1. Introduction
Yttria (Y2O3) has garnered considerable attention in the semiconductor industry owing to its exceptional plasma resistance against radical cationic activation, surpassing that of conventional materials such as quartz, Al2O3, ZrO2, BN, and SiC.1) Furthermore, yttria has been extensively employed in solid-state lasers, infrared transparent ceramics, and high temperature structural components due to its remarkable thermal stability, chemical inertness, and excellent optical characteristics.2) However, the intrinsically high melting point of yttria (2,430 °C) poses a significant challenge to densification, even though achieving full density is essential for optimizing plasma resistance.3)
Various strategies such as nanoparticle synthesis and dopant assisted sintering have been explored to improve its sinterability. Nevertheless, these approaches often involve high processing costs, introduce extrinsic defects, and compromise plasma durability. From this standpoint, the present study establishes a stepwise sintering protocol for high purity yttria ceramics and systematically elucidates the interrelationships among their microstructural evolution, mechanical integrity, optical performance, and plasma resistance. The fabrication of dense, additive free yttria remains a critical challenge, as excessive thermal exposure during high temperature sintering can induce volatility and alter its intrinsic physicochemical properties.4)
Previous studies have demonstrated that near theoretical densities can be achieved under vacuum or hydrogen atmospheres using coprecipitation derived nanoparticles or dopant mediated diffusion.5) Such additives are known to facilitate grain boundary migration by compensating for charge imbalances and promoting cation and solute diffusion.6) Huang et al.7) achieved theoretical density in lanthanum doped yttria via a two-step vacuum sintering process, whereas Choi and Bae8) examined the influence of oxygen vacancies and temperature dependent diffusion on the color, density, and weight variations of yttria ceramics. They attributed the microstructural and chromatic transformations to the volatilization of yttrium ions from oxygen deficient lattice sites at elevated temperatures. Despite these advances, the complete densification of yttria remains inherently difficult owing to its strong tendency for abnormal grain growth during high temperature sintering. To overcome these challenges, various advanced processing techniques including coprecipitation based nanopowder synthesis, microwave flash sintering, hot isostatic pressing, vacuum sintering, and spark plasma sintering have been employed to fabricate transparent polycrystalline yttria.9,10) While most of these studies have focused primarily on nanoparticle fabrication and optical transparency, recent investigations have increasingly emphasized the importance of precursor powder stabilization as a key factor in achieving highly dense and defect minimized yttria ceramics.
In the present work, we systematically investigated the roles of oxygen vacancies and hydration reactions in governing the sintering behavior of yttria. Yttria powders were calcined under controlled hydration conditions, followed by repeated thermal cycling to evaluate their densification behavior, mass variation, phase evolution, and mechanical performance, including hardness and flexural strength. To achieve uniform grains and a dense sintered body, we employed a three-cycle repeated sintering process.11) Finally, the plasma resistance of the fabricated yttria ceramics was comparatively assessed against that of fully dense reference specimens to elucidate the influence of surface and interfacial characteristics on plasma durability.
