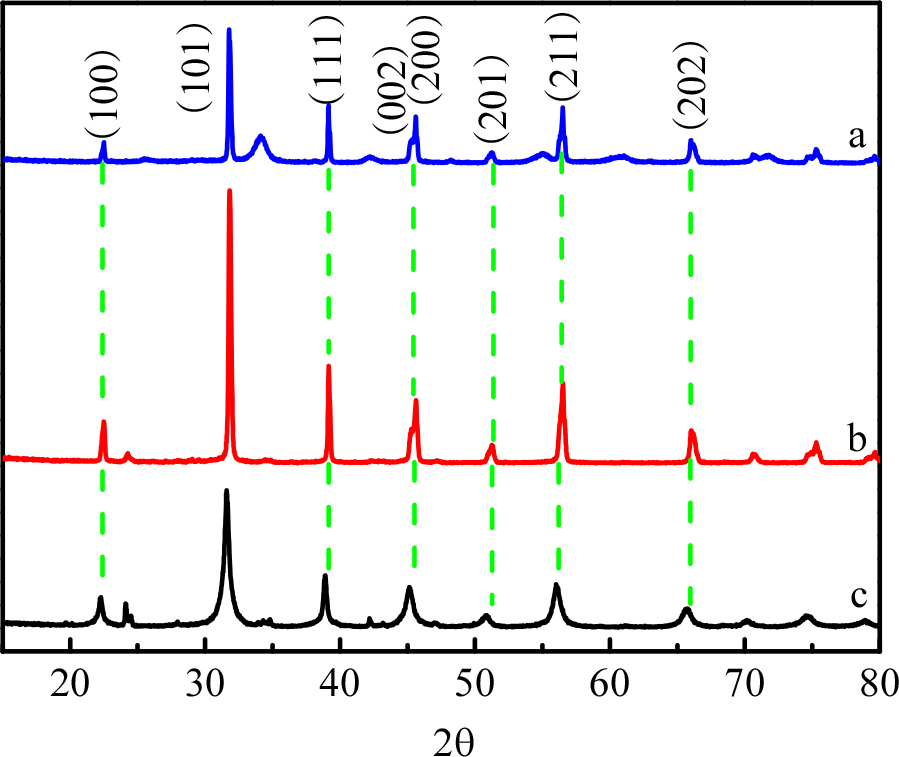
Fig. 2(a-c) show the XRD patterns of BaTiO3 loaded with monoatomic Pd, BaTiO3 before and after calcining, and the typical cubic-phase BTO (PDF#75-0212, space group: Pm-3m, a = b = c = 4.016 Å, a = b = g = 90°). From the (100), (101), (111), (002), (200), (201), (211), (202) and other sharp characteristic peaks, it is noted that the synthesized sample is a perovskite-type cubic BaTiO3. By comparing curves b and c, it is observed that curve b has a bifurcation peak at 2q = 45.2°. It is seen that after calcination, the BaTiO3 crystal changes from cubic to tetragonal phase (PDF#75-2121, space group: P4/mm, a = b = 3.989 Å, c = 4 Å, a = b = g = 90°); from comparison of curves a and b, it is observed that the crystal structure of Pd-BaTiO3 remains unchanged at the tetragonal phase after being calcined once again.21)
