1. Introduction
2. Experimental Procedure
3. Results and Discussion
3.1. X-ray diffraction analysis
3.2. Morphological analysis
3.3. Elemental analysis
3.4. Magnetic Properties
4. Conclusion
1. Introduction
Nano thermodynamic materials with size-dependent features and high surface area-to-volume ratio are revolutionizing the vibrant field of advanced materials science. Spinel nanoferrites belong to the most important class of these materials, owing to their remarkable versatility and adjustable function characteristics.1,2) The parent structure of these belongs to the inverse spinel general space group, with chemical formula AB2O4, where ‘A’ and ‘B’ represent tetrahedral and octahedral coordination sites in a face-centered cubic lattice of oxygen ions. The possibility to fine-tune the proportion of various cations occupying these lattice sites, and hence the magnetic, electrical, and optical controllability through the manipulation of its structure, proffers scientists with opportunities for predesigned material designing prior to the experiments.3,4)
In this regard, cobalt ferrite (CoFe2O4) receives great attention as one of the most promising hard magnetic materials because it has high coercivity and high chemical stability as well as strong magnetostriction, which are suitable for high-density recording media, medical force field devices and magnetic sensors.5,6) The fundamental thing remains the parameter tailoring according to application- specific demands. One such method is to partially substitute the non-magnetic or differently magnetized ions for cobalt (Co2+). The substitutional Al3+-doped is a well-thought-out alternative; since the substitution with octahedral (B-site) ion not only changes the structural valence state but also the weakening of A-B super-exchange interaction effects results in significant tuning in magnetic properties, such as saturation magnetization (Ms), reverse field and O2- bandgap, as well as for the material.7,8)
The octahedral (B) sites are preferentially occupied by Al3+ ions, which cause cation redistribution between A and B sites and local charge-compensation mechanisms, such as Fe2+ ion redistribution between octahedral and tetrahedral sites and/or generation of lattice defects and internal deformation without forming secondary phases.9) In spinel ferrites, when non-magnetic trivalent ions, e.g., Al3+, substitute for the B sites, the superexchange interactions between A-B and B-B are modified, and the magnetic properties change. As a consequence, the chemical formula AlxCo1-xFe2O4 is chosen as this chemical formula for the step and is often used in the ferrite literature.10) Therefore, the objective of this study is to systematically characterize how the extent of aluminum incorporation (x) into the AlxCo1-xFe2O4 structure influences its associated structural, morphological, and magnetic characteristics. A comprehensive knowledge of these relationships is likely to lay the groundwork for constructing ferritic nanomaterials with custom properties.
2. Experimental Procedure
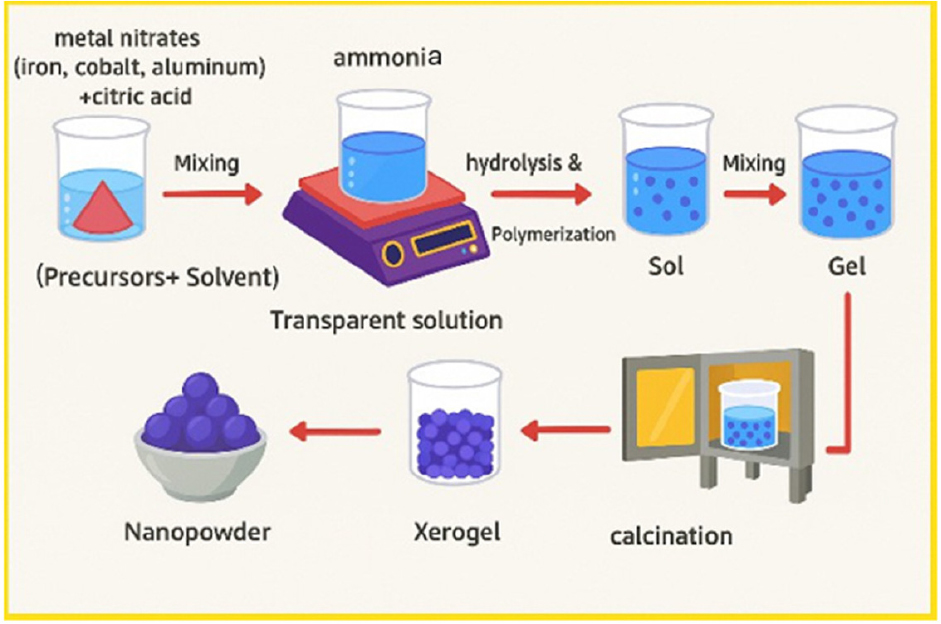
AlxCo1-xFe2O4 nanoferrite samples were prepared using the sol-gel method as shown in Fig. 1. The raw materials to produce AlxCo1-xFe2O4 and their weights are shown in Table 1. In the first step, a small amount of metal nitrates (iron (Fe(NO3)3・9H2O, cobalt Co(NO3)2・6H2O, aluminum Al(NO3)3・9H2O) was weighed and dissolved in 40 mL of distilled water. Citric acid (C6H8O7・H2O) in 40 mL of distilled water was dissolved, served as a chelating agent, and stirred well with a magnetic stirrer to form a homogeneous solution. The pH was made up to about 7 by the addition of a few drops of ammonia solution.