2. Experimental Procedure
Yttria powders (Shin-Etsu Chemical Co., Japan) with a purity of 99.9 % and an average particle size of 1 µm were used as starting materials: (i) as-received green yttria powder (ii) yttria powder calcined at 1,250 °C for 48 h, (iii) fused yttria synthesized via induction melting,8) and (iv) hot-pressed yttria specimen.12)Table 1 summarizes the specimen designations and corresponding descriptions used in this study. For specimen fabrication, 20 g of each powder was loaded into a graphite mold (diameter: 60 mm) and compacted by uniaxial pressing at 20 MPa, followed by cold isostatic pressing (CIP) at 300 MPa to minimize residual porosity. Graphite molds were used due to their non-adhesive properties, allowing demolding without release agents. The resulting green bodies were subsequently sintered at 1,700 °C for three consecutive cycles in air to achieve full densification and pore elimination.11) Both heating and cooling rates were controlled at 5 °C/min throughout the process. The phase composition, microstructural morphology, and bulk density of the sintered specimens were characterized using X-ray diffraction (D/MA2550V, Rigaku, Japan), field emission scanning electron microscopy (JSM-6700F, JEOL, Japan), and helium pycnometry (BRL Pycno, Nikkiso, Japan), respectively. Water absorption of the yttria specimens was measured following ISO 10545-3. The specimens were dried at 110 ± 5 °C to determine the mass (w1) and then immersed in distilled water for over 24 h to measure the mass after immersion (w2). Water absorption was calculated using the equation: Water absorption (%) = (W2 - W1)/W1 × 100. The elastic modulus was measured using the pulse echo technique (5800 Pulser/Receiver, Panametrics, Japan). Fracture toughness (KIC) and Vickers hardness were determined under a 500 g load in accordance with JIS R1610 and ASTM C1327-99 standards. The indentation diagonals were measured using an optical microscope (HV-100, Mitutoyo, Japan). Flexural strength was evaluated by the three-point bending method following JIS R1601 specifications (1311V VRW, Aikoh Engineering, Japan). For comparative analysis, the sintering behavior and plasma resistance of yttria ceramics were benchmarked against reference materials, including Al2O3 (99.6 %, SWI, USA), ZrO2 (99.99 %, SWI, USA), Si wafer (Sumco Co., Japan), and quartz (99.99 %, GE, USA). Plasma erosion resistance was evaluated using a Versiline plasma etching system (UNAXIS Co., USA). The etch rate (ER) and weight loss rate (WLR), which directly reflect the plasma durability of the ceramics, were calculated using the following equations: ER = Etch depth (µm) / Exposure time (min), and WLR = Weight variation (g) × Exposure time (min) / Initial weight (g) × Exposed area (cm2), respectively.
3. Results and Discussion
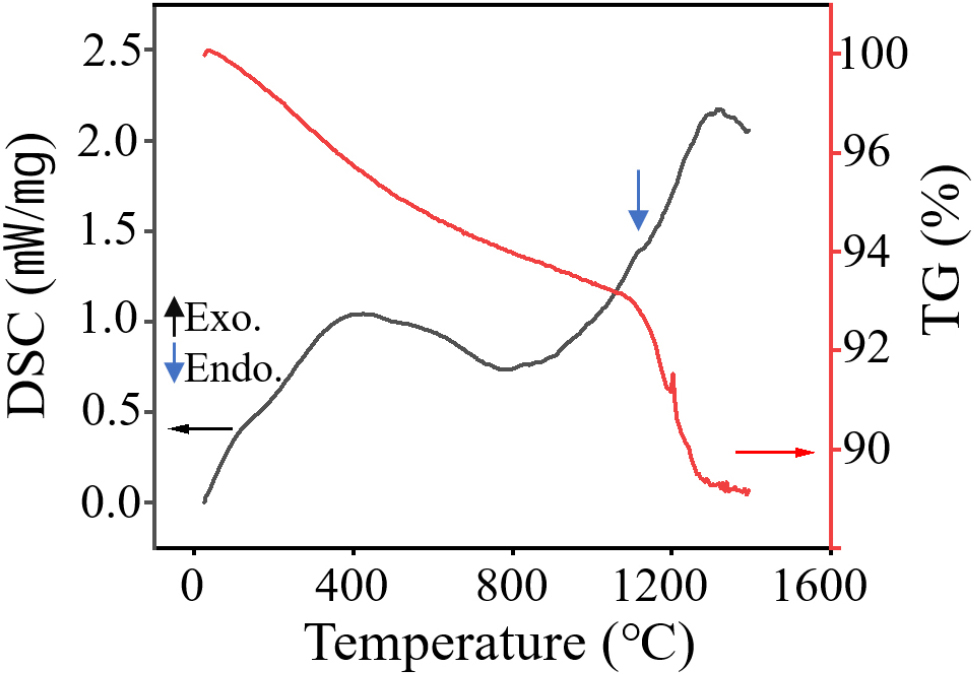
Fig. 1 presents the thermogravimetric (TG) and differential scanning calorimetry (DSC) profiles of the as-received yttria powder. The DSC trace exhibits a pronounced endothermic peak between 1,350 °C and 1,400 °C, corresponding to the desorption of strongly bound hydration water incorporated within the yttria lattice. In parallel, the TG curve indicates a substantial mass loss of approximately 8.98 % near 1,200 °C. Above 1,400 °C, an exothermic event emerges, signifying the complete removal of structural water. Despite these observations, the sintering behavior of calcined yttria specifically treated to suppress hydration reactions, as expressed in equation (1), remains insufficiently elucidated, particularly in relation to oxygen diffusion into lattice vacancies and the suppression of gas evolution during precursor calcination up to 1,250 °C.13)
Here, RE and n denote the rare-earth element and its coordination number, respectively. To further elucidate the interplay between hydration and oxygen vacancy generation during sintering, Saito et al.14) investigated yttria calcined under strictly anhydrous and gas phase free conditions, while intentionally introducing hydration agents into the precursor. Likewise, Huang et al.7) demonstrated that oxygen diffusion in vacancy rich yttria varies significantly upon repeated thermal cycling, directly influencing both mass and density evolution.
