1. Introduction
2. Experimental Procedure
2.1. Extraction
2.2. Preparation of denture base resin coated with Peony extract
2.3. Measurement of microhardness
2.4. Water contact angle measurement
2.5. Measurement of color difference
2.6. Polyphenols content measurement
2.7. Evaluation of cytotoxicity
2.8. Antimicrobial tests
2.9. Statistical analysis
3. Results and Discussion
4. Conclusions
1. Introduction
With an aging population, more people are using dentures.1,2) Polymethyl methacrylate has been widely used as a denture base resin.3) However, bacteria easily adhere to and colonize the denture base surface, which can lead to the chronic inflammatory condition called denture stomatitis.4,5) Denture stomatitis may cause inflammatory redness, edema, and granular hyperplasia in the mouth.1) Various factors are known to be involved in denture stomatitis,6) where bacterial adhesion is an important step in colonization and infection progression.7,8) Among the common pathological microorganisms in the oral cavity, Streptococcus mutans (S. mutans) is the primary causative agent of plaque formation and is involved in initial plaque formation, while Candida albicans (C. albicans) is the primary causative agent of denture stomatitis.9-11)
Therefore, preventing the onset of denture stomatitis is important. These pathogens are currently treated using antimicrobial substances such as chlorhexidine and tetracycline; however, these drugs have various side effects such as tooth discoloration, oral mucosal epithelial exfoliation, and the formation of drug-resistant bacteria.12,13) Thus, many studies have aimed to develop safer antimicrobial agents from natural products to avoid the side effects of existing antimicrobial agents.14,15)
Various natural extracts have been studied for their potential application as antimicrobial agents.14-16) Among them, Peony is an ornamental and medicinal plant, and is known to have excellent antimicrobial, anti-inflammatory, and anti-ulcer pharmacological functions.16,17) Additionally, it contains antioxidants that promote biological activity.18) Plant materials with increased biological activity significantly impact the production process and stability of the final product.19) Although several studies have investigated the antibacterial properties of natural extracts, few studies have investigated product stability and characteristics for application to dental materials.15,20)
Thus, the purpose of this study was to investigate the physical, biological, and antimicrobial properties of a denture base resin coated with Peony extract to prove its potential as an antimicrobial denture base resin containing a natural product. The null hypotheses of this study were that denture base resin coated with Peony extract does not result in significant differences in the: (1) surface properties, (2) biocompatibility, and (3) antimicrobial activity compared to the control.
2. Experimental Procedure
2.1. Extraction
Peony (purity 99.7 %) planted in the Sobaek Mountains in Gyeongsangnam-do, South Korea, was purchased from a commercial supplier. The purchased sample was compared to a standard herbal sample supplied by the National Institute of Food and Drug Safety Evaluation. To obtain the Peony extract, 500 g of Peony leaves were crushed and placed in a solution of 70 % methanol and extracted at 25 ± 1 °C for 24 h. The extract was filtered (Filter paper #2, Whatman, Maidsone, UK) and then concentrated at room temperature using a vacuum evaporator (Vacuum Evaporator, ETELA, Tokyo, Japan). Subsequently, the concentrated extract was pulverized using a freeze dryer (Freeze Dryer, Ilshin Lab, Kyeonggi-do, Korea) for 48 h. To prepare various concentrations of the Peony extract, the pulverized powder was added to dimethyl sulfoxide (DMSO; Amresco, VWR Life Science, MO, USA) at concentrations of 0, 200, 400, and 600 µg/Ml.14)
2.2. Preparation of denture base resin coated with Peony extract
Tokuyama Rebase II (Tokuyama Dental, Tokyo, Japan) was placed on a slide glass, and a polyethylene film was set on top. The resin was mixed in a Teflon mold (10 mm × 10 mm × 0.1 mm) as directed by the manufacturer. Then, the polyethylene film was put up again, and the slide glass was covered and cured for 5 min. Concentrations of 200, 400, and 600 µg/mL of the freeze-dried extract powder were mixed in Plaquit solution (Dreve dentamid GmbH, Unna, Germany) and dissolved with a stirrer. Then, 10 µL of the mixture was thinly spread with a microbrush on a prepared resin specimen. The resin specimen of 0 µg/mL concentration of the freeze-dried extract powder was coated with pure DMSO. The surfaces were irradiated with light at a wavelength of 300~400 nm for 10 min using a photopolymerizer system (Visio Beta Vario, 3M, USA). All experiments were repeated five times with five specimens per group.
