1. Introduction
2. Experimental Procedure
2.1. Materials
2.2. Fabrication of RSD-incorporated PCL/chitosan films
2.3. FT-IR spectroscopy
2.4. FE-SEM observation
2.5. Measurement of contact angle
2.6. Assessment of cell responses
2.7. Statistical analysis
3. Results and Discussion
3.1. Fabrication of PCL/chitosan composite films
3.2. Observation of morphological changes
3.3. Molecular and hydrophilicity changes
3.4. Cellular responses
4. Conclusion
1. Introduction
The use of polymeric composites in drug delivery systems has become an innovative strategy to enhance controlled drug release and bioavailability. These systems typically involve blending two or more polymers to form matrices that exhibit customized mechanical properties, drug release profiles, and biocompatibility. Polymeric blends increase drug encapsulation efficiency, allowing for fine-tuning of drug release rates through the combination of hydrophilic and hydrophobic polymers.1) For example, blends of sodium alginate and polycaprolactone (PCL) have been utilized for controlled drug release in cancer therapies, providing sustained release while minimizing side effects.2) Similarly, incorporating nanoparticles into polymeric composites has shown promise in enhancing drug targeting, as seen with curcumin-loaded nanocomposite systems that improve drug stability and efficacy.3)
PCL is a semi-crystalline, biodegradable polymer valued for its flexibility, slow degradation rate, and compatibility with bioactive molecules. Its mechanical properties are influenced by its molecular weight, with higher weights resulting in greater tensile strength and elongation. PCL has a melting point around 60 °C and a low glass transition temperature, making it ideal for soft and flexible applications, such as drug delivery and tissue engineering. Due to its biocompatibility and slow degradation, PCL is suitable for long-term drug delivery, particularly for hydrophobic drugs.4,5) Blending PCL with other polymers or nanoparticles allows for customization of mechanical and degradation properties, thereby optimizing drug release.6,7) However, PCL’s hydrophobicity limits its ability to encapsulate and release hydrophilic drugs efficiently.8,9)
Chitosan, a natural biopolymer derived from chitin, is mainly sourced from the exoskeletons of crustaceans. It is widely recognized for its biocompatibility, biodegradability, and non-toxicity, making it an excellent candidate for drug delivery systems.10) Chitosan can form films, gels, and nanoparticles, with its chemical properties largely defined by the reactive amino groups, which allow for various chemical modifications that enhance solubility and drug binding.11) Moreover, chitosan’s ability to load charged drugs makes it ideal for controlled and targeted drug release, which is essential for therapies requiring sustained drug levels.12,13) Its strong adhesion to mucosal tissues enhances drug absorption and retention, making it particularly effective in nasal and oral drug delivery systems.
Bisphosphonates are a common class of drugs used to treat bone resorption disorders like osteoporosis. They inhibit osteoclast activity, which is responsible for bone breakdown. Bisphosphonates bind strongly to hydroxyapatite crystals in the bone, preventing bone loss by reducing osteoclast survival and activity.14) Among these, risedronate (RSD) is known for its high potency in reducing bone resorption by inhibiting the enzyme farnesyl pyrophosphate synthase (FPPS) in the mevalonate pathway, which is crucial for osteoclast function.15) However, RSD’s oral bioavailability is low, which has led to the exploration of alternative delivery methods. Nanoparticles, for instance, improve the bioavailability and targeting of RSD to bone tissue, enhancing therapeutic outcomes and reducing side effects.16,17) Besides its effect on osteoclasts, RSD also influences osteoblasts by promoting their survival through reduced production of pro-inflammatory cytokines and reactive oxygen species, thus creating an environment conducive to bone formation.18,19) Moreover, RSD enhances pre-osteoblast differentiation into mature osteoblasts, facilitating bone remodeling and repair, thereby making it a comprehensive therapeutic agent for osteoporosis.20)
2. Experimental Procedure
2.1. Materials
PCL (704605, Mn 45,000), Chitosan (448877, midium molecular weight), and risedronate sodium (1604610) were purchased from Sigma-Aldrich. MG63 preosteoblast cells (ATCC) were used to analyze cellular responses.
