1. Introduction
Energy demand continues to rise every year because of population growth, and it mostly comes from fossil fuels. However, the fossil fuel supply is inherently limited. Therefore, more research is needed to produce alternative energy using renewable raw materials globally, such as biodiesel. Biomass, the energy source to produce biodiesel, can be classified into third generation. The first generation is based on food crops, while the second is from nonfood crops. However, these two generations have weaknesses, among which they lead to soil pollution, competition for food needs, extensive land requirement and forest areas reduction.1) The third generation is considered to be more beneficial because it is derived from the use of possible aquatic biodiversity micro-organisms, namely microalgae. Biomass contains a large quantity of oil, usually 20-50 % of its weight, and has a fast growth rate. In addition, the land use is environmentally friendly and effective.2-4) However, the release of oil from microalgae cells is blocked by rigid cell structures.
Microwave-assisted extraction may be an option due to its ability to penetrate the structure of the cell and efficiently remove lipids.5) When microalgae cells are exposed to a different microwave frequency (around 2,450 MHz), cell molecules will produce rapid oscillations in rapidly oscillating electrical fields, resulting in the production of heat due to the frictional force of inter and intramolecular motion.6) This approach illustrates the advantages of using the microwave, i.e., short reaction times, low operating costs, and effective extraction.7,8) The interaction between triglycerides, solvent, and microwave leads to a substantial reduction in activation energy.9)
Methanol is a fuel that cogently absorbs microwave. However, the O–H groups attached to large molecules typically undergo more localized rotation, dominate the microwave spectrum, and also produce excessive local heating, which helps to complete the reaction more quickly.10) Furthermore, methanol has two functions in microwave-assisted biodiesel production, as a lipid extraction solvent in the microalgae and transesterification reactant.11)
Several methods that have been developed biodiesel production from microalgae biomass are direct transesterification of microalgae using conventional technique with H2SO4 as a catalyst; extraction of microalgae using Bligh and Dyer method; Soxhlet extractor ultrasoundassisted extraction; supercritical fluid extraction and pressurized fluid extraction followed by esterification and transesterification reaction; acid or alkali-catalyzed biodiesel production using transesterification of C. vulgaris and Spirulina sp.; alkali-catalyzed in-situ transesterification of Spirulina sp. using methanol; and microwave-assisted pyrolysis of Chlorella sp. with microwave reception enchancer.12) This present study developed a novel method in the biodiesel preparation performed by simultaneously extraction and transesterification in a microwaveassisted reaction of C. vulgaris to produce biodiesel.
2. Experimental Procedures
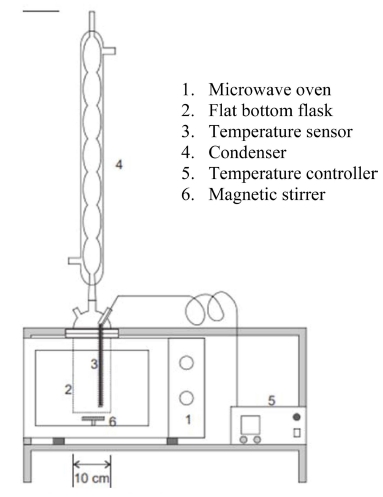
The C. vulgaris was dispersed to the mixture of methanol as solvent and KOH as catalyst and irradiated by a microwave (800 W) at 70 ºC with various microalgaeto- methanol (w/v) ratios of 1:6, 1:9, 1:12, and 1:15 (60, 90, 120, and 150 mL volume of methanol); reaction time of 20, 30, 40, and 50 min and catalyst concentration of 1, 2, and 3 wt%. The mixture was centrifuged for 1 h at a speed of 2,000 rpm. The supernatant was then evaporated. The FAME yield was determined using the following formula.12)
The product with the highest FAME yield was assessed at a various biomass-to-methanol ratio, reaction time, and KOH catalyst concentration. The FTIR Spectrometer (Shimadzu IR Prestige-21) was used to obtain the infrared (IR) spectra. KBr was used to process the microalgae into pellets, and the product was dropped on a thin plate NaCl with a scan range of 4000 - 400 cm−1 at room temperature.
