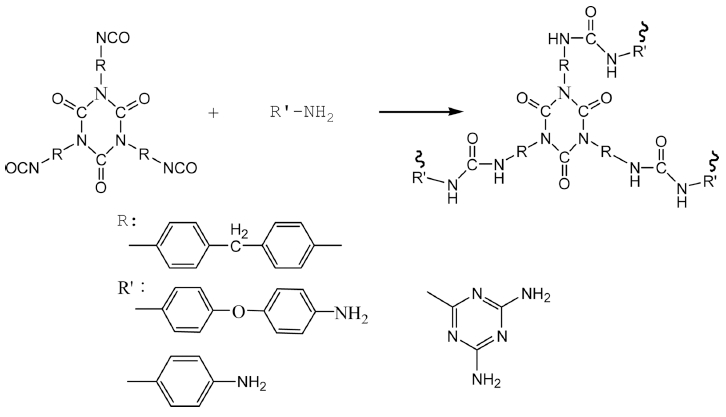
1 Introduction
In view of the increasingly serious environmental problems, researching and synthesizing the new materials to store and adsorb gas are particularly important, especially in the era of fossil energy drying up. Hydrogen is used in many fields as a kind of energy and gradually be taken seriously. Finding out a safe and effective method of hydrogen storage has become the key to the development of hydrogen energy. As we known, the methods of hydrogen storage mainly include compressed hydrogen, liquefaction hydrogen storage, glass microspheres for hydrogen storage and hydrogen adsorption storage etc.1-3) Hydrogen adsorption storage is becoming a hotspot of the current hydrogen storage materials development and research, due to its characteristic of safety, reliability and high efficiency storage etc.4-6) In a variety of adsorption material, pure porous polymers(PPPs) have a great application prospect and got popularly attention because of its large surface area in gas separation and storage.7) So, it is very important for studying and synthesizing pure porous polymer to adsorb and store hydrogen.
The polymers with high surface area should be synthesized first, because the capacity of the adsorption gas will increase with the increase of surface area. In the way, that contains the stabilized isocyanurate and its isocyanate groups have a good reactivity with amino, is chosen to be monomer to synthesize polymer with high surface area.
In this study, we have fabricated pure porous polymers with relatively high surface area by chosen MDI-trimer and different of NH2-containing monomers as monomers, in which is containing the stabilized isocyanurate ring and its -NCO groups with higher reactivity to react NH2- containing monomers. Synthesized polymers show good hydrogen storage properties.
2 Experimental
2.1 Materials and instruments
4,4’-Diphenylmethane diisocyanate(AR), Anhui ANLI Artificial Leather Co., Ltd.; p-phenylenediamine(CP), dimethyl sulfoxide(AR), Sinopharm Chemical Reagent limited corporation; 4,4’-oxydianiline(ODA), TCI(Shanghai) Development Co., Ltd. melamine(AR), Tianjin bo di chemical Co., Ltd; 2,4,6-tris(dimethylaminomethyl)phenol(DMP-30).
Nicolet 6700 Fourier transform infrared spectrometer, 0.2 cm-1 resolution 32 scans; STA409PC simultaneous thermal analyzer, Netisch; JW-BK132F Specific surface analyzer, Beijing JWGB Sci & Tech Co. Ltd; FineSorb- 3110 Hydrogen storage analyzer, Finetec Instruments. Fig. 1
2.2 Synthesis of porous polymer
(1) MDI and ethyl acetate at a ratio were added to four-neck bottles with mixing and reflux, and added drop by drop DMP-30 at 60 °C. After the content of isocyanate (-NCO) groups was to the expected value, by di-nbutylamine method, and the MDI-trimer was synthesized. Then, took out a certain amount of reaction products of MDI-trimers to four-neck bottles with mixing and reflux, added p-phenylenediamine, 4,4’-diaminodiphenyl ether or melamine up to 80 °C and stirred for 12h, there will got the pure porous polymer named as P1, P2 and P31.