2.2. Fabrication of RSD-incorporated PCL/chitosan films
PCL and chitosan were dissolved in 1 M acetic acid at 1 % (w/v) to prepare the respective solutions. Blends of these polymers were created to achieve PCL ratios of 25 %, 50 %, and 75 % (w/w). The blended solutions were poured into 4-well plates and solvent-dried at 37 °C for 19 h. Prior to film formation, RSD was added to the PCL/chitosan blends at concentrations of 10-7, 10-6, 10-5, and 10-4 M to prepare the final solution and referred to as RSD(-7), RSD(-6), RSD(-5), and RSD(-4).
2.3. FT-IR spectroscopy
Fourier-transform infrared (FT-IR) spectroscopy was conducted using an FT-IR spectrometer to analyze the RSD-incorporated PCL/chitosan composite films. The absorption spectra were recorded between 4,000 cm-1 and 400 cm-1, with a focus on the region between 1,800 cm-1 and 1,200 cm-1 to identify characteristic peaks of PCL, chitosan, and RSD.
2.4. FE-SEM observation
The morphology of the PCL/chitosan composite films at different temperatures was observed using field emission scanning electron microscopy (FE-SEM, S-4300, Hitachi, Japan). The films, prepared at 37 °C and 55 °C, were sputter-coated with gold before observation. The acceleration voltage was set to 20 kV.
2.5. Measurement of contact angle
The hydrophilicity of the composite films was assessed by measuring the water contact angle using a Phoenix-300 water contact angle analyzer. Measurements were taken with 10 µL of water placed on the surface of each film containing PCL at 25 %, 50 %, and 75 %, and RSD concentrations ranging from 10-7 to 10-4 M.
2.6. Assessment of cell responses
MG63 cells were used to evaluate cellular responses. The cells were seeded at a density of 1 × 104 cells/mL for proliferation and 2.5 × 104 cells/mL for alkaline phosphatase (ALP) activity assays. The cells were cultured in an incubator at 37 °C under 5 % CO2 and 95 % humidity. The cell population was evaluated using cell counting kit-8 at 3 and 7 d after seeding and ALP obtained from cells lysate at 1 and 2 wk after seeding was incubated with p-Nitrophenyl phosphate (pNPP). Cell population was evaluated using cell counting kit-8 and ALP obtained from cells lysate was incubated with pNPP. The morphological changes were observed using optical microscopy. The cells were cultured in Dulbecco’s Modified Eagle Medium (DMEM) supplemented with 10 % fetal bovine serum and 1 % antibiotic-antimycotic solution.
2.7. Statistical analysis
Data were analyzed using a Student’s t-test to determine statistical significance. Differences between groups were considered significant if the p-value was less than 0.05. All results are expressed as mean ± standard deviation (SD). Statistical analysis was performed using OriginLab 2023b software, and p-values are reported to indicate the level of significance (*p < 0.05).
3. Results and Discussion
In this study, hydrophilic and hydrophobic polymers, PCL and chitosan, were blended in various ratios, and the resulting films were evaluated for their miscibility, morphology, and hydrophilicity. Furthermore, the cellular response of MG63 cells was assessed in the presence of RSD incorporated at different concentrations to optimize release for bone repair. The range of RSD concentrations was selected based on its cytotoxicity. The previous studies reported the cytotoxic limits of RSD to be approximately 5 × 10-8 to 10-5 M.21,22,23) Therefore, we set the RSD concentration range at 10-7 to 10-4 M for incorporation into various film compositions.