3.2. TEM and HAADF-STEM observations
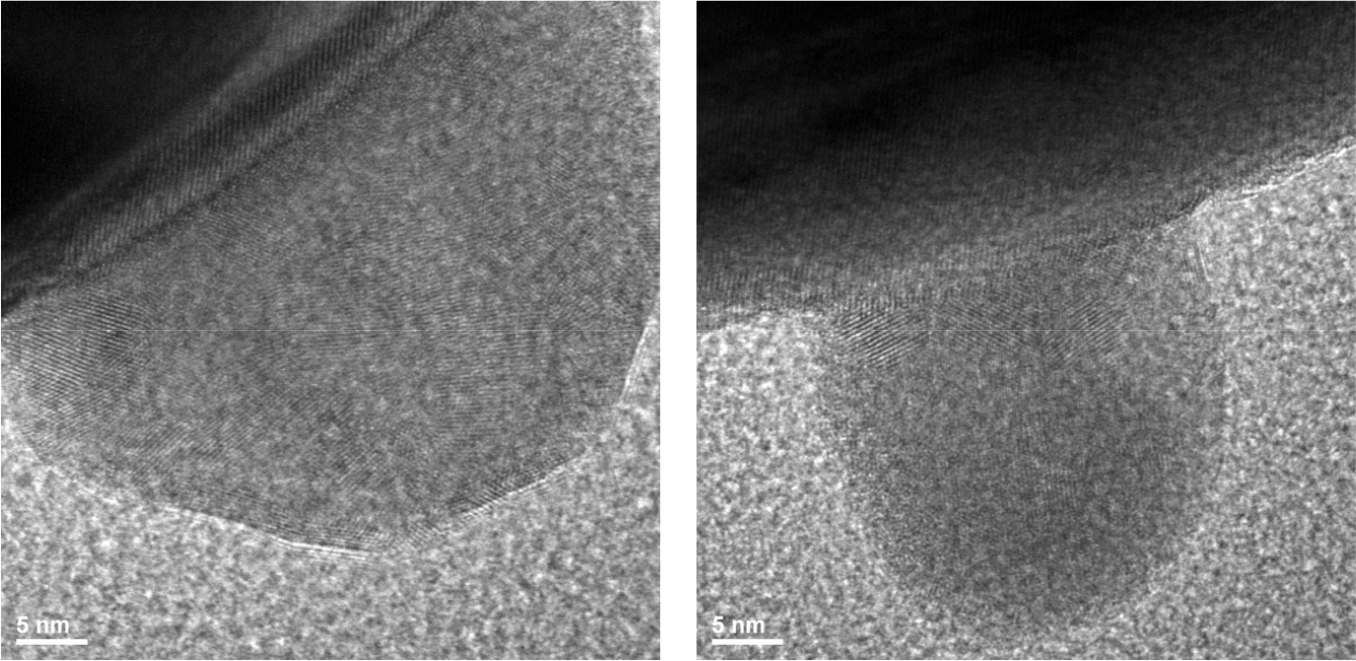
Transmission electron microscopy (TEM) provides further information on the crystallography of BTO. As shown in Fig. 3(a), BaTiO3 nanoparticles are crystallized well, with a size of about 200 nm; obvious regularly arranged lattice fringes are observed in the HRTEM image throughout the single crystal grain. The lattice fringe spacing of the tetragonal BaTiO3 is measured as 0.28 nm, which corresponds to the standard PDF card (JCPDS Card No. 79-2264) (101) crystal plane fringe spacing.22)
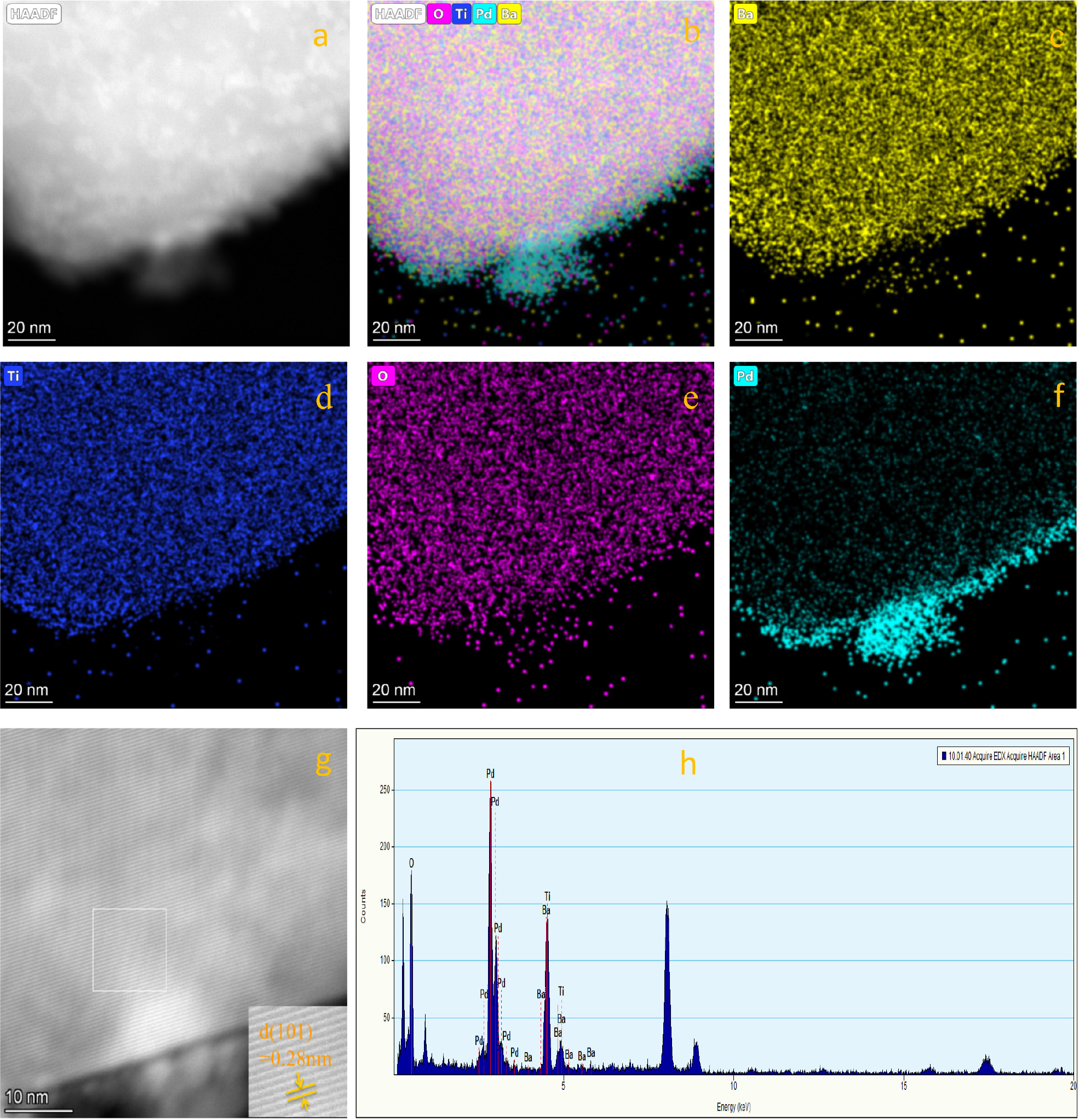
In addition, the morphology and composition of Pd-BaTiO3 were further determined by HAADF-STEM and EDX techniques. As shown in Fig. 4, based on the selected-area electron diffraction patterns [Fig. 4(b, c)], it is determined that the distance between the (001) crystal planes (d001 = 0.402 nm) is slightly larger than that of the (100) planes (d100 = 0.393 nm).23) The divergence of the lattice parameters confirms the tetragonal symmetry of the perovskite structure. The HAADF-STEM image [Fig. 4(d)] and corresponding element mapping image [Fig. 4(e-h)] clearly show that Ba (red), Ti (green), O (blue), and Pd (yellow) are evenly distributed in the nanostructures. On the surface, this indicates that the single-atom clusters Pd was successfully introduced into BaTiO3. The atomic ratios of O, Ti, Ba, and Pd in the monoatomic Pd-BaTiO3 are 61.45, 24.31, 13.94, and 0.3 %, respectively. The ratio of metals (Ba + Ti + Pd) to oxygen is 2:3.18, which is close to 2:3, and the atomic stoichiometric ratio is reasonable.
