1. Introduction
2. Experimental Procedure
2.1. Materials
2.2. Preparation of Silica Suspensions
2.3. Preparation of CNT-Silica Suspensions
2.4. Characterization
3. Results and Discussion
3.1. Rheological Behavior of Silica-Based STFs
3.2. Effect of CNT Incorporation on Rheological Properties
3.3. Influence of Acid-Functionalized CNTs
3.4. Microstructural Analysis of Silica-CNT Systems
4. Conclusion
1. Introduction
Shear thickening fluids (STFs) are a class of non-Newtonian suspensions in which viscosity increases dramatically once a critical shear rate is exceeded.1,2,3,4,5,6,7,8,9,10,11,12,13,14) This unique property enables STFs to behave as free-flowing liquids under low-stress conditions while transitioning into solid-like states under sudden impact or deformation. Such characteristics make STFs attractive for applications in personal protection systems including body armor, stab-resistant textiles, vibration control system and impact-resistant composites.15,16,17,18,19,20,21)
Conventional STFs are typically prepared by dispersing silica (SiO2) nanoparticles in a polymeric matrix such as polyethylene glycol (PEG). A primary limitation in maximizing the STE of traditional STFs is the dependence on a high particle volume fraction (𝜙). While increasing 𝜙 enhances the thickening performance, it invariably leads to a higher suspension density, restricted flexibility, and severe particle agglomeration, which compromises long-term colloidal stability and processing viability. Therefore, a core challenge for next-generation STFs is achieving powerful and immediate shear thickening performance while maintaining material lightness and dispersion quality at reduced base particle concentrations.
The shear thickening effect (STE) is governed by numerous factors, including the dispersed phase properties such as particle size, particle shape, particle size distribution, and volume fraction,22,23) as well as medium properties like viscosity, pH value, and temperature.24,25,26) Among these, adjusting the parameters of the dispersed phase offers the most versatile and effective route for formulating high-performance STFs. Recent research has demonstrated this efficacy through various nanofiller strategies. For example, Huang et al.27) prepared a new type of STF by incorporating graphene oxide into a silica-based system, successfully achieving significant improvements in viscosity, critical shear rate, storage modulus, and dissipation modulus. Similarly, Laha and Majumdar28) utilized silica/halloysite nanotubes to create composite STFs that exhibited notable STE and enhanced the impact resistance of aramid fabrics impregnated with the new material. Furthermore, Ghosh et al.29) investigated the incorporation of cellulose nanofibers (CNFs) to modulate the rheological behavior of silica-based STFs.
Recent studies have highlighted the potential of carbon nanotubes (CNTs) as secondary fillers in STF systems.30,31,32,33,34) Owing to their exceptionally high aspect ratio (up to 2,000:1), large surface area, low density, and superior mechanical properties, CNTs are considered ideal candidates to enhance rheological responsiveness and simultaneously contribute to the lightweight design of STFs. Mechanistically, these nanofillers are expected to form supplementary internal structures. For instance, Nakonieczna et al.,31) Nakonieczna-Dąbrowska et al.32) reported that adding 0.25 vol% multi-walled CNTs (MWNTs) to a silica-based STF increased maximum viscosity from 2,128 Pa・s to 12,213 Pa・s, enhancing impact energy absorption up to 74 %. Similarly, Wei et al.33) observed that 0.8 wt% MWNTs raised viscosity by 191 % and reduced the critical shear rate by more than 60 %. Wang et al.34) noted that CNTs are more effective than other carbon nanofillers, such as graphene nanoplatelets (GNs), primarily due to stronger lubrication interactions with PEG and increased particle winding effects, which effectively accelerate the onset of cluster formation. However, this effect is highly sensitive: Hasanzadeh and Mottaghitalab35) observed in their studies that CNT addition could also sometimes delay the onset of shear thickening due to complex surface interactions that increase the number of hydrogen bonds between the MWNT, silica, and PEG components, highlighting the challenge of achieving stable enhancement.