In the physical property evaluation experiment, parameters such as microhardness, contact angle, and color difference were evaluated in five groups: an uncoated group and 0, 200, 400, and 600 µg/mL. Cytotoxicity evaluation, antimicrobial test, and polyphenol content measurement were also conducted in four groups (0, 200, 400, and 600 µg/mL). All experiments were repeated five times with five specimens per group.
2.3. Measurement of microhardness
The Vickers hardness was measured using a microhardness tester (Dmh-2; Matsuzawa Seiki, Tokyo, Japan) to confirm the difference in hardness between the experimental and control groups. A load of 0.09 MPa was applied to the specimen surface for 20 s with a diamond pyramid indenter with a face angle of 136°. The measurements were repeated three times per specimen.
2.4. Water contact angle measurement
The water contact angle of each specimen was measured using a Phoenix 300 (SEO, Suwon, Korea) to determine the change in the hydrophilicity of the surface after coating. The water contact angle was immediately measured after dropping 5 µL of distilled water, and the difference in the water contact angle was confirmed for each experimental group.
2.5. Measurement of color difference
To determine the color difference in the specimens, color tone measurements were performed using spectrophotometry (CM-3500d; Minolta, Kyoto, Japan). A white plate was used as the standard; the L*, a*, and b* values of each specimen were calculated to obtain the color difference value ΔE* as follows.
2.6. Polyphenols content measurement
The polyphenols content was measured using a solution obtained by eluting the specimen in distilled water for 7 d at 37 °C. For this purpose, 650 µL of distilled water was added to 50 µL of the sample solution. Then, 50 µL of the Folin-Denis reagent was added, and the resulting reaction was allowed to proceed at room temperature for 3 min. Next, 100 µL of saturated 10 % Na2CO3 solution was added, followed by adding 150 µL of distilled water to adjust the final volume to 1 mL, and the solution was mixed. After allowing the reaction to proceed for 1 h in a constant temperature water bath in a dark room at 37 °C, the absorbance was measured at 725 nm using ultraviolet-visible (UV-vis) spectrometry (X-ma 1200 Spectrophotometer, Human, Korea). The polyphenols content was calculated from a standard curve that was drawn using standard gallic acid (Sigma Aldrich, USA).
2.7. Evaluation of cytotoxicity
2.7.1. Sample preparation
To evaluate the cytotoxicity, the specimen was eluted in a solvent (i.e., cell culture medium) at 37 °C for 24 h with an elution ratio of 3 cm2/mL according to ISO 10993-11. The obtained eluate was diluted 2× and 4× in the solvent. Therefore, the substances used in the MTT assay were the eluate stock solution and diluted solutions.
2.7.2. MTT assay
The cytotoxicity of the eluate from the specimen was evaluated according to the MTT cytotoxicity test method specified in ISO 10993-5. The number of L929 cells per well was adjusted to 1 × 104, and then 100 µL of the cell solution was dispensed into wells and cultured for 24 h. After incubation, 100 µL of the eluate solution was applied to the cells for 24 h. After 24 h, the solution was removed and washed with 100 µL of Dulbecco’s phosphate-buffered saline (DPBS; Gibco BRL, Life Technologies, NY, USA). The DPBS was then removed, and a culture solution containing 1 mg/mL of MTT (3-[4,5-dimethylthiazol-2-yl]-2,5-diphenyltetrazolium bromide) formazan; Sigma, UK) was added at a concentration of 50 µL per well, followed by further incubation for 2 h. To dissolve the formed MTT, 100 mL of isopropanol (Sigma, UK) was added at 100 µL/well and reacted for 20 min. Then, the absorbance was measured and analyzed at 570 nm using spectrophotometry. The results were expressed as a percentage by standardizing the MTT reduction rate of the control group to 100 %.