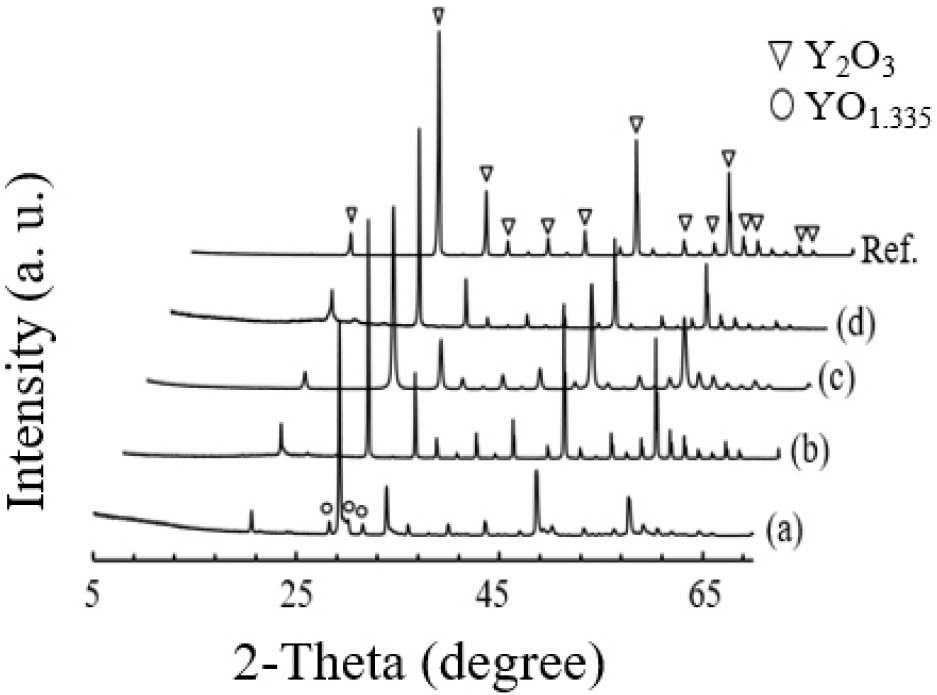
Fig. 2 shows the X-ray diffraction (XRD) patterns of the sintered specimens. The CY samples (derived from calcined yttria) exhibited a single Y2O3 phase, whereas the GY samples revealed a mixed composition of Y2O3 and oxygen-deficient YO1.335, indicative of reduced oxygen occupancy in GY relative to CY. This observation is consistent with previous reports.15) The self-diffusion coefficient of oxygen in Y2O3 is approximately 6 × 10-6 cm2/s, with an activation energy of 82 kJ/mol within 1,050-1,250 °C.14,16) Such structural discrepancies can thus be primarily attributed to the oxygen vacancy concentration. Yttria crystallizes in the c-type rare earth sesquioxide structure, derived from the cubic fluorite lattice by removing one quarter of the oxygen atoms.17,18) Each unit cell comprises 32 Y3+ and 48 O2- ions, together with interstitial voids comparable in size to the oxygen anions, and remains cubic (α-type) up to approximately 2,325 °C.