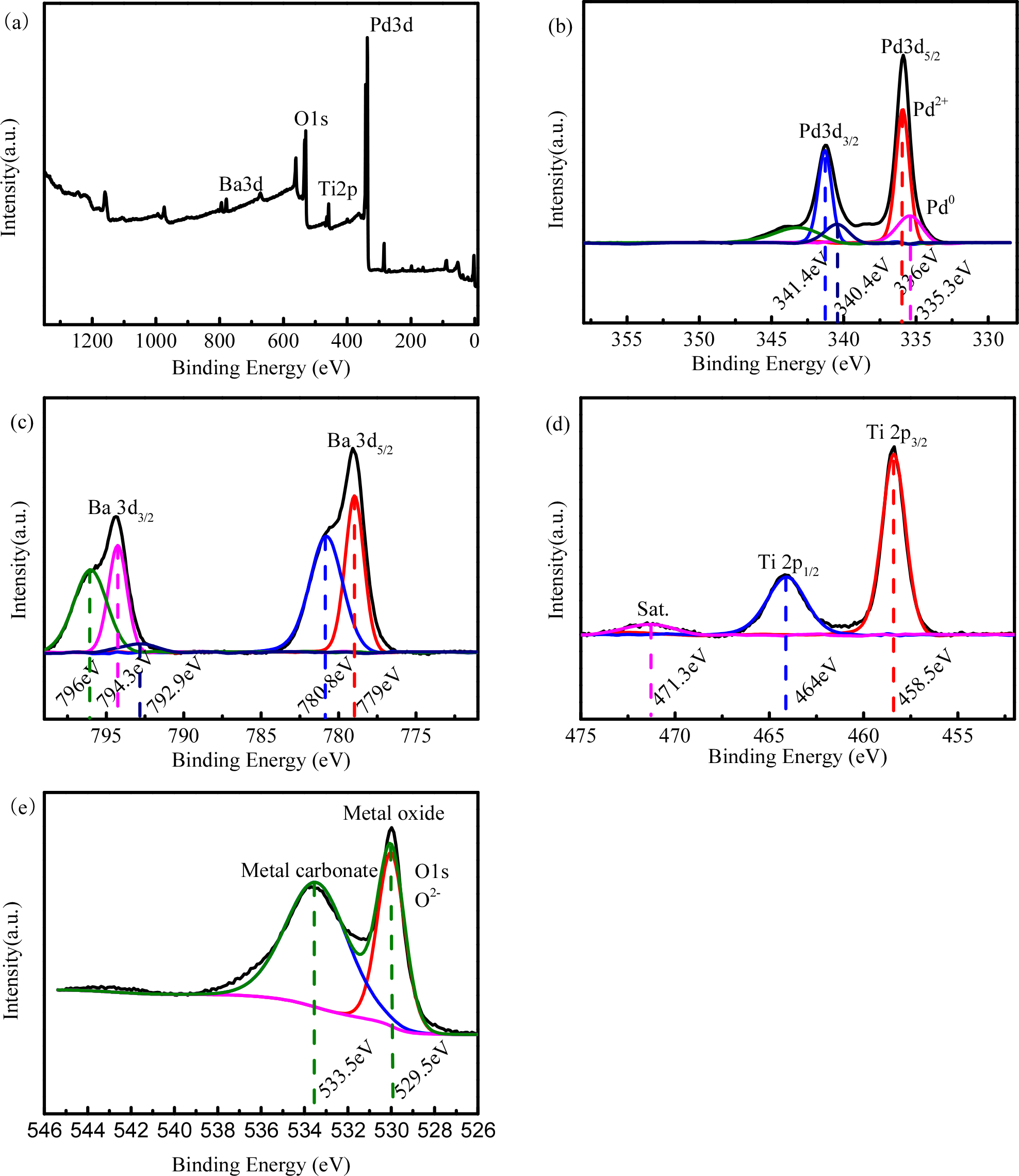
3.3. XPS characterization
The XPS data show that Ba 3d, Ti 2p, Pd 3d, and O 1s are present in Pd-BaTiO3. The high-resolution XPS spectra of each of the elements are shown in Fig. 5(a-d). From the figure, it is seen that the sample contains Ba, Ti, O, and Pd, which are consistent with the EDX results in Fig. 4(h). Fig. 5(a) shows the high-resolution XPS image of Ba 3d; the electron affinity at 778.4 eV is the characteristic peak of Ba 3d5/2, and the satellite peak electron affinity is 780 eV. Fig. 5(d) shows that the characteristic peaks at 458.5 and 464.0 eV are from Ti 2p. Fig. 5(b) is the high-resolution electron energy spectrum of Pd 3d; in the figure, the peaks of Pd 3d5/2 and 3d 2p3/2 are located at 336.0 and 341.4 eV, respectively. No satellite accompanying peaks appear, indicating that the sample contains monovalent Pd species, Pd0 or a mixture of the two. The characteristic peaks of O 1s are seen at 529.5 eV.24,25)
3.4. Piezoelectric properties of the samples
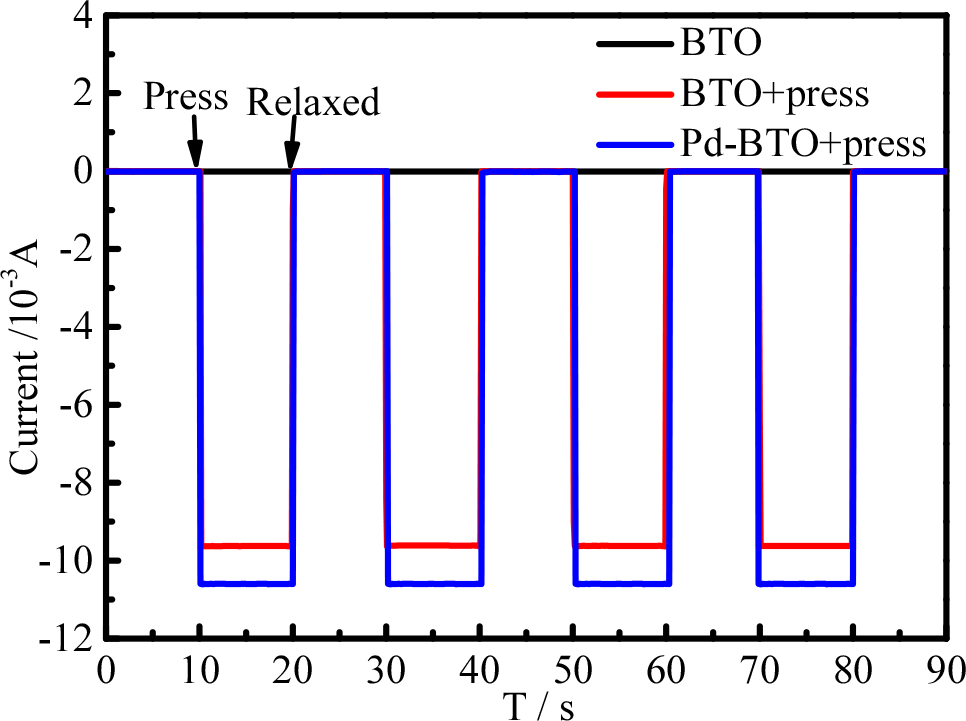
BaTiO3 without pressure, BaTiO3 with pressure, and Pd-BaTiO3 were used in a computer-controlled electrochemical workstation (CHI660C) for transient the piezoelectric current response tests to investigate the reasons for the observed improvement in the catalytic activity of the Pd-BaTiO3 binary composite. In order to confirm the piezo-electric effect, the piezo-currents of the BaTiO3 and Pd-BaTiO3 were measured by exerting a pressure of 1.1 ± 0.1 kg cm2 between the ITO electrodes. As seen in Fig. 6, the piezoelectric current density of the prepared Pd-BaTiO3 binary composite is significantly higher than that of the single BaTiO3, implying that the binary composite material has a higher carrier separation efficiency and better electron mobility such that it can be used as an efficient piezoelectric synergistic active catalyst.