Prior research has often lacked a systematic approach to controlling CNT dispersion, especially within the confines of high-concentration silica matrices (e.g., 𝜙≥0.45). This failure to control the CNT-silica interface leads to the detrimental effects of slip or premature aggregation. This study addresses this critical gap by systematically analyzing the synergistic influence of varying MWNT loading and the necessity of acid functionalization to improve dispersion stability. We aim to identify the optimal processing parameters required to leverage the CNT’s structural reinforcement capabilities, even under challenging high-𝜙 conditions. The findings offer crucial guidelines for designing lightweight, high-performance STFs for robust protective applications.
2. Experimental Procedure
2.1. Materials
Silica particles (SG-SO500, Sekyung AT, Korea) with an average diameter of 500 nm and a density of 2.32 g/cm3 were employed as the dispersed phase. Polyethylene glycol (PEG, MW 200; product code 6550-1405, Daejung Chemicals, Korea) was used as the dispersing medium. Ethanol (EP grade, Daejung Chemicals, Korea) was added as a diluent to facilitate particle dispersion. Multi-walled carbon nanotubes (MWNTs, A-Tube-M95, produced by catalytic chemical vapor deposition, CCVD) were supplied with diameters of 5-20 nm, lengths up to 10 μm, and a purity of 95 wt%.
The acid functionalization was performed as follows: Pristine MWCNTs were mixed with sulfuric and nitric acids for 5 min and then sonicated for 1 h to ensure dispersion. The mixture was subsequently refluxed at 110 °C for 12 h. Finally, the acid-functionalized MWCNTs were obtained by washing and filtering the mixture with distilled water, followed by drying in a freeze dryer.
2.2. Preparation of Silica Suspensions
To investigate the effect of silica concentration on shear thickening behavior, suspensions were prepared at 30, 35, 40, and 45 vol% silica loadings. Silica particles were added to PEG along with ethanol, followed by ball milling at 500 rpm for 8 h to ensure homogeneous dispersion. The resulting mixtures were subjected to solvent evaporation at 50 °C under stirring (150 rpm, 12 h) using an overhead stirrer to obtain the final silica suspensions.
2.3. Preparation of CNT-Silica Suspensions
For CNT-modified suspensions, the silica content was fixed at 30, 35, 40, or 45 vol%, while MWNTs were incorporated at 0.2, 0.5, 1.0, 1.5, 2.0, 2.5, and 3.0 wt% relative to the suspension. Ethanol (30 wt% of total suspension) was added as solvent, and sodium dodecyl benzene sulfonate (SDBS) was introduced at a 5:1 ratio with respect to CNTs to improve dispersion stability. Pre-dispersion was performed using ball milling at 500 rpm for 30 min, after which PEG was added following the same procedure used for silica suspensions. Acid-functionalized MWNTs were also incorporated using the same method, with the surface modification process conducted according to a previously reported procedure.36)
2.4. Characterization
The rheological properties of the suspensions were evaluated using a rotational rheometer (Discovery HR-2, TA Instruments, USA) equipped with a cone-plate geometry (diameter: 40 mm, angle: 2°). Flow sweep measurements were performed at 25 °C, and each sample was tested 2-3 times to ensure reproducibility, particularly in cases of poor dispersion.
To elucidate the role of CNTs in shear thickening behavior, the microstructures of the suspensions were analyzed using field emission scanning electron microscopy (FE-SEM, JSM-6500F, JEOL, Japan). Special attention was given to suspensions with low CNT loadings, where suppression of shear thickening was observed. The morphology and interaction between silica particles and CNTs were also examined.