Swamy et al.19) proposed that irregular oxygen vacancies promote the formation of fluorite type phases, as confirmed by high temperature XRD. Their results also indicated that while additives primarily affect optical transmittance, the emergence of YO1.401 and YO1.335 phases, as well as monoclinic transitions, are predominantly governed by the heat treatment conditions. Despite these distinctions, the underlying mechanism remains consistent: the generation and migration of oxygen vacancies within the yttria lattice.
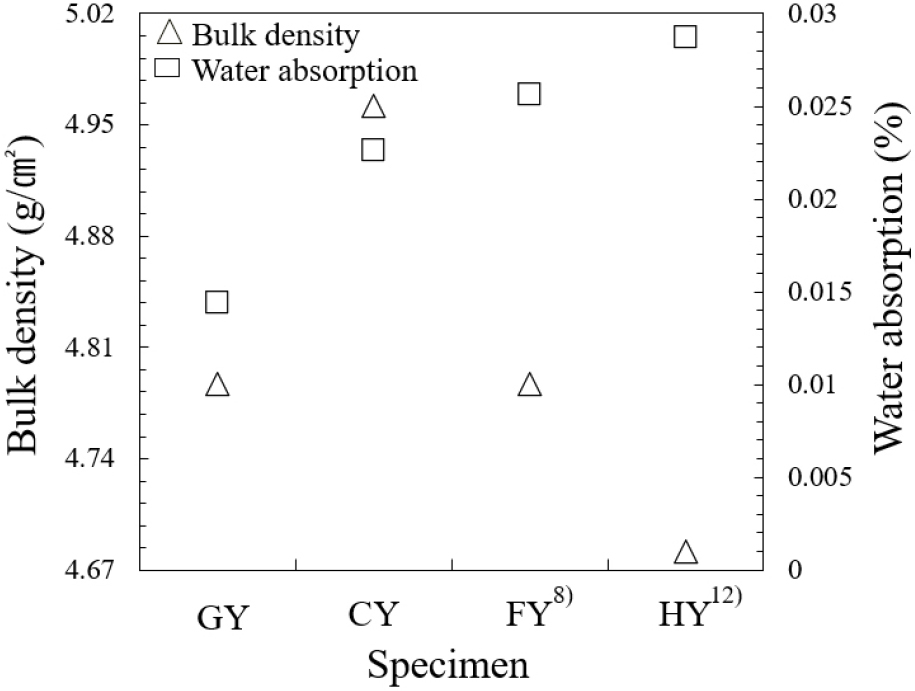
Fig. 3 represents the bulk densities and water absorption characteristics of the sintered bodies. The CY specimens achieved a density of 4.9 g/cm3, slightly exceeding that of GY (4.8 g/cm3). These values are comparable to commercial yttria (Y0100A, Kyocera, Japan; 4.9 g/cm3) and approaching the theoretical density of 5.01 g/cm3. The GY and CY specimens showed relatively higher water absorption, while the FY8) and HY12) specimens exhibited significantly lower values. Sintered ceramics contained residual water or porosity associated with hydroxyl (OH-) groups at the grain boundaries.20) Consequently, water absorption was found to directly affect surface reactivity, thereby promoting plasma erosion. Adsorbed water, in particular, is expected to play a significant role in this process and will be further analyzed in the plasma etching test. These results validate the effectiveness of the applied sintering strategy. During repeated heat treatment, a transient liquid phase is presumed to form locally near 1,522 °C (Fig. 4) and subsequently decompose at higher temperatures. This phase likely crystallizes when the Y:O atomic ratio approaches 60:40, consistent with the Y-O binary phase diagram.21) To obtain a uniform particle distribution, a three-step repeated sintering process was employed to promote densification while suppressing abnormal grain growth.22) Repeated cycle sintering promotes refinement of initially coarse particles into a uniformly distributed grain structure, supporting a liquid phase assisted recrystallization mechanism.23) The progressive disappearance of coarse grains, as shown in Fig. 5, can be attributed to stoichiometric recovery via oxygen diffusion. Rather than persisting as a metastable melt, the system stabilizes through recrystallization, yielding the homogeneous microstructure observed after sintering, consistent with the indentation morphologies shown in Fig. 6. Fig. 6 displays representative Vickers indentation patterns for the GY, CY, FY,8) and HY12) specimens. The FY8) and HY12) samples exhibit distinct crack propagation along cleavage planes and a mist-like texture, suggesting partial glassy phase formation. The hardness of the sintered bodies increases with decreasing grain size, consistent with the Hall-Petch relationship,24,25) whereas excessive grain coarsening leads to a reduction in hardness. The balance between fracture toughness (KIC) and hardness is governed by both grain size and residual porosity.