3.1. Fabrication of PCL/chitosan composite films
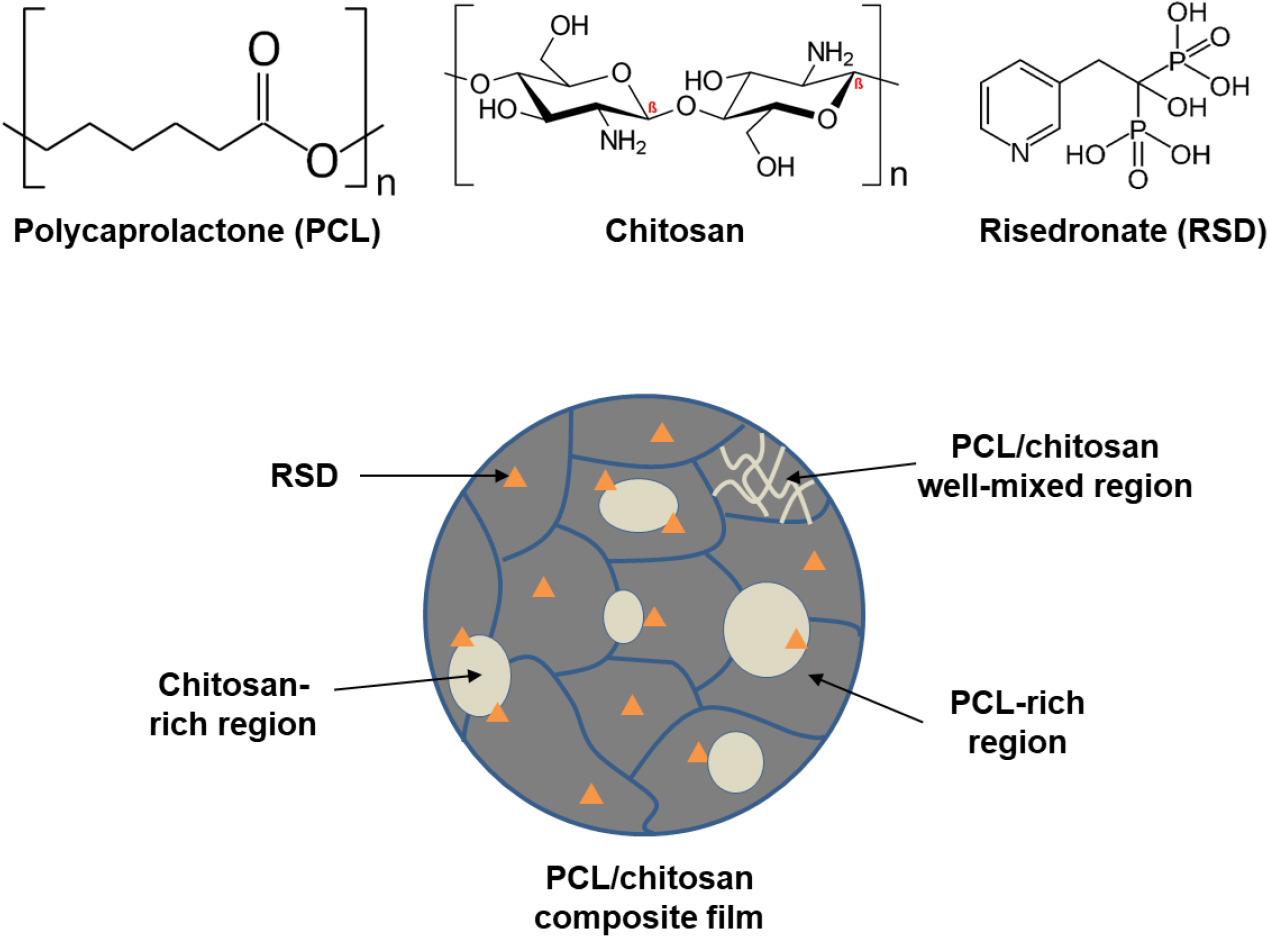
Fig. 1 presents a schematic illustration of the RSD-incorporated PCL/chitosan film, along with its chemical structure. Given the differences in miscibility, hydrophilicity, and solvent susceptibility between PCL and chitosan, optimizing the blending conditions and ratios is crucial for achieving uniform mixing and efficient drug loading. PCL’s aliphatic chains and oxygen groups facilitate hydrophobic interactions and hydrogen bonding, while chitosan’s amine and hydroxyl groups promote ionic interactions and hydrogen bonding. These distinct properties lead to the formation of PCL- or chitosan-rich regions during the blending and solvent evaporation process, necessitating careful control of the conditions to ensure a homogeneous film.