Table 1.
The raw materials to produce AlxCo1-xFe2O4
The solution was heated for 2 h at 90 °C under stirring and then maintained at this latter temperature until it gelled. The temperature was then raised to 110 °C to dry the gel. At 120 °C, the dried gel began to burn spontaneously, leading to a fast self-combustion reaction.
This produced a ferrite powder, which was then allowed to cool before being calcined in an electric furnace at 600 °C for 2 h to clarify the crystals and eliminate any organic matter.
In the present investigation, a range of sophisticated instruments was employed for the characterization of the as-prepared nanoferrite AlxCo1-xFe2O4 samples to uncover their atomic, structural, morphological, and magnetic parameters from which component properties could be extricated. The crystal phase of the nanoproducts was determined by X-ray diffraction (XRD), and the lattice constants and size of crystals were obtained. The surface morphology and size distribution of the particles at the nanoscale were examined by a field emission scanning electron microscope (FESEM). The elemental compositions and distributions of the as-prepared samples were characterized by energy-dispersive X-ray analysis (EDX) to demonstrate their chemical purity. Then, the magnetic properties of the samples, for example, saturation magnetization (Ms), remanent magnetization (Mr), and coercive field (Hc), were analyzed using a vibrating sample magnetometry (VSM). The combination of these devices contributed to establishing a complete image of the microstructure and physical properties of the studied nanoferrite and its relationship with the degree of substitution of aluminum.
3. Results and Discussion
3.1. X-ray diffraction analysis
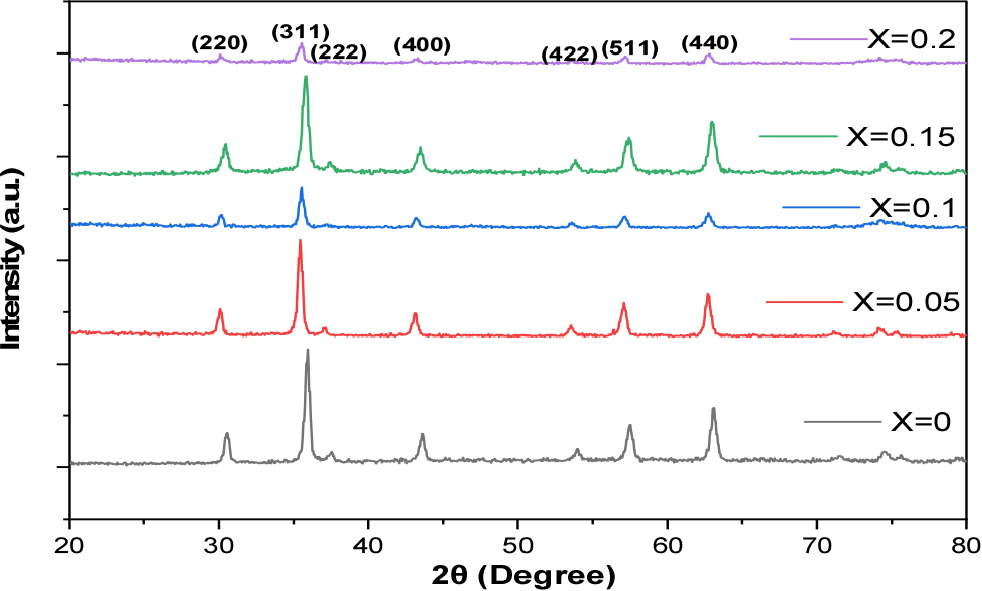
XRD patterns in Fig. 2 indicate the formation of a distinct single-spinel phase in all samples, with characteristic peaks of the spinel structure appearing at planes (220), (311), (222), (400), (422), (511), and (440),11,12) with JCPDS Data (Card No. 22-1086)13) belonging to the Fd-3m (227) space group. The absence of obvious additional peaks or secondary phase peaks within the measurement sensitivity confirms that the introduction of aluminum ions occurred within the spinel material without independent phase formation. This phase stability is important because it ensures that the change in microstructural properties is primarily due to cation exchange within the same structure and not to the formation of new phases.14) The absence of secondary phases in XRD patterns, systematic lattice parameter variations, and the detection of Al3+ signals in EDX spectra collectively support the successful incorporation of Al3+ ions into the spinel lattice.15) The observed non-monotonous variation in lattice parameters is attributed to competing effects such as ionic size difference, lattice strain, and cation redistribution, which are commonly reported in doped spinel ferrites.16)