3. Results and Discussion
3.1. Rheological Behavior of Silica-Based STFs
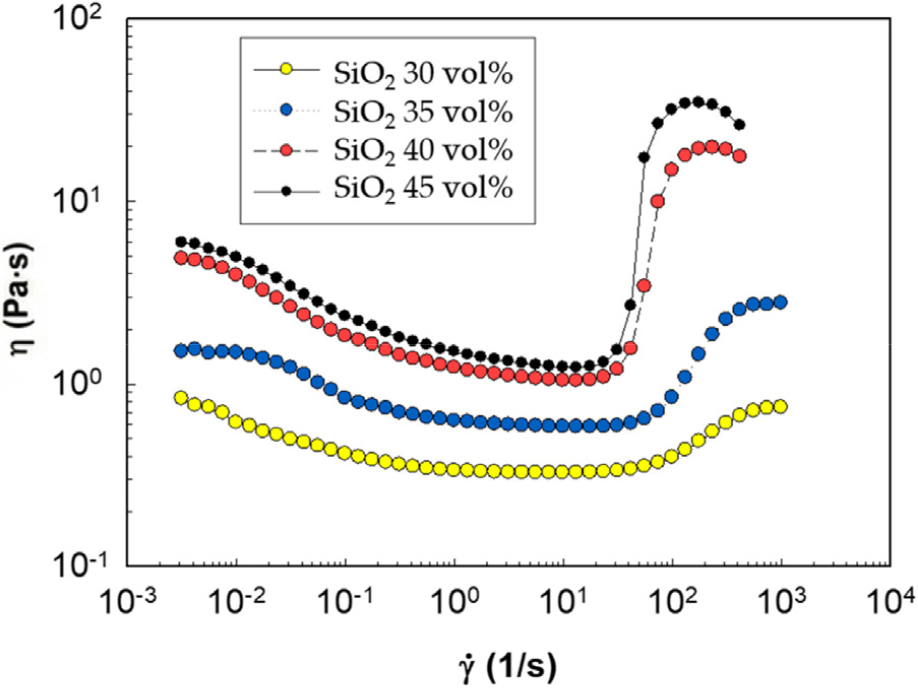
The influence of silica concentration on rheological properties was evaluated using a rotational rheometer over a shear rate range of 1.0 × 10-4 to 1,000 s-1. As expected, increasing silica loading intensified the shear thickening response. The rheological behavior of the baseline suspensions, consisting of silica particles in PEG, was first established to understand the fundamental shear thickening mechanism (Fig. 1). Shear thickening in such systems is driven by the shear-induced formation of transient, jammed particle structures known as hydro-clusters.
Fig. 1 presents the viscosity profiles of suspensions containing 30, 35, 40, and 45 vol% silica. For lower concentrations (30 and 35 vol%), viscosity changes were relatively modest, showing only gradual increases with shear rate. At low particle concentrations, the interparticle spacing is relatively large. Under shear, particles do not collide frequently or forcefully enough to overcome repulsive forces and the lubricating PEG layer, meaning the conditions necessary for strong hydro-cluster formation are not satisfied. This results in only modest, gradual viscosity increases.
In contrast, at 40 and 45 vol%, the system is at a higher particle packing density, closer to the critical volume fraction for jamming. As the shear rate increases, hydrodynamic forces overwhelm stabilizing forces, promoting “frequent particle-particle collisions”. These collisions lead to the formation of extensive hydro-clusters, which trap the dispersing medium and resist flow, manifesting as a “pronounced viscosity jump” in the 101-103 s-1 range. This confirms that a sufficiently high silica concentration is a prerequisite for strong STF behavior.