The GC-MS test was performed by Shimadzu QP-2010 with a capillary column Rtx-5MS using He carrier gas at 70 °C. The identification of the component was determined using a database library. Each component was quantified based on the percentage of area at each GC-MS peak. An analytical test using 1H-NMR (JEOL Delta spectrometer) was performed by dispersing 100 μL product in 700 μL deuterated chloroform (CDCl3). The mixture was put into the NMR tube and tested on an autosampler. The SEM instrument (Phenom ProX) was performed for analysis of the surfaces of C. vulgaris before and after microwaveassisted in-situ transesterification reaction.
3. Results and Discussion
3.1 Microwave-assisted In-situ Transesterification
In an in-situ transesterification reaction, methanol was used as a solvent due to its high dissipation factor. It gives low ability to block microwave and high ability to dissipate the microwave into heat.13) However, in-situ transesterification converts triglycerides directly from microalgae to methyl esters, and since methanol acts as lipid extraction solvent and transesterification reagent. The use of a homogenous catalyst can produce high conversions of biodiesel due to its susceptibility properties to microwave irradiation.14,15)
In this study, the microwave oven reactor was modified by a reflux system to maximize the reaction process. The condenser causes the solvent to evaporate when it reaches its boiling point temperature so that it becomes condensed, and the solvent reacts again. Therefore, the solvent quantity remains, and the maximum reaction occurs without a shortage of solvent to produce the maximum product. In this reactor, we have added temperature controller to control the temperature change. This type of microwave oven reactor has been used for the transesterification reaction of bio-crude oil from spirulina (Arthrospira platensis) into biodiesel and successfully converted 88% of biodiesel yield.16)
3.2 Effect of Various Biomass-to-methanol Ratios
The effect of various biomass-to-methanol ratios (reaction time of 20 min; the catalyst of 2 %) on FAME yield is presented in Fig. 3(a). The results confirm that the optimum biomass-to-methanol ratio of 1:12 (g/mL) produced the highest yield of FAME (6.42 %). The reaction at biomass-to-methanol ratios of 1:6 and 1:9 was found to be ineffective in shifting the reaction equilibrium and gave lower amount of FAME due to the rigid cell structure.
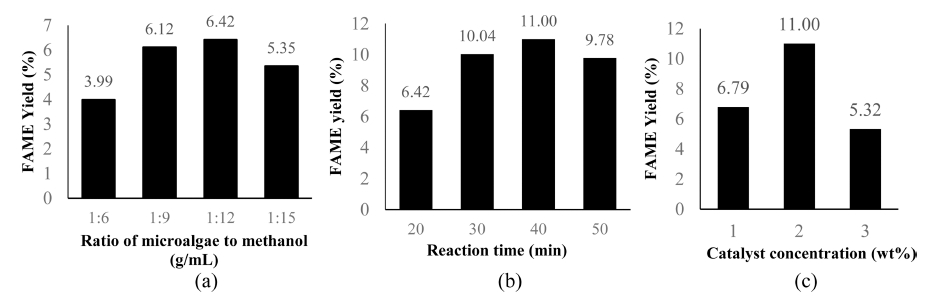
Fig. 3
The effect of various (a) biomass-to-methanol ratios, (b) reaction time, and (c) catalyst concentrations on FAME yield from microwave-assisted in-situ transesterification reaction.
In this study, methanol acted as an extraction solvent for transesterification reaction. With a limited amount of solvent, it only served to extract lipids from the cell, while the remaining reacted with triglycerides to form a small amount of FAME. The biomass-to-methanol ratio needs to be increased to enlarge the contact area of methanol with microalgae and extracted lipids to produce higher FAME. However, a high amount of methanol can cause too much microwave irradiation to be absorbed by the solvent, leading to inefficient extraction of lipids from microalgae. According to Zhang et al.,17) the excessive amount of methanol can also reduce the catalyst concentration, leading to product yield decreasing.
3.3 Effect of Various Reaction Time
The effect of various reaction times (biomass-to-methanol ratio 1:12; the catalyst of 2 wt%) on FAME conversion is presented in Fig. 3(b). A more prolonged reaction time led to a longer reactant exposed to irradiation from the microwave. A reaction time for 40 min produced the highest amount of FAME (11 %). However, an excessive reaction time significantly increased the reaction heat, thus causing solvent removal and by-product formation. The thermal effect from microwave enhances the ability of methanol to extract the lipids from suspended microalgae biomass. The excessive microwave induces the penetration of the cell, thus force the lipids out to the solvent.