Lattice parameters were calculated using Match! software (version 3), and the Scherrer equation was used to get the mean crystallite size.17,18)
Where D is the crystallite size, λ is the X-ray wavelength (0.154 nm), β is the full width at half maximum (FWHM), and θ is the Bragg angle.
The following equation’s X-ray density must be calculated19,20):
Where (MW) is the molecular weight, (NA) is the number of Avogadro, and (a) is the lattice constant. The lattice constant (a) values listed in Table 2 exhibit minor variations in response to variations in the x ratio.
Table 2.
Lattice constants, crystallite size and density of AlxCo1-xFe2O4 nanoferrites.
| x | a (Å) | ρ (g/cm3) | D (nm) |
| 0 | 8.3660 | 5.393 | 25.5 |
| 0.05 | 8.3690 | 5.387 | 29.5 |
| 0.10 | 8.3734 | 5.379 | 25.4 |
| 0.15 | 8.3694 | 5.387 | 25.0 |
| 0.20 | 8.3709 | 5.384 | 30.11 |
In general, the introduction of Al3+ ions (with ionic radii smaller than Co2+) is expected to result in a slight contraction in the lattice constant and a shift of the XRD peaks to larger 2θ angles; therefore, any slight shift in the peak positions can be explained by such lattice contraction resulting from ionic substitution. However, the observed changes in the table are small and nonlinear, suggesting that the influence of cation distribution, potential oxidation states, and internal stress may contribute to modifying a value in complex, non-monotonous ways.21)
The crystallite size values according to the Scherrer equation exhibit non-monotonous behavior: 25.5 nm at x = 0, rising to 29.5 nm at x = 0.05, then decreasing at x = 0.10-0.15 (≈25 nm), and then rising again to ≈30.11 nm at x = 0.20. This fluctuation can be explained by the interaction of several simultaneous mechanisms during the sol-gel preparation model and the calcination stage: First, the change in the Al3+ ratio affects the number of nucleation sites during gel formation and spontaneous combustion; increasing the number of nuclei typically results in smaller crystals, while faster growth conditions or coarsening yield larger sizes.22) Second, microstrains and lattice defects resulting from ionic substitution increase the width of XRD peaks (FWHM) and reduce the calculated D Scherrer; therefore, samples with higher strain will exhibit smaller Scherrer crystal sizes. Third, differences in cation distribution or the presence of surface aggregates/secondary phases in trace amounts may actually alter the grain growth mechanisms from one sample to another.23)
The slight variation in the arithmetic density reported in the table corresponds to small changes in the lattice constant and relative element abundance. The density changes are small and do not indicate the occurrence of large voids or density variations, but they support the idea that the chemical modification is minor within the measured range. Experimentally, whether the particle size from the FESEM images matches or conflicts with the Scherrer D values will help explain whether the differences are due to true crystal size (coherent domains) or to the fragmentation of the grains into multiple particles (agglomeration).24)