(2) Melamine and dimethyl sulfoxide in different proportion dissolved by stirring and heating in four-neck bottles with thermometer and reflux device, and adding a certain amount of MDI-trimer was keeping at 180 °C for 24h. After filtrating, washing several times with acetone, vacuum drying at 180 °C for 24h, reflux in chloroform at 70 °C for 12h, and vacuum drying, the powder of porous polymer was obtained named as P32.
3 Results and Discussions
3.1 FTIR patterns and XRD patterns
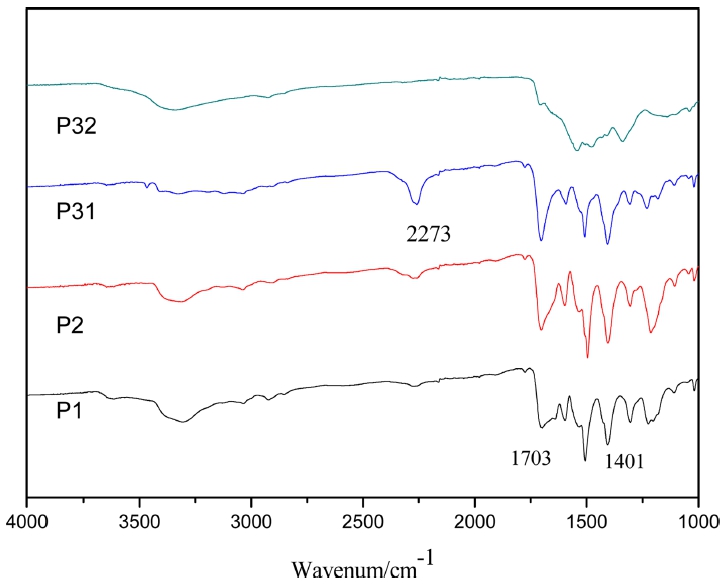
Firstly, polymers were characterized by FT-IR as shown in Fig. 2, OCN- in MDI trimer and NH2- in amino monomers will react to connect into polymer in the form of the imino groups. The IR absorption peak at 3340 cm-1 in the infrared spectra of P1, P2, P31 and P32 corresponds to N-H stretching vibration, that indicate the two monomers reacting to form a new substance. And the obvious IR absorption peak at 1401 cm-1 and 1703 cm-1 correspond to the characteristic absorption peaks of isocyanurate.8) What calls for special attention is that the obvious IR absorption peak of isocyanate groups at 2273 cm-1 only appear in the infrared spectra of P31. The reason is that melamine is not soluble in ethyl acetate, and the reaction is not complete under the same reaction conditions of MDI trimers. On the other side, in melamine the quadrant stretching vibration peak (1541 cm-1) and the semicircle stretching vibration peak (1472 cm-1)9) of thiotriazinone both appear in the infrared spectra of P32.