2.8. Antimicrobial tests
2.8.1. Sample preparation
To evaluate the antimicrobial activity of the resin specimen coated with the antimicrobial Peony extract, S. mutans (ATCC 25175) and C. albicans (ATCC 10231) were used. S. mutans and C. albicans were respectively inoculated in a brain heart infusion (Becton Dickinson and Co., Sparks, MD, USA) and yeast mold (Becton Dickinson and Co., Franklin Lakes, NJ, USA) liquid medium and then incubated at 37 °C for 24 h.
2.8.2. Growth inhibitory effect
The liquid medium dilution method was used to analyze the growth inhibitory effect of the experimental group coated with the antimicrobial agent. The specimen was immersed in 600 µL of phosphate-buffered saline and eluted for 24 h. One mL of bacterial suspension has a cell density of 1 × 105 cells/mL. The bacterial culture was diluted to obtain the OD600 value of 0.4~0.6. The eluate and bacterial culture were mixed in a 1:1 ratio, and the absorbance was measured at 600 nm using an ELISA reader (Epoch, BioTeck, Winooski, VT, USA) at 24 and 48 h after incubation in an incubator at 37 °C.
2.9. Statistical analysis
For statistical analysis, SPSS ver. 21.0 (IBM Co., Armonk, NY, USA) was used. A one-way analysis of variance was performed to determine the significant difference between each experimental group. Further, a Tukey’s post-hoc test was performed. The statistical significance level was set to 0.05.
3. Results and Discussion
In this study, surface characterization of a denture base resin coated with Peony extract (as an antimicrobial agent) was performed, and its efficacy in terms of cell cytotoxicity and antimicrobial properties was evaluated. The control was an uncoated resin, and the experimental groups were resins coated with a dimethyl sulfoxide (DMSO) solution with 0, 200, 400, and 600 µg/mL of concentrated Peony extract.
As shown in Fig. 1, the hardness difference between the uncoated and coated specimens is statistically significant (p < 0.05), whereas the differences between the experimental groups are not significant (p > 0.05). These results imply that Peony extract does not adversely affect the surface hardness of the denture base resin and exhibits stable surface characteristics.
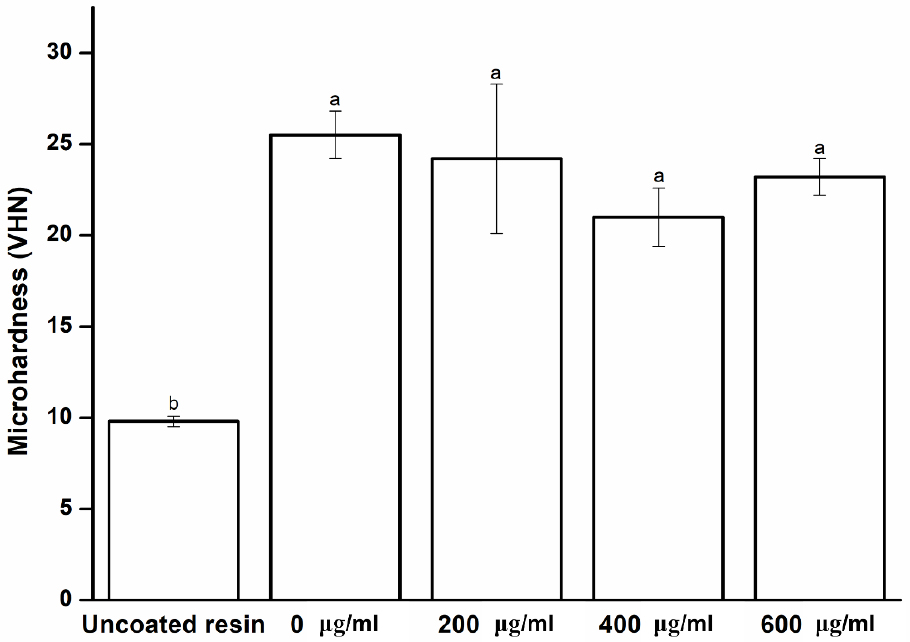
Fig. 1
Microhardness of experimental and control groups. Each value represents the mean of five measurements, and the error bar shows the standard deviation of the mean value. The lowercase letters, a and b, indicate no significant differences between the values of the experimental and control groups (p > 0.05).