Finally, the XRD spectra and accompanying data suggest that Al substitution affects the lattice structure and crystallization quality in nonlinear ways: certain ratios (such as x = 0.05 or x = 0.20) may encourage the growth of larger grains or reduce local strain, while intermediate ratios increase strain and reduce the calculated crystal size.25)
3.2. Morphological analysis
The FESEM images in Fig. 3 with magnification pictures at 70 kX reveal that, on the surface morphology, the undoped sample (x = 0.0), semi-agglomerated irregularly shaped grains with a fairly close size are formed. The particles are well-associated and dense, which shows the agglomeration due to stronger inter-particle forces of the nanoparticles. This is compactness due to the natural growth of cobalt ferrite particles fabricated by the sol-gel technique, with the evaporation time and spontaneous combustion stage determining grain formation.26)
In the presence of small amounts of aluminum ions (x = 0.05), there is a distinct transformation in surface morphology. The particulates look rougher and are densely packed, with secondary agglomerates and heterogeneous aggregates, as well as platelet- or flake-like structures.27) This displacement means that the “crystallization and particle growth kinetics have been modified” as a consequence of the partial substitution of aluminum, causing more accentuated particulate agglomeration processes, perhaps the internal strain would be increased, which is reflected in the morphology.28)
Such changes may be attributed to the introduction of Al3+ ions, which have a radius smaller than that of Co2+, into the crystal lattice and inhomogeneous growth of the grains during calcination. This in its turn results in wider particle size distribution and larger aggregates. From a practical point of view, the higher porosity and roughness could be advantageous for certain applications (e.g. gas sensing or catalytic reactions) where a more effective surface becomes available, and adsorption sites are promoted.29)
In summary, the FESEM results above prove that Al substitution (even at very low doping of 5%) not only influences crystal size changing, as observed from XRD data, but also strongly alters particle morphology and agglomeration state, which may lend strength to establishing a link between nanoferrite microstructure and expected physical characteristics.30)
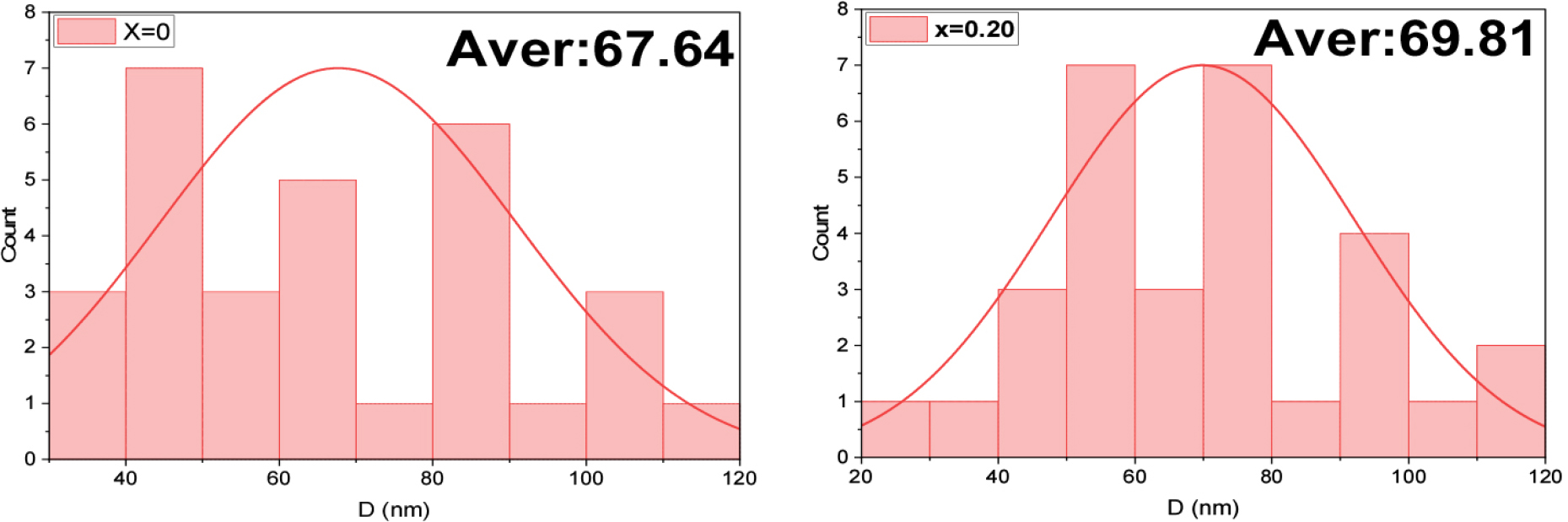