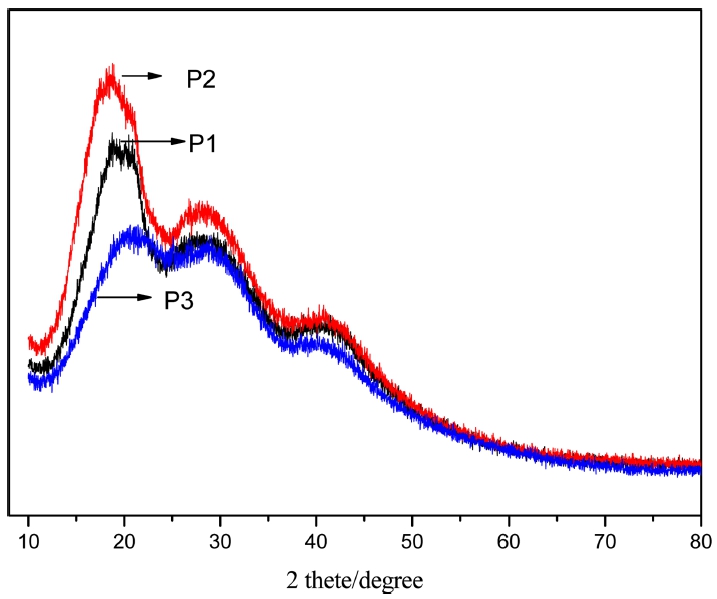
Combination with XRD pattern, as shown in Fig. 3, form the view of the shape of the diffraction pattern, there is no symmetrical peak, only a series of broad peaks appear continuously, indicating that the formation of the formation of the material is not stereotyped state. From the spatial configuration, because of the large steric hindrance of the synthesized polymers, it is not a fixed form of the connection between the two bodies and the NH2- in amino monomers, such as, the random polymers of P1 and P2 linked by the imino groups organic materials that have been reported. They are all noncrystalline.
3.2 N2 adsorption-desorption performance
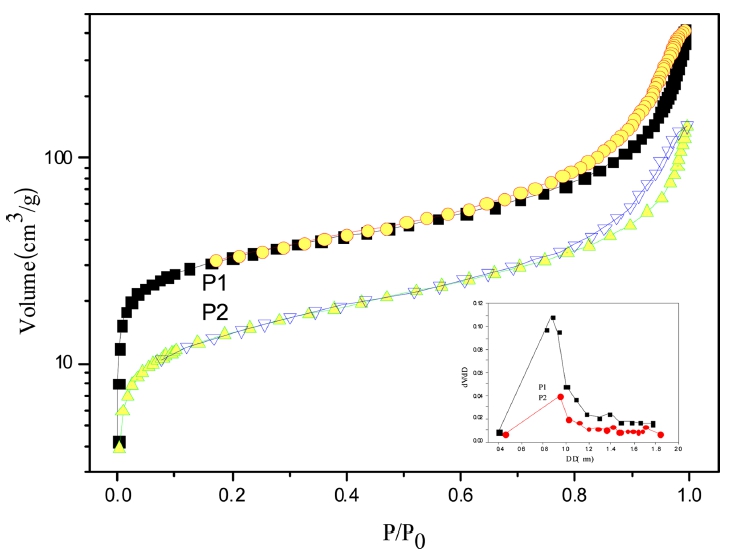
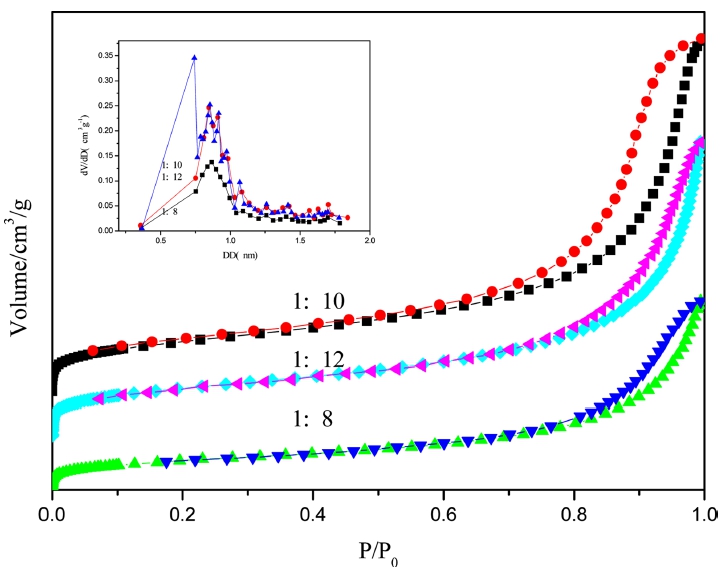
Specific surface area and pore properties of the polymers were characterized by nitrogen adsorptiondesorption, as shown in Fig. 4, where adsorption and desorption isotherm was got by using N2 as molecules probe at 77k. The isotherm of P1, P2 and P3 all conform to the IV type by contrasting with six types(IUPAC). The hysteresis loops in the high pressure(P/P0 = 0.8-1.0) show that there are mesoporous or macroporous. In the low pressure(P/P0 = 10-5-10-2) N2 adsorption has a relatively fast growth attributing to micropore adsorption of N2 molecules.10)
The specific surface area and porosity properties is listed in Table 1. The specific surface area of P1 is larger than P2, which results from the degrees of freedom of pphenylenediamine monomer is greater than ODA monomer so as easier to connect molecular chains into closed pores. From the pore size distribution the most probable pore size of P1 at 0.9 nm and P2 at 0.98 nm, they are very close, but form the point of micropore volume of P1 is bigger.