The coating material must have a good crack resistance and stable mechanical and physical properties while being resistant to the aqueous conditions in the oral environment.21) Previous studies have suggested that maintaining surface wettability is crucial for good denture retention.22) The water contact angle was measured to confirm the chemical changes of the surface of a specimen coated with Peony extract.11) As shown in Fig. 2, compared to the uncoated specimen, the coated specimens have a lower water contact angle (p < 0.05), indicating that the coating provides a hydrophilic resin surface, which has been shown to reduce the adhesion of microorganisms.11,23) Furthermore, the group with and without the antimicrobial coating does not show a significant difference (p > 0.05). Although the contact angle is lower in the groups with the antimicrobial coating, this difference is not statistically significant (p > 0.05), indicating that its surface becomes more hydrophilic.
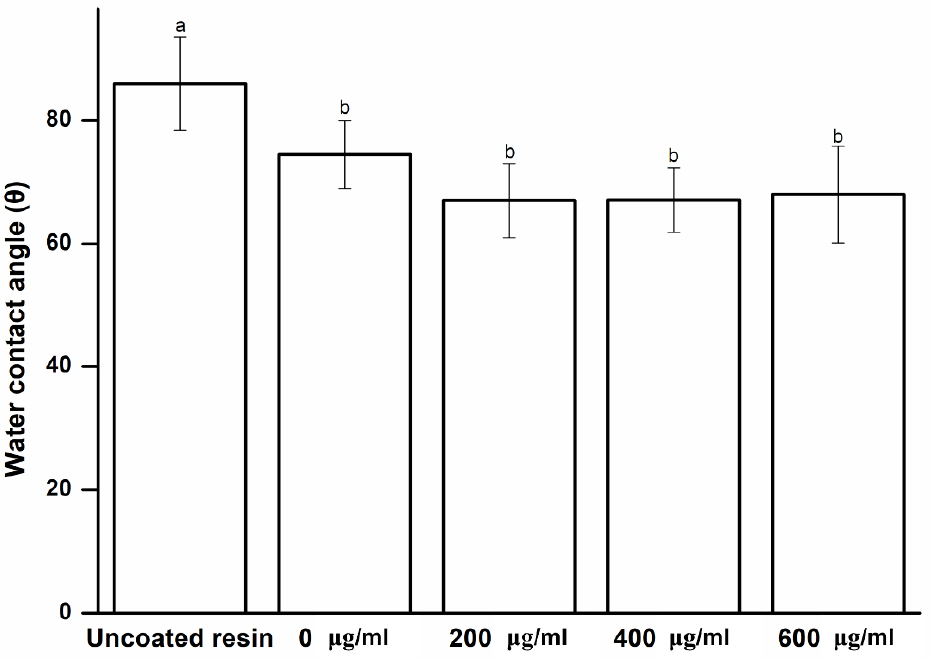
Fig. 2
Water contact angle of experimental and control groups. Each value represents the mean of five measurements, and the error bar shows the standard deviation of the mean value. The lowercase letters, a and b, indicate no significant differences between the values of the experimental and control groups (p > 0.05).