3.4 Effect of Various Catalyst Concentration
The mechanism of catalyst in an in-situ transesterification reaction takes place in several steps. First, the alkoxide ion attacks to the carbonyl carbon of the triglyceride molecule which results the formation of a tetrahedral intermediate. Then in the second step, the reaction between tetrahedral intermediate and an alcohol produces the alkoxide ion. In the final step, the rearrangement of tetrahedral intermediate produces an ester and a diglyceride. In a similar way, diglyceride is transesterified to form methyl ester and monoglyceride, which is further converted to methyl ester and glycerol.
The effect of various catalyst concentrations (biomassto- methanol ratio of 1:12; reaction time of 40 min) on FAME yield is shown in Fig. 3(c). It shows that the use of catalyst concentration of 2 wt% produced the highest amount of FAME (11 %). However, a higher catalyst concentration was not able to increase the reactions effectively. This maybe due to the presence of various organic compounds from the extraction of microalgae biomass, such as lipids, olefins, fatty alcohols, phytols, sterols, and pigments.5)
Rushan et al.18) reported that FAME yield of 14.92 % by the extraction of C. vulgaris using methanol and chloroform as the solvent, and the transesterification reaction over hydrochloric acid catalyst using methanol and hexane as solvent. Purkan et al.19) also reported that 13.68 % of FAME yield was successfully produced by the extraction of C. vulgaris lipid using ethanol and hexane as a solvent, and the transesterification reaction over sulfuric acid using methanol as solvent. However, in this study, the microwave-assisted in-situ transesterification method offered a cost-effective and energy-efficient storage by using a one-step transesterification process of C. vulgaris to produce biodiesel.
3.5 FTIR Characterization
Fig. 4(a) shows FTIR spectrum of microalgae after extraction. The peak at 3,425 cm−1 is attributed to O–H streching vibration of fatty acid. Peaks at 2,924 and 2,854 cm−1 are corresponded to symmetric and asymmetric stretching vibrations of –CH2–, respectively. These vibrations confirm the presence of lipid in microalgae. The peak at 1,651 cm−1 is ascribed to the presence of C=C stretching vibration, while another peak 1,450 cm−1 is assigned to C–H bending vibration. The peak at 1,111 cm−1 is ascribed to asymmetric stretching vibration of C–O.
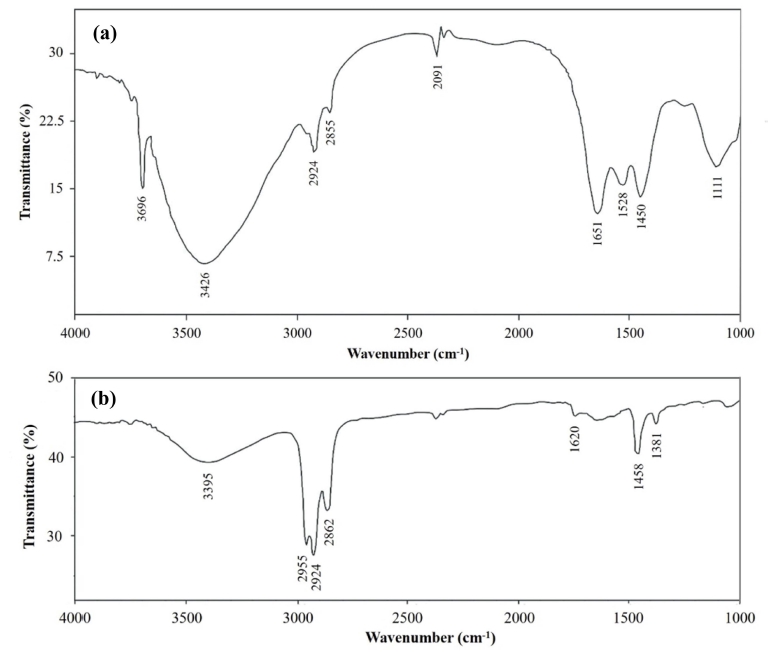
Fig. 4
FTIR spectra of (a) microalgae after extraction and (b) microwave-assisted in-situ transesterification product.