Table 1
The specific surface area and porosity properties.
| Samples | SBET (m2/g) | Vtotal (cm3/g) | Vmicro (cm3/g) | DD (nm) |
|---|---|---|---|---|
| P1 | 117.2 | 0.6 | 0.045 | 0.90 |
| P2 | 53.4 | 0.2 | 0.020 | 0.98 |
| P31 | 15.1 | \ | \ | \ |
The specific surface area of P32 samples were synthesized by different reactive functional groups molar ratio(n(-NCO):n(NH2)) and various reaction times are listed in Table 2. When molar ratio at 1:8 reacting for 12h, the specific surface area is largest, followed by its molar ratio of 1:12. When reaction time is 24h, the specific surface area with a molar ratio at 1:10 is largest, followed by its ratio of 1:12. The reasons why the changes of molar ratio and reaction time both have influences on the specific surface area are that the polymer molecular chains link in the form of random and MDI-trimer has a large steric hindrance. Table 3
Table 2
the specific surface area of P32.
| n(-NCO):n(-NH2) | Reaction for 12h | Reaction for 24h | ||
|---|---|---|---|---|
| SBET(m2g–1) | SLangmuir(m2g–1) | SBET(m2g–1) | SLangmuir(m2g–1) | |
| 1:8 | 188 | 291 | 144 | 173 |
| 1:10 | 132 | 216 | 243 | 366 |
| 1:12 | 158. | 259 | 230. | 279 |
Table 3
Porosity properties of P32 reacting for 24h.
| n(-NCO):n(NH2) | SBET (m2/g) | Vtotal (cm3/g) | Vmicro (cm3/g) | DD (nm) |
|---|---|---|---|---|
| 1;8 | 144 | 0.4 | 0.05 | 0.88 |
| 1:10 | 276 | 0.6 | 0.11 | 0.85 |
| 1:12 | 230 | 0.6 | 0.09 | 0.75 |
As shown in Fig. 5, the pore size distribution of P32 with molar ratio of 1:8 is uniform, mainly concentrating in the range of 0.8 to 0.9 nm. The pore size distribution of the sample of 1:12 is widest, concentrating in the two regions: 0.75 nm and 0.8 nm to 1.0 nm. Comparatively speaking, when molar ratios are 1:10, the specific surface area and microporous volume are largest, so 1:10 is best molar ratio of raw material.