Fig. 4 for grain size analysis: when x = 0, i.e., without aluminum, it can be observed that the grain size distribution is very uniform and lies in the range 40-120 nm. Apparently, a sharp peak is found at the 67.64 nm mean value, signifying that the resulting grain size uniformity is excellent. This uniformity is proof of the preparation procedure and that the manufacturing technique employed was successful, by obtaining nanoparticles with satisfactory distribution and sizes very close to each other. This uniformity is due to the well-regulated reaction conditions and regulation of parameters such as temperature, pressure, and concentration of reactants during production.31)
The aluminum-containing sample (x = 0.20), on the other hand, revealed a shape difference in the grain size distribution of the histogram deduced from the FESEM image. The size of the average grain is in the range of 20-120 nm, and the average calculated grain size is around 69.81 nm. This distribution has larger scatters and more peaks than the sample above. This distribution of sizes is broad, implying the action on grain growth dynamics and the crystallization process of the substitution by aluminum. This may be due to alteration of surface energy or growth rates caused by aluminum ions in the crystal lattice.32)
When we contrast the two samples, it can be observed that the mean grain size has slightly grown from 67.64 to 69.81 nm by doping aluminum material. This small enhancement may be attributed to the fact that Al substitution promotes the process of grain growth, which is likely due to modifications in crystallization kinetics or stress induced through surface energy effects. However, the broader size distribution of the x = 0.20 sample results in non-uniform physical and magnetic properties of the material. Such inhomogeneity may decrease the performance of the material in many applications requiring property homogeneity.33)
This shift in grain size distribution can be related to the large reduction of magnetic properties seen previously. The broader distribution of the x = 0.20 sample size may induce many features, such as inhomogeneity due to differences in grain size and magnetic properties. This distribution also results in differences in the antiparallel field values between grains, where smaller grains have larger values of the antiparallel field than the larger ones. In addition, this inhomogeneous distribution also generates variation between the magnetization and magnetic reorientation mechanisms from grain to grain.34)
The grain size plays an important role in controlling the nanoscale properties of a material. Grains with smaller sizes cause a larger surface-to-volume ratio and large effects on the magnetic properties due to the surfaces. These effects consist of an enhanced surface magnetism and a dead layer-induced reduction in the saturation magnetization. Larger grains tend to be more bulk-like, i.e., with weaker surface effects and higher magnetic stability. Homogeneous size distribution induces uniform and regular magnetic properties, whereas inhomogeneously sized particles offer complex magnetic behavior.35)
3.3. Elemental analysis
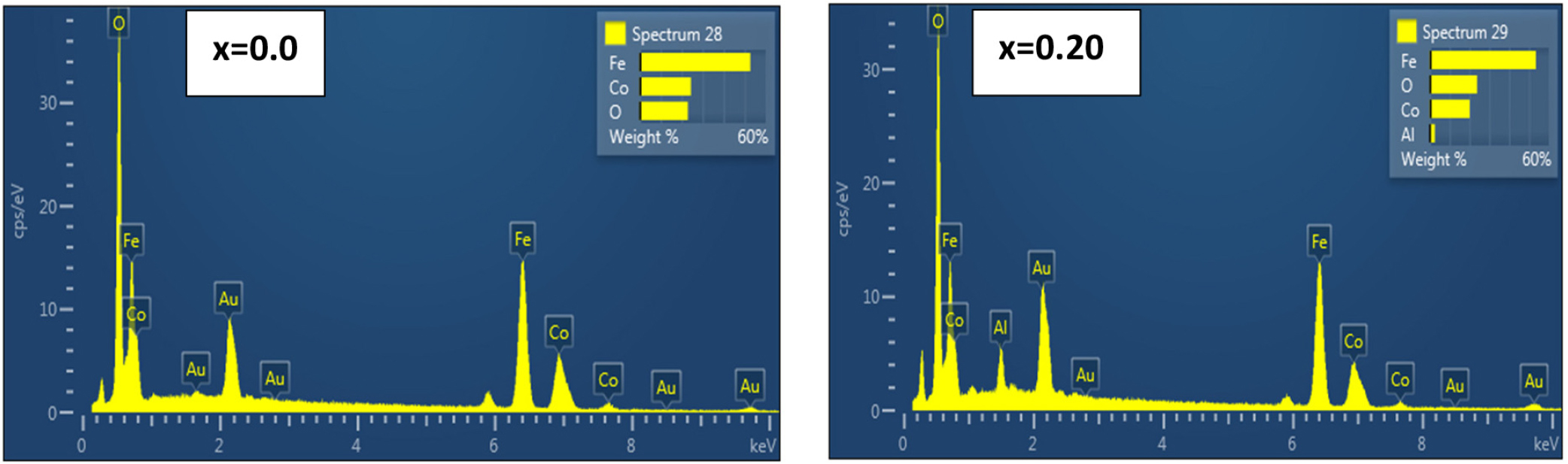