Fig. 4(b) shows FTIR spectrum of microwave-assisted in-situ transesterification product. The presence of unsaturated chains in methyl ester is confirmed by the peak at 2,954 cm−1 associated with olefin group (=CH–). The peak at 2,924 and 2,862 cm−1 are attributed to C–H vibration in methyl esters. The peak at 1,740 cm−1 is ascribed to asymmetric stretching vibration of C=O. The absorption peaks at 1,458 and 725 cm−1 are assigned to – CH2– vibration. The peak at 1,381 and 1,165 cm−1 are resulted from O–CH2 and C–O vibrations, respectively.
3.6 GC-MS Characterization
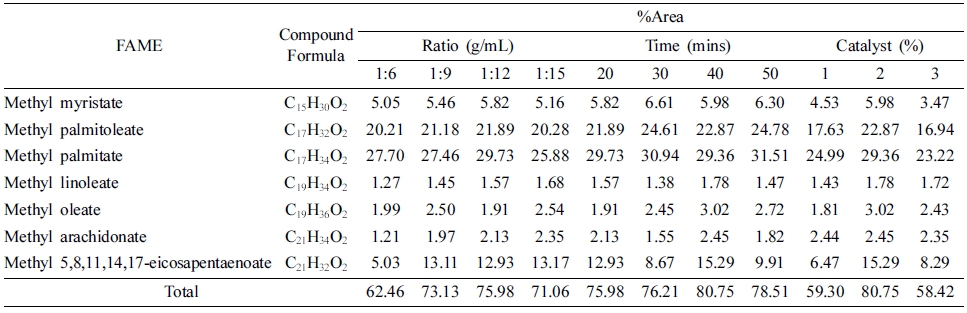
According to GC chromatogram of microwave-assisted in-situ transesterification product at an optimum condition (biomass-to-methanol ratio of 1:12, reaction time of 40 min and catalyst concentration of 2 %), the product contains of phytols, olefin, fatty alcohol, sterol, pigment, vitamin, saturated and unsaturated FAME.
According to GC-MS data, the product contains of methyl tetradecanoate (methyl myristate), methyl 9- hexadecenoate (methyl palmitoleate), methyl hexadecanoate (methyl palmitate), methyl 9,12-octadecadienoate (methyl linoleaate), methyl 9-octadecenoate (methyl oleate), methyl 5,8,11,14-eicosatetraenoate (methyl arachidonate) and methyl 5,8,11,14,17-eicosapentaenoate. Table 1 lists the total percentage of FAME compounds with various biomassto- methanol ratio, reaction time, and catalyst concentration.
3.7 1H-NMR Characterization
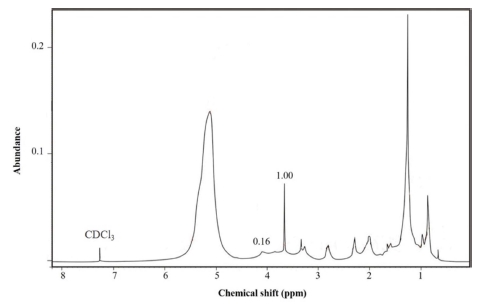
Fig. 5 presents 1H-NMR of microwave-assisted in-situ transesterification product. The peak at 4.1 ppm is indicated to the presence of triglycerides20) with a relatively low integration value of 0.16, while a peak at 3.66 ppm is ascribed to proton from methoxy group (–OCH3). Methoxy is a typical biodiesel peak with an integration value of 1.00. Methyl ester conversion was calculated using the following formula.21) The result indicates that the triglyceride was successfully converted into methyl ester with a total conversion of 77.64 %.
3.8 SEM Characterization
Fig. 6 shows SEM image of C. vulgaris microalgae and microwave-assisted in-situ transesterification product. It can be seen that the surface morphology of microalgae was significantly changed. It indicates that the cell of microalgae had been damaged by microwave irradiation, then extract lipids from microalgae to the solvent, and followed by lipid transesterification with the reagent.22)
4. Conclusions
Microwave-assisted in-situ transesterification of C. vulgaris was successfully conducted with methanol as a reagent. Biomass-to-methanol ratio of 1:12, reaction time of 40 min, and catalyst concentration of 2 wt% were determined to be the optimum condition to obtain the highest amount of FAME (11 %). The transesterification reaction of C. vulgaris produced FAME conversion of 77.64 %. Biodiesel consisted of methyl myristate, methyl palmitoleate, methyl palmitate, methyl linoleate, methyl oleate, methyl arachidonate, and methyl 5,8,11,14,17 eicosapentaenoate.