Color stability is essential for all dental materials24) because it strongly influences dental aesthetics.25) Color changes can be visually confirmed with the naked eye or via spectrophotometry.24) The disadvantage of the former approach is that the subjectivity of the inspector cannot be excluded; therefore, the latter approach is more widely used.26) Spectrophotometry methods can be used to quantitatively measure the color change of a material, thereby providing an objective evaluation.27) In the present study, a spectrophotometer was used to examine the color difference of the experimental specimens coated with Peony extract. As shown in Fig. 3, no significant difference is observed between the uncoated and coated specimens (p > 0.05), and no color change occurs with the change in concentration in the experimental group (p > 0.05). These results indicate that the Peony extract does not discolor the resin surface at the baseline. Nevertheless, polymers are subjected to different degrees of staining and future studies may investigate color stability of extract-treated specimens immersed in different staining solutions.28)
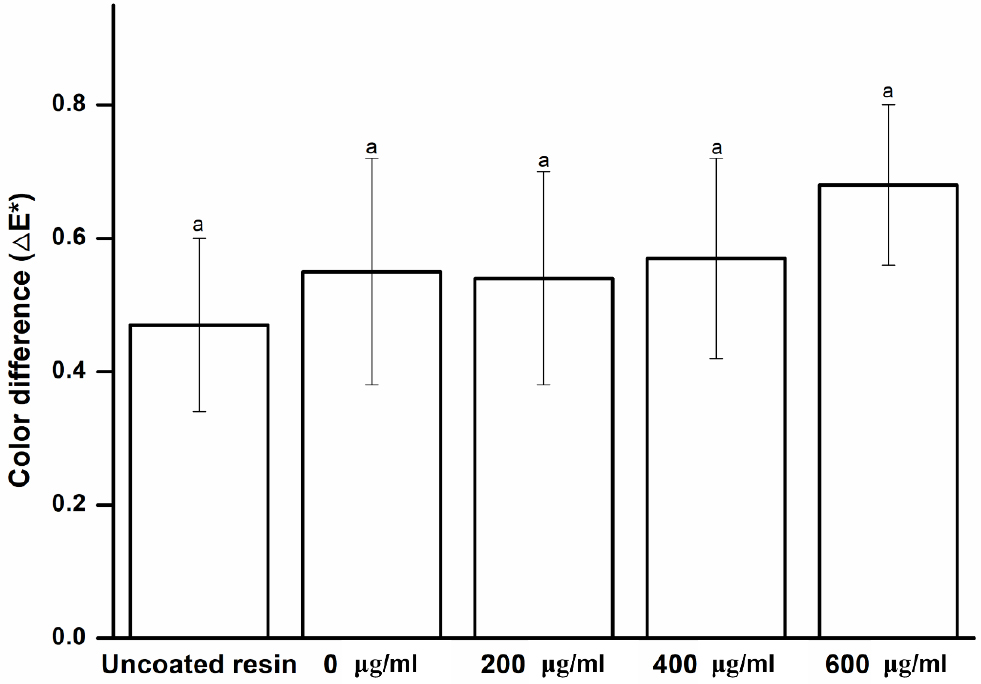
Fig. 3
Color change of the experimental and control groups. Each value represents the mean of five measurements, and the error bar shows the standard deviation of the mean value. The lowercase letter, a, indicates no significant difference was observed between the values of the experimental and control groups (p > 0.05).
Based on the results presented thus far, our first null hypothesis is accepted, i.e., the denture base resin coated with Peony extract would not result in significant differences in the surface properties compared to the control.
Toxicity of the denture base material may cause irritation, inflammation, and allergic reactions to oral tissues.29) These factors can also be due to the local effects of toxic substances liberated from the denture base.30) To prevent these side effects, it is important to determine whether toxic substances are released from the oral mucosa or saliva.31) Our study assessed the effect of the Peony extract coating on cell viability and cytotoxicity using the MTT assay. According to the ISO 10993-5, the MTT assay is recommended when selecting cytotoxicity test of medical devices.29) Peony extracts of three different concentrations were used for cytotoxicity evaluation, and one group without Peony extract was used as the control group. As shown in Fig. 4, all the experimental groups show no significant difference in cytotoxicity compared to the control group (p > 0.05). Therefore, the Peony extract is not considered cytotoxic. The second null hypothesis is accepted, i.e., the denture base resin coated with Peony extract would not result in significant differences in biocompatibility compared to the control.