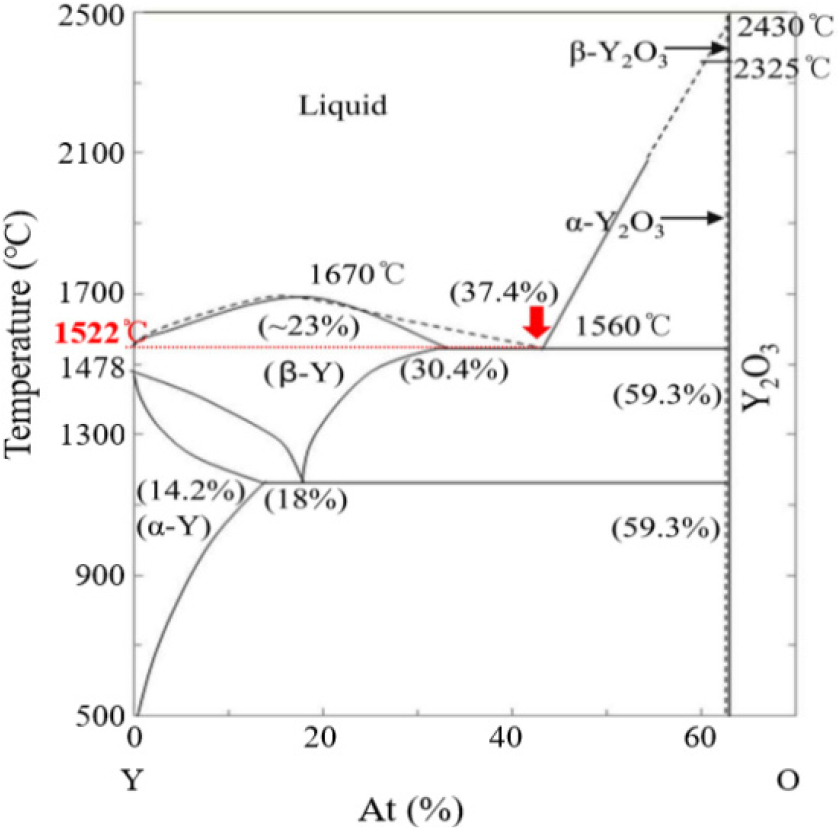
Fig. 4.
Phase diagram of yttrium-oxygen.21) The dashed line represents the thermo dynamically stable liquid phase locally formed at 1,522 °C.