Fig. 5 of the EDX spectrum of the undoped (x = 0.0) sample reveals identifiable peaks for major elements as anticipated in the cobalt ferrite structure - iron (Fe), cobalt (Co), and oxygen (O). The given weight content (Fe ≈ 52.7 wt%, Co ≈ 24.4 wt%, O ≈ 22.9 wt%) corresponds to the stoichiometric composition of the ferrite-metallic part; high amounts of oxygen are present as an oxide. The respective atomic percentages reveal the excess of oxygen in atomic percentage (~51 %), as imposed by a higher number of oxygen atoms within the oxide lattice, while Fe and Co ratios reflect the specific cationic distribution into the spinel sites.36) Following the addition of a small amount of x = 0.05 aluminum, the EDX spectrum has an obvious peak existing at ≈2.53 wt% (≈3.25 at%), which should be Al. This feature verifies that aluminum is present as part of the general composition sample, even in values less than expected for this experiment. On the contrary, there is a small increase in the Fe ≈51.46 wt% and a small decrease in the Co ≈ 20.16 wt% observed with respect to the pure sample. This means that part of the Co2+ sites have been replaced by Al3+ ions or that there has been a partial redistribution of cations to balance charges, which is consistent with only slight variations in weight and calculated atomic (%) values.37)
The presence of oxygen even at high atomic percentages (~51-52 at%) is due to the nature of the compound (spinel oxide) and because EDX measurements detect what happens in near-surface layers. Hence, the figures are quite satisfactory in giving a general idea of the chemical composition. Minor Au peaks are also shown, which generally originate from the coating films for microscopic preparation (coating), while these are not structural constituents of the specimen.38) The absence of secondary phases in XRD patterns, combined with the detection of Al signals in EDX spectra and systematic changes in lattice parameters and magnetic properties, strongly suggests that Al3+ ions are incorporated into the spinel lattice rather than forming segregated Al2O3 clusters.39)
3.4. Magnetic Properties
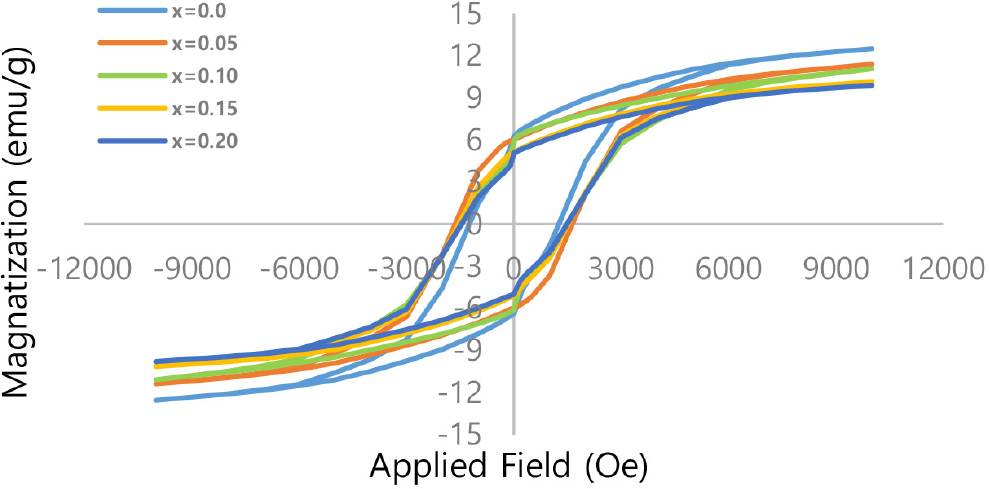
Hysteresis loops for the nanoferrite samples AlxCo1-xFe2O4 (x = 0, 0.05, 0.10, 0.15, and 0.20) at room temperature are shown in Fig. 6. The Ms value decreases with the increasing doping concentration of Al3+ ions for Co2+ ions. The Ms value reduced from 12.5 emu/g at x = 0.00 to 9.9 emu/g at x = 0.20, as shown in Table 3. This behavior can be interpreted in terms of the magnetic moment of replacing ions. The aluminum ion that is Al3+ is diamagnetic, where the magnetic moment value of it is equal to 0. Replacement of the powerful magnetic moment-possessing cobalt ion (Co2+) diminishes the resultant general formula for ferrite. This magnetic dilution directly results in a reduction of the overall magnetization of the material, as is indicated by its decreasing Ms.40)
Table 3.