3.2. Effect of CNT Incorporation on Rheological Properties
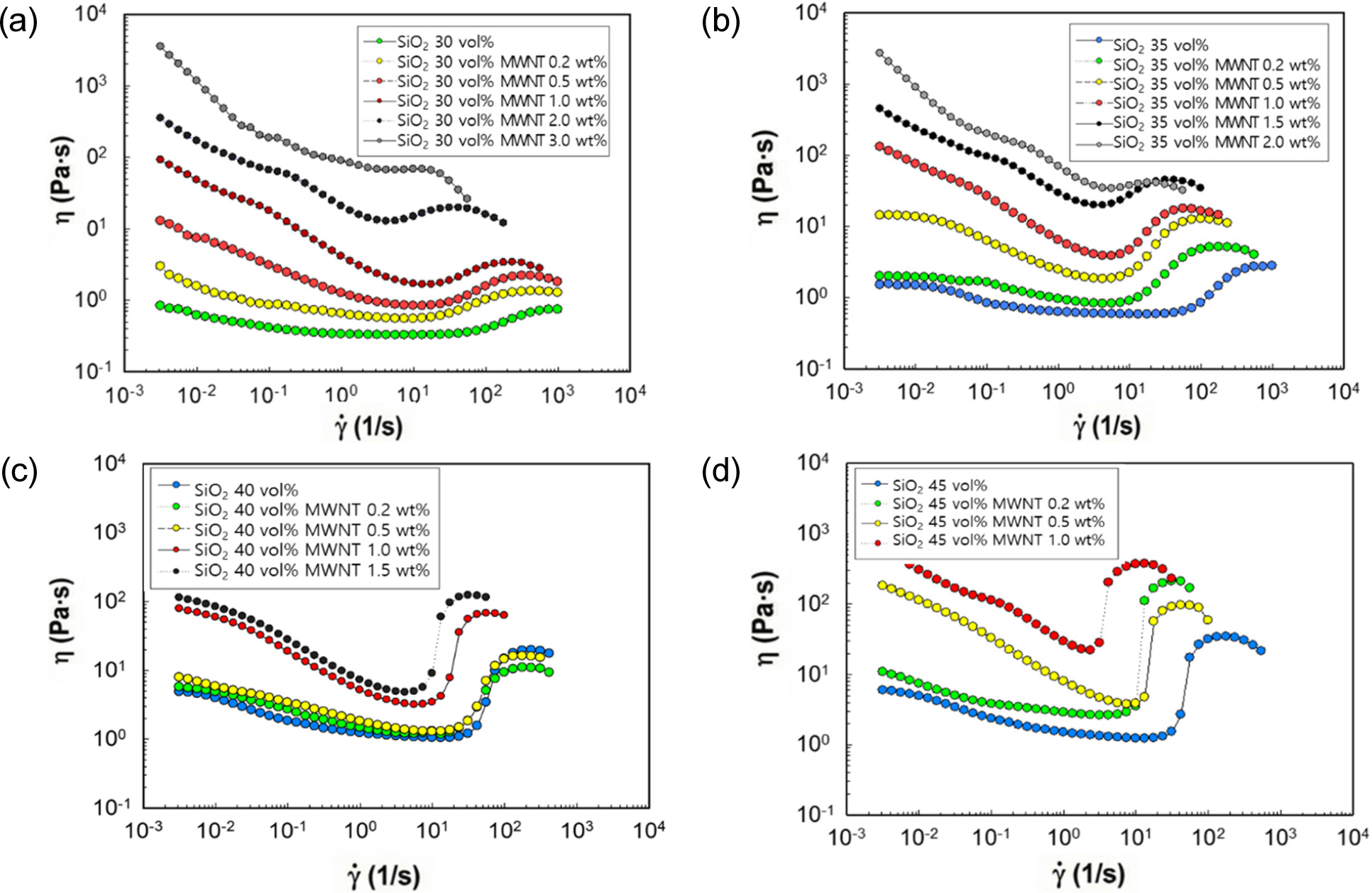
The effect of MWNT incorporation on the rheological behavior of silica-PEG suspensions is shown in Fig. 2. For suspensions containing 30 vol% silica [Fig. 2(a)], increasing MWNT loading progressively reduced the critical shear rate. For example, the critical shear rate, , decreased from 67.7 s-1 without CNTs to 7.0 s-1 at 2.0 wt% CNT. This suggests that CNTs facilitate interparticle interactions under shear, promoting the formation of particle clusters even at relatively low shear rates. The high aspect ratio of CNTs enables them to act as bridging agents, inducing network-like structures. This enhancement is attributed to the high aspect ratio of the CNTs, which allows them to act as bridging agents. Even at low shear rates, the CNTs form a nascent network structure that links silica particles, effectively pre-organizing the suspension. This pre-organization means less shear energy is required to initiate the transition to a jammed state, thus accelerating shear-induced clustering and lowering . As shear continues, these CNTs become entangled within the silica hydro-clusters, promoting stable cluster formation. This results in a synergistic enhancement where the CNT network reinforces the silica hydro-clusters, making them more robust and increasing the maximum viscosity.