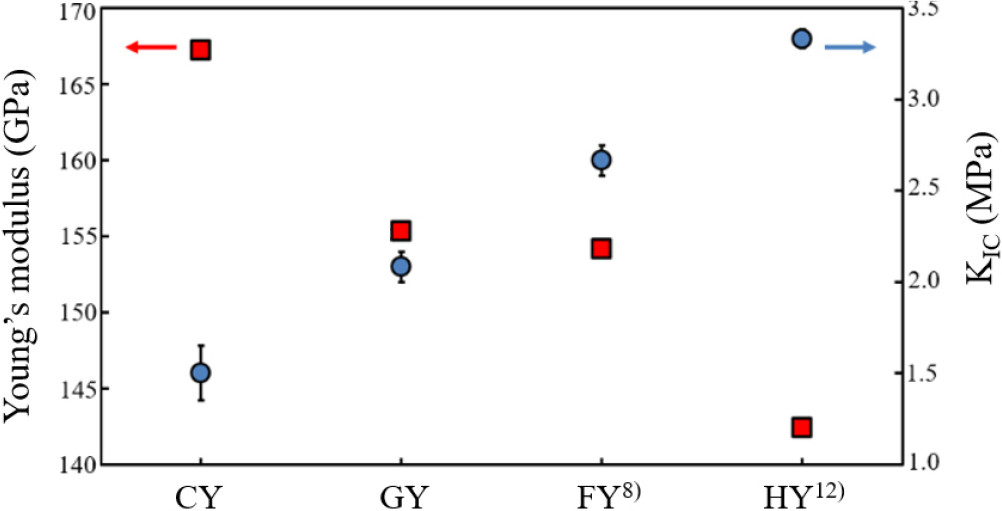
Fig. 7 shows the relationship between Young’s modulus and KIC for yttria ceramic specimens. Young’s modulus is largely governed by microstructural parameters such as ceramic volume fraction, particle size distribution, and porosity.26) Although it generally scales with the ceramic fraction, Young’s modulus is considered relatively insensitive to particle size distribution. Despite the similar particle size distributions observed in Fig. 5(a-c), the HY12) specimen [Fig. 5(d)] exhibits a microstructure with a notably reduced porosity. The present results suggest that the reduction in Young’s modulus mainly results from changes in pore distribution and grain coarsening associated with the development of a coarse grained microstructure. The KIC values were obtained from more than ten repeated indentations under a 500 g load. Notably, the HY12) specimen exhibited relatively high fracture toughness despite its fine microstructure, attributable to its dense, mixed structure composed of both crystalline and amorphous phase [Fig. 6(c, d)].
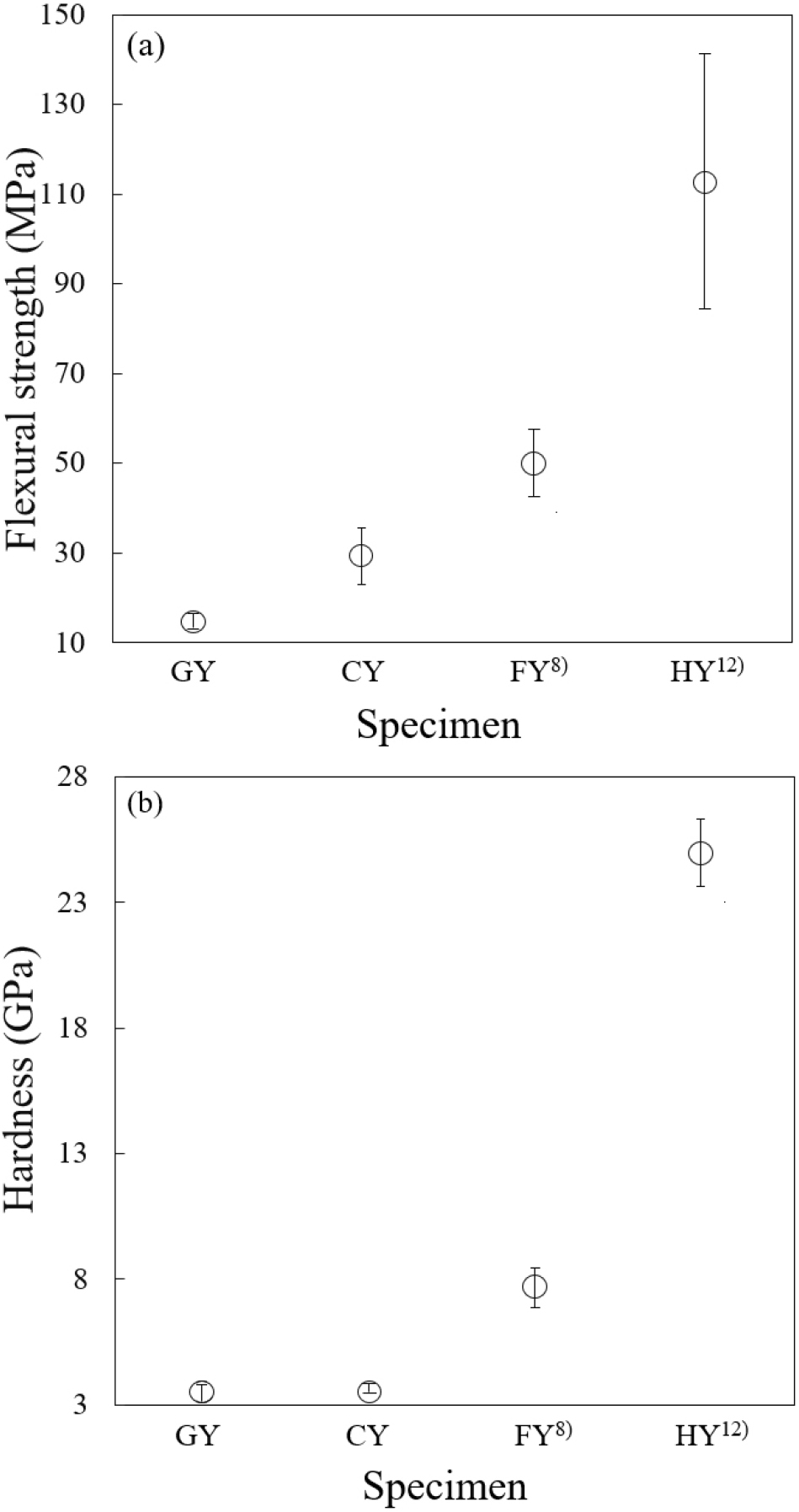
As illustrated in Fig. 8, both flexural strength and hardness correlate positively with bulk density, finer grain size, and reduced porosity. Enhanced densification and reduced porosity yield superior mechanical integrity. Microstructural analysis (Fig. 5) revealed residual pores in GY and CY, whereas FY8) and HY12) were nearly pore-free. Flexural strength and hardness slightly increase with increasing squeeze pressure, except for specimens GY and CY, which exhibited extremely low strength at 40 MPa.