3.3 Hydrogen adsorption properties
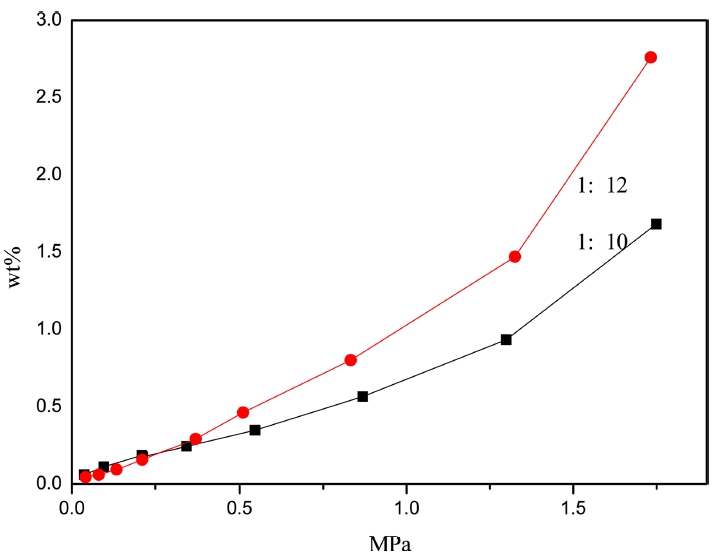
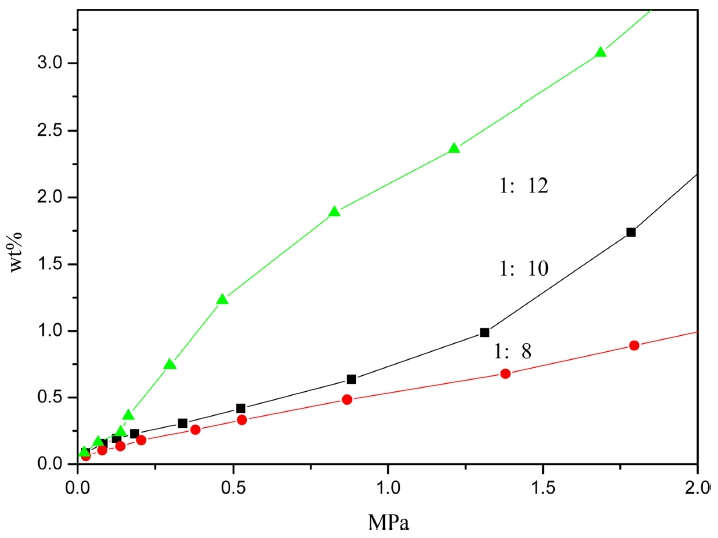
In order to test polymer hydrogen adsorption, the larger specific surface area of P32 was chosen to study. That’s because the more microporous of organic porous materials, whose hydrogen adsorption are based on physical adsorption, will show the better hydrogen adsorption properties. Fig. 6 shows the hydrogen adsorption capacity of P32 is relatively larger in high pressure when the molar ratio is 1:12 and its increases with the pressure. Under the condition of 77 k/1bar, the hydrogen adsorption capacities were respectively 0.1 wt% and 0.07 wt% when the molar ratios were 1:10 and 1:12. As shown in Fig. 7, the hydrogen adsorption capacity of P32 reacting for 24h is larger than reacting for 12h due to the more microporous. And a introduction of nitrogen heterocyclic to the organic skeleton increases the enthalpy of hydrogen adsorption, which results in the hydrogen adsorption capacity increases with the increase of the weight ratio. Under the condition of 77 k/1bar, the hydrogen adsorption capacities were 0.14 wt%, 0.16 wt% and 0.18 wt% when the weight ratios were 1:8, 1:10 and 1:12, respectively. Compared with the adsorption capacity in the practical application, there is still some way to go.
4 Conclusions
In summary, the amorphous polymers of P1, P2 and P3, containing isocyanurates and connected by the imino groups were synthesized by using solvothermal method and had good stability. P3 is more stable due to it’s including of thiotriazinone. Specific surface area of P1 is greater than P2,because the smaller monomer molecules easily access to the larger specific surface area of polymer. Synthesis of P32 indicated that the choice of solvent and temperature has a great influence on the synthesis of porous polymer and different specific surface area of polymer can be got through adjusting the proportion of synthetic raw materials. Moreover, P32 prove to be a certain amount of adsorption performance for hydrogen, just adsorption has yet to be improved. But synthesis of high specific surface area of porous polymer and developing the ability of adsorption still have a great studying significance.