However, when the CNT content reached 3.0 wt%, the suspension exhibited an abnormally high viscosity (maximum 3640.0 Pa・s), resembling a rubbery solid rather than a fluid. Under such conditions, shear thickening was suppressed, most likely due to agglomeration and poor dispersion stability.
At 35 vol% silica [Fig. 2(b)], a similar trend was observed: the addition of CNTs lowered the critical shear rate and increased the baseline viscosity. Nevertheless, at CNT loadings above 2.0 wt%, sample mixing became difficult due to solidification, indicating a dispersion limit under high-viscosity conditions. At these concentrations, the CNTs’ strong van der Waals attractions dominate, forming a percolated, solid-like gel that solidifies the mixture. This static gel structure prevents the dynamic formation and dissolution of hydro-clusters, which is the very mechanism of shear thickening.
At 40 vol% silica [Fig. 2(c)], shear thickening was clearly observed. Interestingly, at 0.2 wt% CNT loading, shear thickening initiated at lower shear rates than in the 0.5 wt% system. This is attributed to a slip phenomenon. At this low, likely insufficiently dispersed concentration, the CNTs fail to form a bridging network. Instead, they are postulated to align with the flow, creating “interlayer sliding” planes between silica-rich domains. This slip hinders the particle-particle collisions necessary for hydro-cluster formation, weakening the thickening response.
The most remarkable performance was achieved at 45 vol% silica with 1.0 wt% CNT [Fig. 2(d)]. In this case, the critical shear rate dropped to 2.6 s-1, while the maximum viscosity reached 24,417.0 Pa・s. Such suspensions behaved more like viscoelastic solids than liquids, suggesting that CNT-silica networks had formed robustly. However, higher CNT loadings at this silica content rendered the suspensions too viscous for practical processing or applications.