The magnetic properties of the AlxCo1-xFe2O4 nanoferrites.
| x | Ms (emu/g) | Mr (emu/g) | Mr/Ms | Hc (Oe) |
| 0.0 | 12.5 | 6.2 | 0.496 | 1250 |
| 0.05 | 11.4 | 6 | 0.526 | 1650 |
| 0.10 | 11 | 5.87 | 0.533 | 1500 |
| 0.15 | 10.1 | 5.13 | 0.508 | 1560 |
| 0.20 | 9.9 | 5.07 | 0.512 | 1495 |
The Mr value is found to have a similar tendency of saturation magnetization with increasing alloying elements, i.e., aluminum content (x). This reduction is anticipated since Mr is directly proportional to Ms.41) However, the most important was the squareness ratio (Mr/Ms), which varied between 0.496 and 0.533. These values prove that all samples have the same random magnetic domain orientation (magnetocrystalline anisotropy). The Stoner-Wohlfarth model gives Mr/Ms~0.5 for single-domain particles with cubic symmetry (and random distribution of easy axis configuration). The slightly increased value at x = 0.10 indicates that little change in the particle size distribution or improvement of interaction between particles occurs at this ratio.42)
Hc shows interesting behavior, increasing from 1,250 Oe at x = 0.00 to 1,650 Oe at x = 0.05, where it reaches its peak value; then it decreases slightly later to be 1,495 Oe at x = 0.20. Coercivity is essentially a function of magnetic crystal anisotropy, crystal dimensions, and lattice stress. The first stage of the Hc increase was assigned to the creation of internal stress and deviations in the crystal lattice due to small Al3+ ions that affect magnetic walls’ movement and make demagnetization harder, rendering higher values of Hc. The following decrease of Hc after an initial enhancement with increasing x likely stems from the overwhelmingly minimized contribution to magnetic crystal anisotropy as a result of the substitution of Co2+ ions (having a large anisotropy) by Al3+ ions (anisotropy = 0), which allows for easier magnetization reversal and decreases coercivity.43)
The general tendency is that non-magnetic aluminum is substituted for cobalt ferrites, and it weakens the basic magnetic properties, such as Ms and Mr, but it has a complicated effect on the coercivity by manipulating crystal anisotropy and lattice stress, which could open the possibility toward controlling the material properties.44)
In this work, the results of the current research were compared with published research, and it was found that the Ms values were within the range of 9.9-12.5 emu/g, much lower than the value reported for pure CoFe2O4 ferrite (~50-80 emu/g, even growing up to 69.67 emu/g in some reports), as shown in Table 4. This reduction can be ascribed to the non-magnetic behavior of Al3+ ions; compared with that for Co2+ and Fe3+, the magnetic exchange between Co2+ and Fe3+ is decreased by replacing these cations in the B sites along with a significant decrease of Ms.45) The low calcination temperature (600 °C), which may result in incomplete crystallization and enhanced spin disorder, and the nanoscale particle size, which encourages surface spin canting and magnetically dead layers due to the high surface-to-volume ratio, are both responsible for the lower saturation magnetization values (9.9-12.5 emu/g) seen in this investigation.46) Furthermore, A-B superexchange contacts are weakened when non-magnetic Al3+ is substituted for magnetic Co2+/Fe3+, and long-range magnetic ordering is further disrupted by potential cation redistribution and internal strain. Secondary non-magnetic phases were not found by XRD investigation; however, surface disorder or nanoscale amorphous areas cannot be completely ruled out.53)
Table 4.