3.5. Degradation of tetracycline
3.5.1. Comparison of the degradation rate
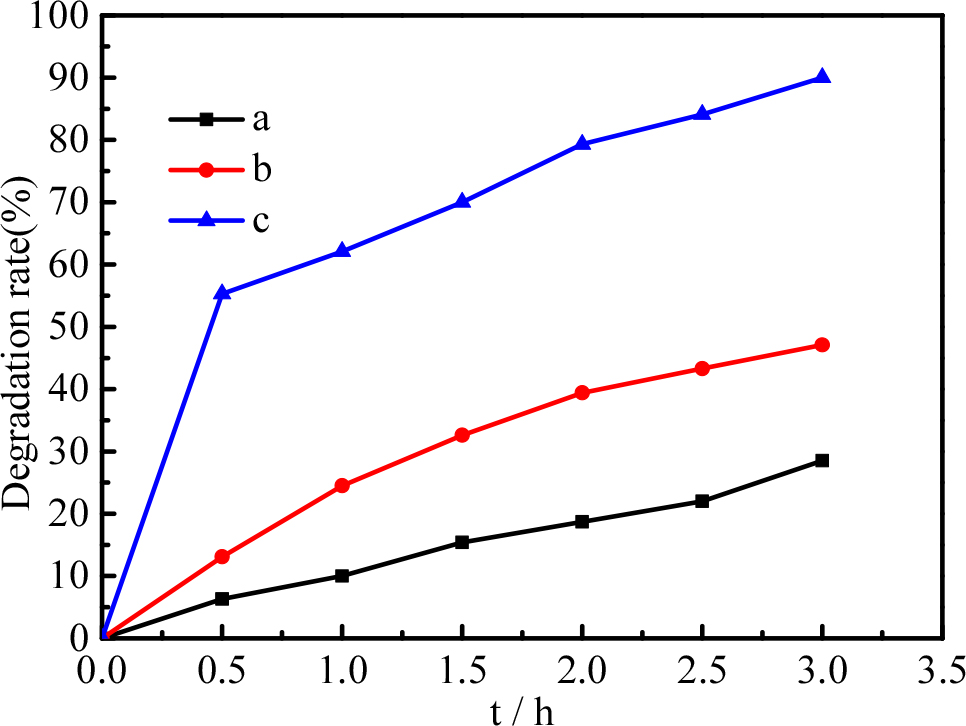
To prove the synergistic effects of Pd-BaTiO3 composites on the catalytic capacity, the degradation of tetracycline solution is investigated, as shown in Fig. 6. For comparison, the adsorption ability of pure BaTiO3 as well as the catalytic abilities of pure BaTiO3 and Pd-BaTiO3 are tested under the same experimental conditions. In Fig. 7, line (a) is the adsorption ability of pure BaTiO3 under dark conditions, (b) is degradation of tetracycline by pure BaTiO3 under ultrasonic vibration, (c) is degradation of tetracycline by the Pd-BaTiO3 composite under ultrasonic vibration. Predictably, limited degradation degrees of the tetracycline solution are observed for the pure BaTiO3, which shows only adsorption ability [Fig. 7(a)] or under ultrasonic vibration [Fig. 7(b)].
As indicated in Fig. 7, about 28.5 % of the tetracycline was adsorbed by pure BaTiO3 under dark conditions [Fig. 7(a)]; under ultrasonic vibration [Fig. 7(b)], the degradation rate of tetracycline increased to 48.2 % by the pure BaTiO3. After doping with Pd, the piezocatalytic oxidation activity of the Pd-BaTiO3 composite increased from 28.5 to 91.3 %. Owing to the synergistic effects of Pd and BaTiO3, the Pd-BaTiO3 composite showed the best degradation effects under ultrasonic vibration.
Pd-BaTiO3 (c) shows a rapid increase in degradation rate until the first 30 minutes, and after that, the slope of the degradation rate appears to be not significantly different from other samples (a, b), this is because the concentration of tetracycline is low, the concentration is also can affects the effectiveness of the degradation.
3.5.2. Effects of Pd content