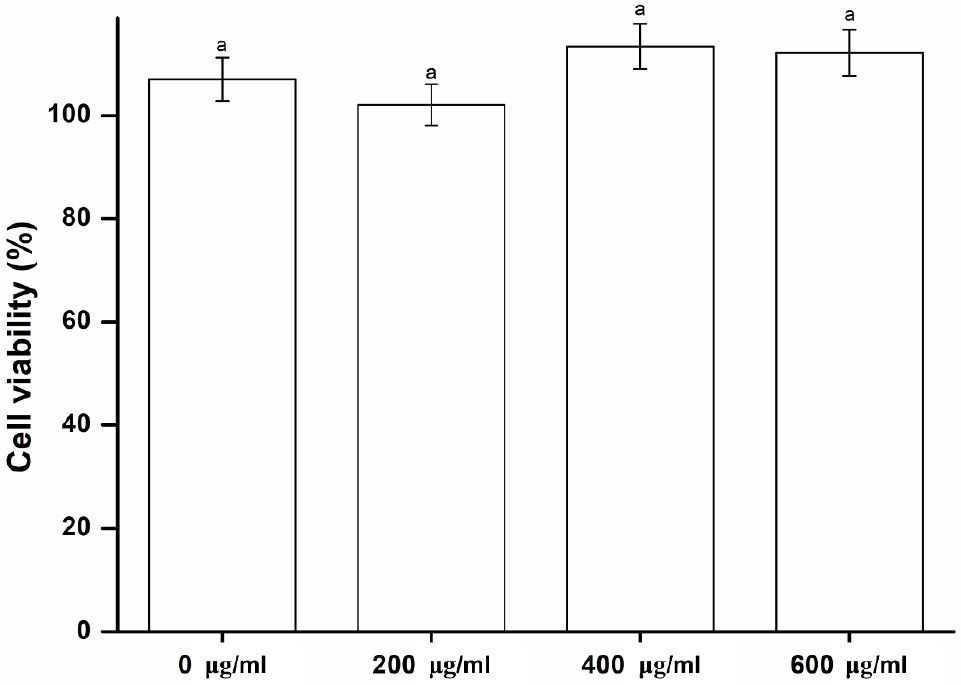
Fig. 4
Biocompatibility of the experimental and control groups. Each value represents the mean of five measurements, and the error bar shows the standard deviation of the mean value. The lowercase letter, a, indicates no significant differences between the values of the experimental and control groups (p > 0.05).
The third null hypothesis is rejected, i.e., the denture base resin coated with Peony extract would not result in significant differences in antimicrobial properties compared to the control. Denture stomatitis can easily occur because dentures are clinically used in close contact with tissues such as the gingival mucosa.14) Therefore, there is a need for a novel denture base resin with antimicrobial activity. In evaluating the antimicrobial activity of the novel denture base resin developed in this study, a microbial suspension was directly inoculated on the specimen to model the microbial adhesion scenario when a denture is installed in the oral cavity and then cultured for 24 and 48 h. Subsequently, the antimicrobial activity was evaluated by counting the number of viable cells. This study focused on S. mutans and C. albicans as these are the most common bacteria and fungi in the oral cavity, and are known to attach to denture base resins easily.32,33) Therefore, assessing the antimicrobial properties of these microorganisms is a clinically relevant outcome.34) As shown in Fig. 5, experimental specimens containing Peony extract significantly affect the growth of the oral pathogens. After 24 h of culture, the number of viable S. mutans cells substantially decreases in the experimental group compared to that in the control group, whereas that of C. albicans does not show a significant difference (p > 0.05). However, after 48 h, the number of viable S. mutans and C. albicans cells significantly decreased in the experimental group compared with the control group (p < 0.05). This result confirms the antimicrobial properties of the specimen coated with the Peony extract. However, the antimicrobial of the denture base resin was not evaluated using longer contact times and without the cyclic fatigue test. Therefore, antibacterial evaluation considering these cases will be investigated in future studies. Despite this limitation, we believe that Peony extract can be used as an antibacterial coating agent.