Comparison of the magnetic properties of the current research results with the results of previous research.
| Composition | Ms (emu/g) | Mr (emu/g) | Mr/Ms | Hc (Oe) | Reference |
| AlxCo1-xFe2O4 (x = 0.0-0.20) | 9.9-12.5 | 5.07-6.2 | 0.49-0.53 | 1250-1650 | This work |
| CoFe2O4 nanoparticles | ~50-80 | ~20-40 | ~0.40-0.50 | 200-2000 | 47) |
| CoFe2O4 (T = 300, 500, 1000 °C) | 42.42-78.06 | 8.94-27.4 | 0.21-0.35 | ~434-461 | 48) |
| CoFe2O4 (co-precipitation method) | 69.67 | 16.4 | 0.235 | 775.27 | 49) |
| NiFe2O4 nanoparticles | 0. 838-57 | 0.0173-15.2 | 0.021-0.268 | 32.77-148.8 | 50) |
| Al1-xZnxFe2O4 (x = 0.0-1.0) | ~0.54-2.1 | ~0.01-0.12 | ~0.005-0.043 | ~10-3703 | 51) |
| AlFe2O4 | 16.07 | 0.1287 | 0.008 | 8.67 | 52) |
Regarding the Mr, it may be mentioned that in this study Mr values were found between 5.07-6.2 emu/g, like some studies for low Ms but smaller compared to pure CoFe2O4 (up to 16.4 emu/g or even higher). It tells us that the order of the doping ratio restricts the magnetism pinned after removal of the external field. The Mr/Ms ratios range between 0.49-0.53, average values of Mr/Ms, and indicate that the sample is still quite ferrimagnetic material. However, the relative enhancement in this ratio with respect to the undoped CoFe2O4 (0.21-0.35), justified a substantial increase in stability of the magnetic field due to ionic distribution change.54)
The Hc varied from 1,250 to 1,650 Oe, higher than CoFe2O4 (~30-150 Oe) but lower than some highest values reported for CoFe2O4 for particular preparation conditions. This partial hardness would imply that the addition of aluminum makes selection for some magnetic hardness over soft substances like CoFe2O4, but also suppression of magnetic strength reaching CoFe2O4.55) In general, the results of this work indicate that for CoFe2O4, Al doping resulted in a decrease of Ms while keeping satisfactory Hc and Mr/Ms, which could provide some references to apply them in special magnetic fields requiring moderate magnetic hardness and controllability. In contrast with other studies, the AlxCo1-xFe2O4 system exhibits a compromise between the hard and soft magnetic properties and presents some potential applications in electronic and magnetic characteristics that do not require very high Ms.56)
It should be noted that the hysteresis loops presented in this study correspond to single-cycle VSM measurements performed at room temperature, following standard experimental protocols. The loops obtained are well-defined, symmetric, and closed, indicating a stable magnetic response and reliable measurement conditions.57) The consistency of magnetic parameters (Ms, Mr, and Hc) across different compositions further supports the reproducibility of the magnetic behavior.58)
4. Conclusion
In summary, the paper highlights the following: It was shown that nanosized powder samples of aluminum-cobalt ferrite AlxCo1-xFe2O4 (x = 0.0-0.20) can be effectively obtained by the self-combustion sol-gel method with a homogeneous contamination-free product. Structurally, a pure cubic spinel structure was observed for all prepared samples according to XRD analysis, along with little variation in the crystal lattice constant due to the increase of Al content. This is because the magnetic cobalt ions were substituted by non-magnetic aluminum ions having smaller ionic diameters.
Morphologically, FESEM observations indicated a clear change in the samples’ grain size due to the presence of aluminum. An enhancement of the average grain size and a heterogeneous distribution were found, indicating an evident effect of aluminum on the crystal growth mechanism and crystallization dynamics. With respect to magnetic properties, the saturation and Mr values reveal a continuously decreasing tendency as aluminum content is increased, mainly because of the diluting influence due to the substitution of non-magnetic ions for magnetic ones. By contrast, opposition to the coercive field showed an anomalous behavior with increasing value at low aluminum content and a decrease with increasing aluminum; such a characteristic is due to competition between the internal stress and loss of crystalline anisotropy.
This work demonstrates the possibility of precisely controlling the structural and magnetic properties of these compounds by adjusting the aluminum content, offering a promising avenue for designing materials with controllable properties.