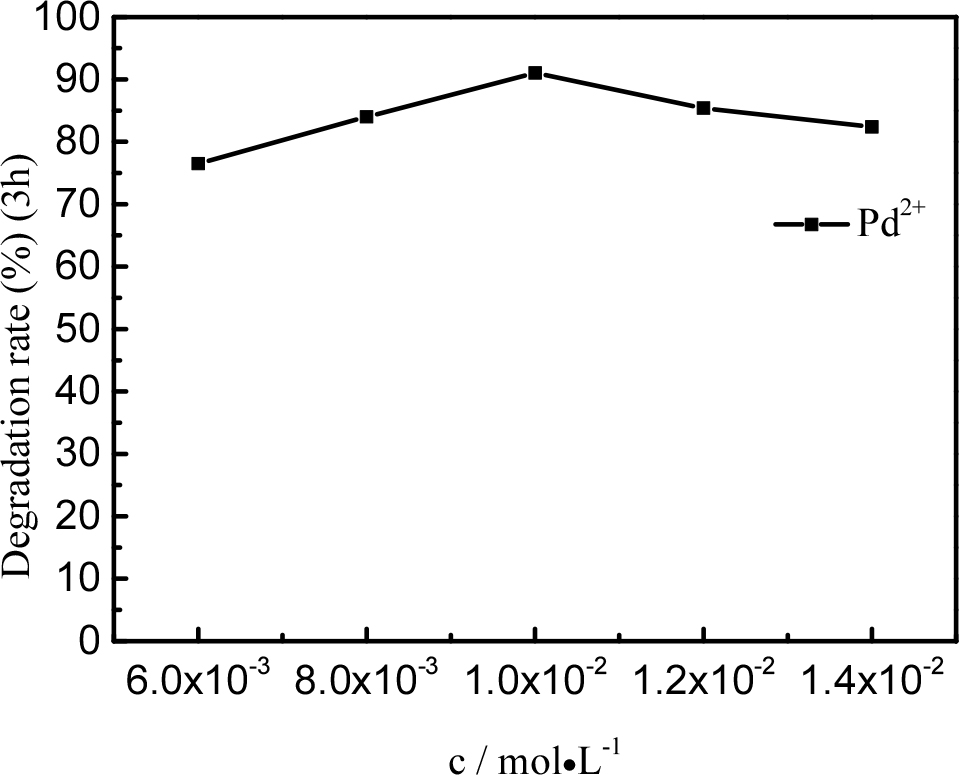
Fig. 8 shows the degradation of the tetracycline solution with samples containing different amounts of Pd. We prepared used PdCl2 solutions with different concentration (0.006, 0.008, 0.01, 0.012, and 0.014 M). The degradation efficiencies of tetracycline were significantly influenced by the amount of Pd, which increased from 75.6 to 91.3 % after ultrasonication for 7 h when the Pd dosage was correspondingly increased from 0.006 to 0.01 mol/L. The degradation efficiency of tetracycline decreased from 91.3 to 78.7 % after ultrasonication for 7 h when the Pd dosage was increased from 0.01 to 0.014 mol/L. This is because the more content of Pd, the aggregation effect of Pt can decrease the degradation effect.
To evaluate the cycle stability of the piezoelectric catalyst, the recovered Pd-BaTiO3 was used in degradation experiments (Fig. 9). After four cycles of the degradation experiments, the degradation efficiency of tetracycline hardly changed, indicating that the Pd-BaTiO3 piezoelectric catalyst has high cycle stability.
3.5.3. Mechanism discussion
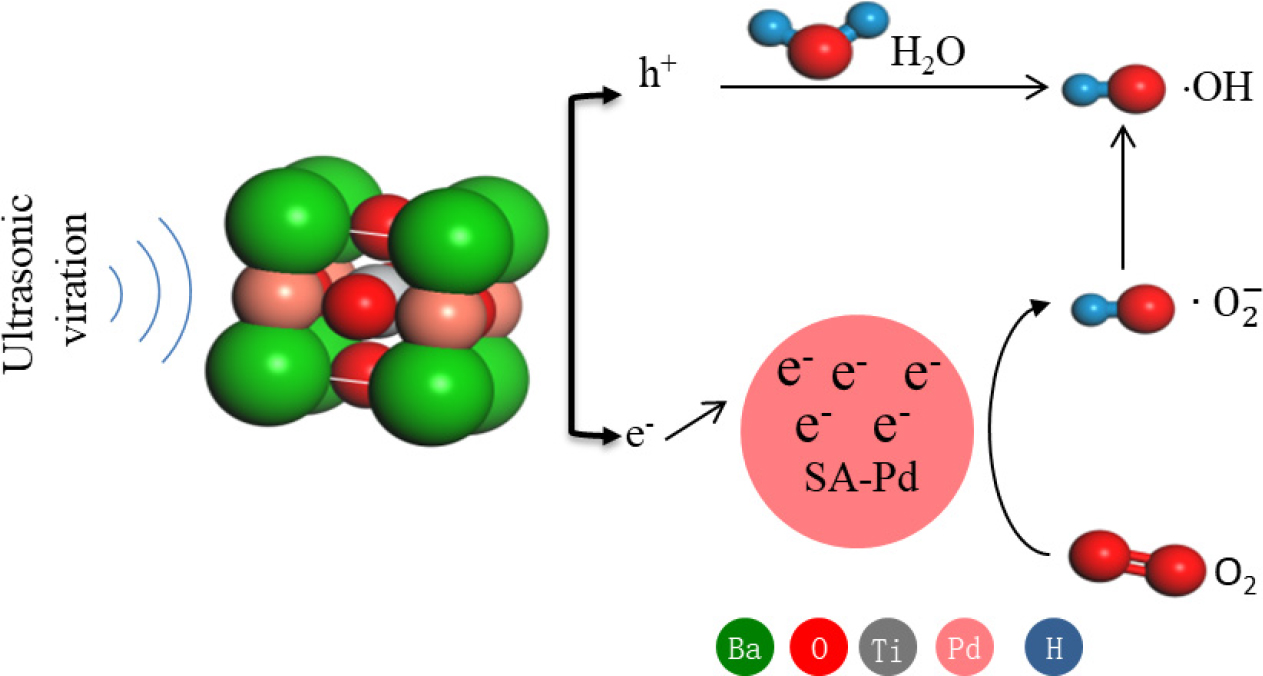
The piezoelectric properties owing to the non-centrosymmetric nature of the material (i.e., crystals not having a center of symmetry in their structure) results in electric dipoles within the material. In most crystals, the unit cell is symmetrical but not in the piezoelectric crystals. Despite their asymmetric arrangement of atoms in the lattice, the piezoelectric crystals are electrically neutral because of canceling of the positive charges by their neighboring negative charges. However, if a piezoelectric crystal is subjected to stress by squeezing or stretching, the atoms are displaced from their original positions, which results in a net electrical charge