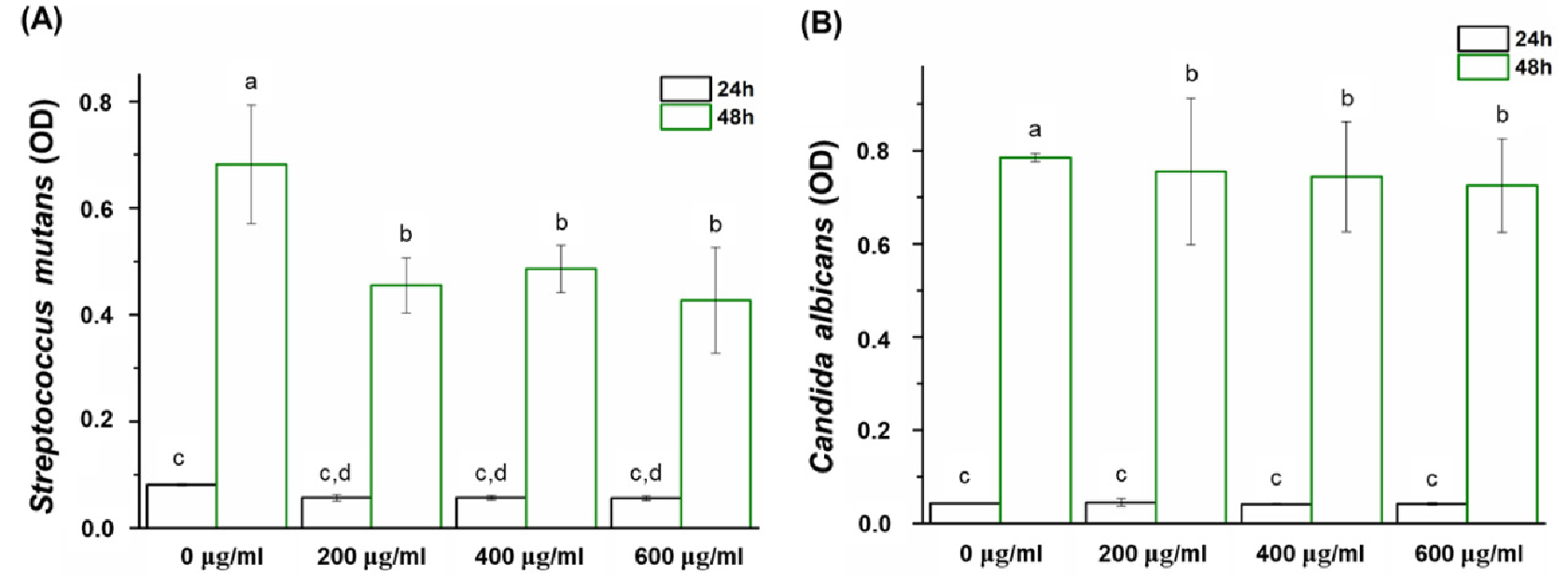
Fig. 5
Comparison of the optical density (OD) of (A) S. mutans and (B) C. albicans between the experimental and control groups. Each value represents the mean of five measurements, and the error bar shows the standard deviation of the mean value. Lowercase letters indicate no statistically significant difference between the values of the control group and those of the experimental group within the same time group (p > 0.05).
The polyphenol content in the Peony extract used to coat the specimens was evaluated. The results, shown in Table 1, indicate that polyphenols (from the Peony extract-coated denture base resin) leach out into the liquid environment (p < 0.05). Although the polyphenol contents of the experimental groups do not correlate with the coating amount of concentrated Peony extract, there is a correlation between the control and experimental groups. Previous studies have noted that polyphenols have important antioxidant effects and biological activities; specifically, many phenolic compounds have an inhibitory effect on microbial growth.14,35) This study confirms the antimicrobial effect of Peony extract-coated denture base resins against oral pathogens for the first time, even though only a short test period was used. Therefore, it is necessary to further investigate the surface characteristics, such as microhardness, contact angle, color stability, biocompatibility, and long-term antimicrobial activity.14,19) And, it is necessary to consider a wide range of efficacy through various microorganisms. Measurement of the minimal inhibitory concentration of a plant extract will be also investigated in future studies. Phenolic compounds are believed to be the source of many natural antioxidants and are widely found in different plants.20) Previous studies revealed a high correlation between the presence of phenolic compounds and antimicrobial or antioxidant activity.19) Therefore, natural antimicrobial substances have great potential for dental applications and should be investigated further.
4. Conclusions
In this study, we investigated the surface properties, cytotoxicity, and antimicrobial performance of the Peony extract coated on a denture base resin. The results of this study are summarized as follows:
(1) No statistically significant differences in the surface properties or cytotoxicity were observed between the denture base resin coated with the Peony extract and the control group (p > 0.05).
(2) Conversely, a statistically significant difference was observed in the antimicrobial efficacy between the denture base resin coated with the Peony extract and the control group (p < 0.05).
From our results, the antimicrobial activity was confirmed through the coating of an antibacterial agent on the denture base resin, which enables bacterial propagation, and it was confirmed that there was no cytotoxicity.
Based on the results of this study, Peony extract can be used as an antibacterial coating agent in the future, and the denture base resins coated with Peony extract are a promising dental material for preventing stomatitis.