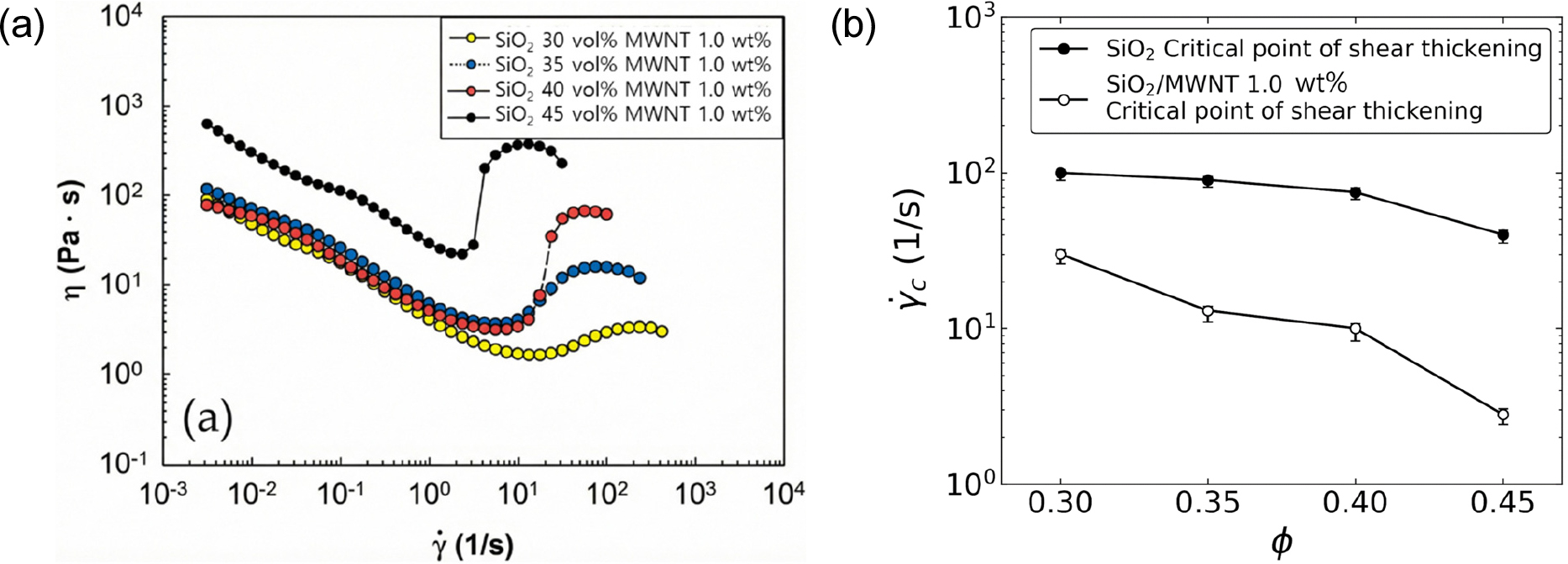
Fig. 3(a) compares the viscosity profiles of CNT-modified suspensions (1.0 wt% CNT) with varying silica contents. All systems displayed typical shear thickening, with stronger effects at higher silica loadings. This reflects the synergistic enhancement between CNT-induced network formation and silica hydro-clustering. Fig. 3(b) highlights the reduction in critical shear rate across silica contents. Without CNTs, critical shear rates were maintained in the 60-100 s-1 range. With 1.0 wt% CNTs, critical shear rates decreased significantly across all silica loadings, dropping to as low as 2.0 s-1 at 45 vol% silica. This demonstrates that CNTs accelerate shear-induced clustering, thereby increasing the sensitivity and responsiveness of STFs to applied stress.
3.3. Influence of Acid-Functionalized CNTs
To mitigate the agglomeration observed with untreated CNTs, acid-functionalized MWNTs (m-MWNTs) were introduced. The functionalization serves two purposes: 1) it introduces electrostatic repulsion between CNTs, counteracting van der Waals attractions to improve CNT-CNT dispersion, and 2) it improves surface polarity, which is theorized to promote hydrogen bonding or electrostatic interactions between the CNTs and the silica particles.
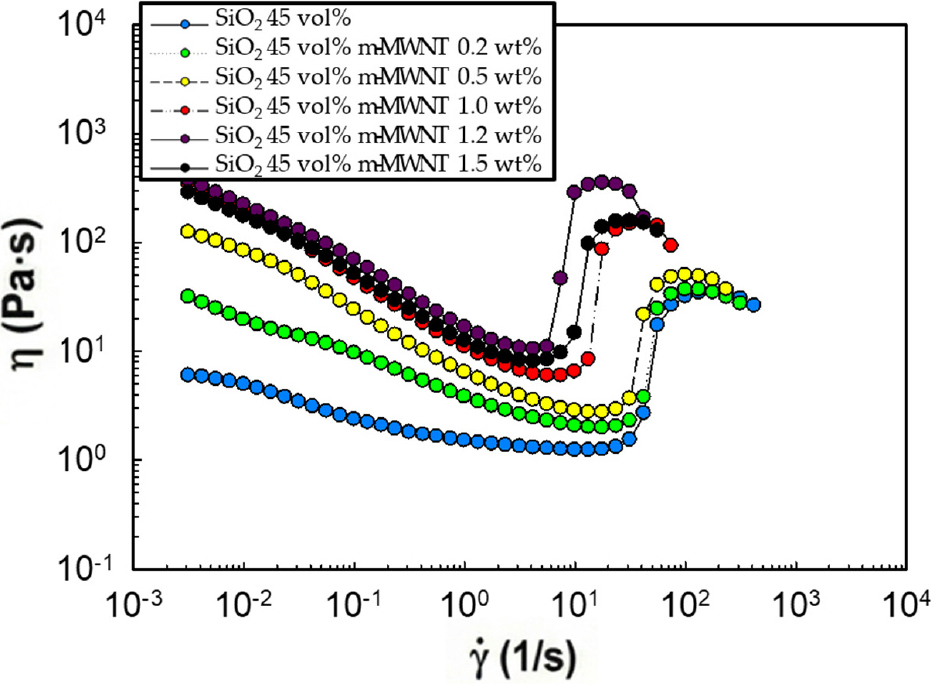
Fig. 4 illustrates the rheological behavior of suspensions containing 45 vol% silica with various m-MWNT loadings. Acid treatment allowed stable incorporation of CNTs up to 1.5 wt%, compared to only 1.0 wt% for untreated CNTs. With 1.5 wt% m-MWNTs, the critical shear rate decreased to 7.8 s-1, and shear thickening occurred more rapidly and strongly than in untreated systems. This indicates that acid functionalization promotes hydrogen bonding or electrostatic interactions between CNTs and silica particles, strengthening hydro-cluster formation.