for the entire crystal, based on which net positive and negative charges appear on the opposite outer faces of the crystal.26,27) When stress is applied to the unit cell, the original atomic positions of the O2- anions and Ba2+ cations are relatively displaced, which would lead to gaining a dipole moment in the unit cell. Hence, the crystal gains a piezoelectric potential by the collective induced polarization of charges in entire unit cells in the crystal, which is the piezoelectric potential (piezopotential). The piezoelectric crystal is excited by mechanical stress, and the positive and negative potentials are created on opposite surfaces of the polarized crystal, which are called positive polarization domains (PPDs) and negative polarization domains (NPDs), respectively; then, the piezogenerated built-in electric field can be used to separate the carriers for catalytic degradation. The generation of free charges and their participation in the redox reactions in the piezocatalytic process are shown in reactions Eq. (1) to Eq. (11).
Anode (negatively charged sides of the BaTiO3):
Overall:
Cathode (positively charged sides of the BaTiO3):
Overall:
Tetracycline decomposition:
Accordingly, an inferred schematic diagram is proposed to reveal the variation of charge carrier transport processes after introduction of Pd into the BaTiO3 crystal face. Schematic diagrams of the charge carrier separation and transport processes at the interfaces of the Pd-BaTiO3 composites are shown in Fig. 10. In the Pd-BaTiO3 system, under ultrasonic vibrations, the BaTiO3 primary cell is doubled in the direction of XYZ to build a 2 × 2 × 2 supercell; when the BaTiO3 suffers an external force, a nonzero dipole moment is propagated, and the stretched and compressed side surfaces exhibit positive and negative