Significant grain coarsening and the presence of a glassy phase were observed in the FY8) and HY12) specimens compared to GY and CY [Fig. 5(c, d)]. These microstructural features contributed to the observed changes in mechanical properties. Specimen HY12) has higher flexural strength and hardness (120 MPa and 25 GPa, respectively) than specimens GY, CY, and FY8) (< 60 MPa and < 8 GPa, respectively). These results clearly demonstrate that mechanical strength and hardness are primarily governed by the degree of microstructural homogeneity and densification, both of which are determined by precursor powder treatment.
Finally, Fig. 9 compares the plasma erosion behavior of the sintered specimens. The specimens were exposed to inductively coupled plasma (ICP) for 60 min, after which the etch depth was measured, and the corresponding ER and WRL were calculated. The test regions were defined by abrasive masking. The plasma durability of yttria ceramics was benchmarked against Al2O3, ZrO2, Si(100) wafer, and quartz. After 10 min of plasma irradiation, quartz and Si (100) exhibited severe etching (2.94-3.14 µm), necessitating early test termination. In contrast, Al2O3 and ZrO2 displayed slower erosion rates (1.31-1.45 µm after 60 min). Yttria ceramics demonstrated exceptional plasma resistance, with etch depths of only 0.41-0.54 µm, exhibiting durability approximately three times greater than that of conventional oxide ceramics. These findings conclusively establish yttria’s outstanding plasma stability under optimized sintering conditions, highlighting its strong potential for next-generation plasma applications.