The acid treatment grafts functional groups onto the CNT surface, which serves two critical purposes for dispersion stability: first, it introduces electrostatic repulsion between adjacent CNTs, thereby counteracting the van der Waals attractions that drive agglomeration; second, it significantly enhances surface polarity, promoting hydrogen bonding and electrostatic compatibility with the silica particles and the PEG medium. This improved interfacial interaction is crucial for achieving a homogeneous distribution of CNTs within the high-concentration silica suspension.
However, an optimal balance exists. At 2.0 wt% m-MWNTs, performance deteriorated again. This suggests that even with functionalization, “saturation effects and renewed agglomeration” occur when the total filler volume (silica + CNTs) becomes too high, leading to processing difficulties and a gel-like state similar to the untreated, over-loaded samples.
3.4. Microstructural Analysis of Silica-CNT Systems
FE-SEM images of MWNTs, silica particles, and CNT-silica mixtures are shown in Fig. 5. Silica particles were observed as spherical aggregates (~500 nm), while CNTs appeared as fibrous bundles. At low CNT content (0.2 wt%), CNTs were distributed as thin layers among silica particles. This structure facilitated interlayer slip under shear, delaying cluster formation and weakening shear thickening.
In contrast, at higher CNT loading (1.5 wt%), silica particles were embedded within a CNT network, with entangled CNTs bridging multiple silica particles. This morphology enabled robust cluster formation under shear, consistent with the pronounced thickening observed in rheological tests.
These microstructural findings confirm that both CNT concentration and dispersion state critically influence STF performance. Poorly dispersed CNTs induce slip and suppress thickening, whereas well-dispersed CNT networks reinforce hydro-clusters and enhance rheological sensitivity.
4. Conclusion
The incorporation of carbon nanotubes (CNTs) has emerged as a promising strategy to enhance thickening efficiency and simultaneously reduce the suspension weight of silica-PEG STFs. The CNTs employed in this study possessed an exceptionally high aspect ratio (up to 2,000:1) and large surface area, with a low tap density (0.22 g/mL) relative to their true density (2.1 g/mL). These characteristics enabled small amounts of CNTs to occupy a significant effective volume.
As a result, CNT addition provided a dual benefit: it intensified shear thickening behavior by reinforcing hydro-cluster networks, and it contributed to the partial replacement of silica volume, thereby reducing the overall weight of the fluid. Acid functionalization was shown to improve CNT dispersion, enabling stable incorporation of up to 1.5 wt% CNTs in suspensions containing 45 vol% silica, which further enhanced shear thickening compared to untreated systems.
However, the findings also highlight critical challenges. At lower CNT loadings, slip phenomena were observed, leading to delayed cluster formation, an effect that became more pronounced at higher silica concentrations. At excessive CNT loadings, agglomeration dominated, suppressing the STF effect. Overall, the findings demonstrate that while CNT incorporation is a valuable tool for designing lightweight, high-performance STFs, effective control of dispersion and slip phenomena is essential. Optimization must be achieved within an appropriate concentration window to fully realize the advantages of CNT-modified STFs.