piezopotentials, respectively, thereby forming a built-in electric field across the cross section of the c-axis-oriented BaTiO3. Because the electric field is formed by the nonmobile, nonannihilative ionic charges, the polarization charges (static and nonmobile) remain in the crystal as long as the stress remains.28,29) In the piezoelectric field, the PPD draws compensating electrons toward the surface, decreasing the bandgap, whereas the NPD repels electrons from the surface, increasing the bandgap under a compressive strain.
To further prove the decisive effects of the charge density redistribution inside the BaTiO3 nanoparticles, Pd is selectively deposited onto the BaTiO3 crystal face. As shown in Fig. 10, plenty of piezo-induced electrons escape to the Pd-doped BaTiO3 interfaces owing to the excellent conductivity of Pd, so the charge carrier diffusion channels form at the Pd-BaTiO3 interfaces. This in turn restrains the piezo-induced charge recombination and strengthens the piezocatalytic oxidation activity ultimately.
4. Conclusion
In summary, monoatomic clusters Pd nanoparticles anchored on tetragonal BaTiO3 were prepared, and their piezoelectric catalytic activity was studied through degradation of tetracycline solution. Under mechanical vibrations, the nanomaterial showed a tetracycline decomposition rate of ~95 % within 7 h, which is much higher than the degradation rate of 56.7 % observed for pure BaTiO3. Plenty of piezo-induced electrons escape to the Pd-doped BaTiO3 interfaces owing to the excellent conductivity of Pd, so single-atom clusters Pd promotes separation of the piezo-induced electrons to achieve synergistic catalysis. The prepared Pd-BaTiO3 nanomaterials also show excellent piezoelectric catalytic activities and stabilities over four consecutive piezoelectric catalytic processes.