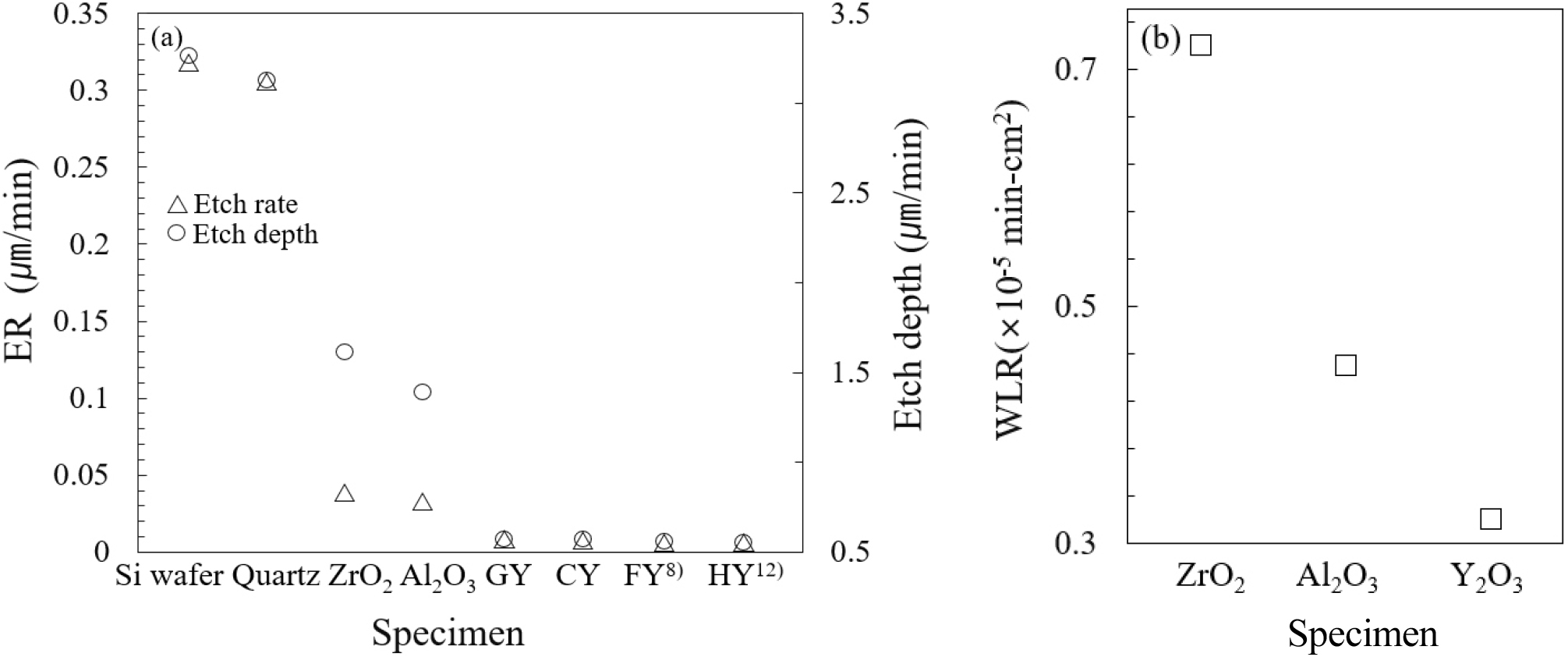
Fig. 9.
Coverage of plasma-etched yttria ceramics compared with quartz, silicon, and ZrO2: (a) etch depth and etch rate. Etching times were 10 min for Si wafer (100) and quartz, and 60 min for ZrO2 and yttria ceramics, GY, CY, FY,8) and HY.12) (b) comparison of weight loss rate (WLR) among the specimens. The etch rates for the wafer and quartz were evaluated after 10 minutes, whereas those for the Al2O3, ZrO2, and yttria specimens were determined after 60 minutes.
4. Conclusion
This study examined the structural, mechanical, and plasma-resistant properties of yttria ceramics fabricated through a three cycle sintering process in air. XRD confirmed that the calcined precursors were primarily composed of Y2O3, with slight deviations associated with oxygen vacancy formation. Repetitive heat treatments effectively enhanced densification, accompanied by transient liquid-phase formation and stoichiometric adjustments near a Y:O ratio of 60:40 due to oxygen diffusion, leading to grain refinement and microstructural homogenization. Mechanical characterization revealed that fracture toughness and hardness were strongly governed by grain size and residual porosity, while Young’s modulus increased with decreasing particle size, underscoring the positive effects of densification. Plasma etching tests demonstrated superior plasma resistance of yttria ceramics compared with conventional oxides, following the etching-rate sequence: Si wafer > quartz > ZrO2 > Al2O3 > Y2O3. These findings highlight the critical role of optimized sintering and microstructural control in enhancing the mechanical integrity and plasma durability of yttria ceramics for advanced semiconductor applications.