3.2. Observation of morphological changes
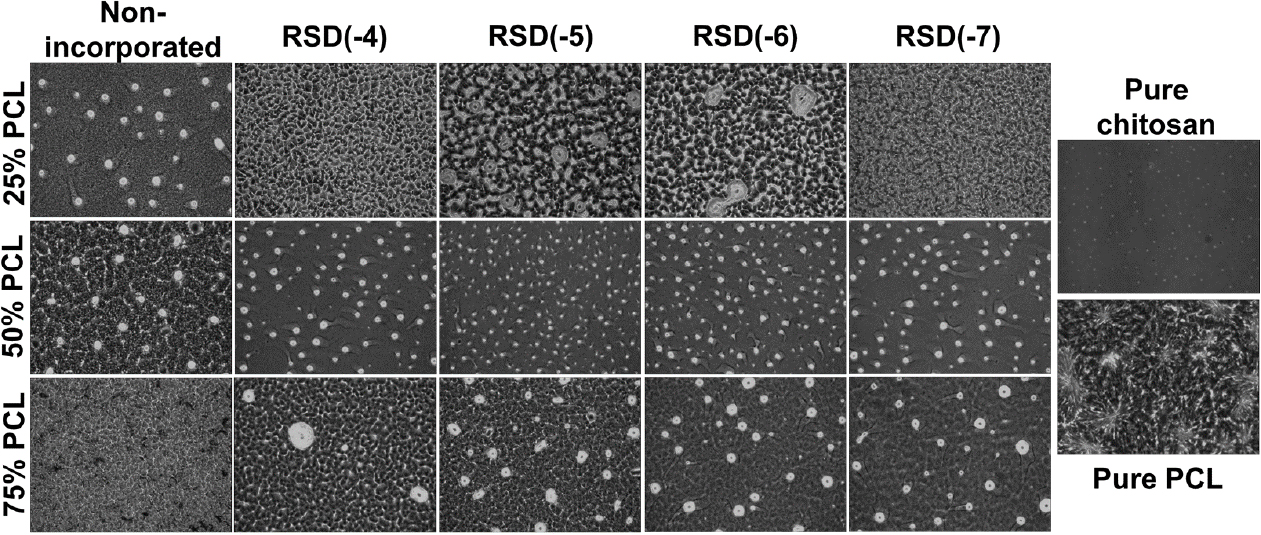
Fig. 2 shows optical microscopy images of PCL/chitosan composite films with varying PCL content (25 %, 50 %, and 75 %) and RSD concentrations ranging from 10-7 to 10-4 M. Pristine chitosan formed small, spherical domains, while pristine PCL displayed typical spherulite formation.
In the 25 % PCL blend, RSD influenced the chitosan-rich regions, with the RSD(-4) concentration producing a relatively homogeneous morphology, minimizing chitosan entanglement. In the 50 % PCL films, distinct chitosan-rich regions were present across all samples, with similar size and density. For the 75 % PCL films, both the non-incorporated and RSD(-4) samples exhibited well-mixed morphologies with fewer chitosan-rich areas. In contrast, the RSD(-5), RSD(-6), and RSD(-7) films showed greater phase separation in the chitosan regions, likely due to phosphate and nitrogen groups in RSD’s pyridine ring, which may disrupt chitosan aggregation via ionic and hydrogen bonding. Blending PCL and chitosan poses significant challenges due to their differing chemical and physical properties. A major issue stems from the immiscibility between hydrophobic PCL and hydrophilic chitosan, leading to poor miscibility and inhomogeneous blends. This immiscibility can result in mechanical weaknesses and non-uniform drug dispersion, negatively impacting the drug release profile.24,25) Additionally, PCL and chitosan have significantly different degradation rates; PCL degrades slowly, while chitosan degrades more rapidly. This discrepancy can compromise structural integrity over time, resulting in inconsistent drug release and variable therapeutic outcomes.26)
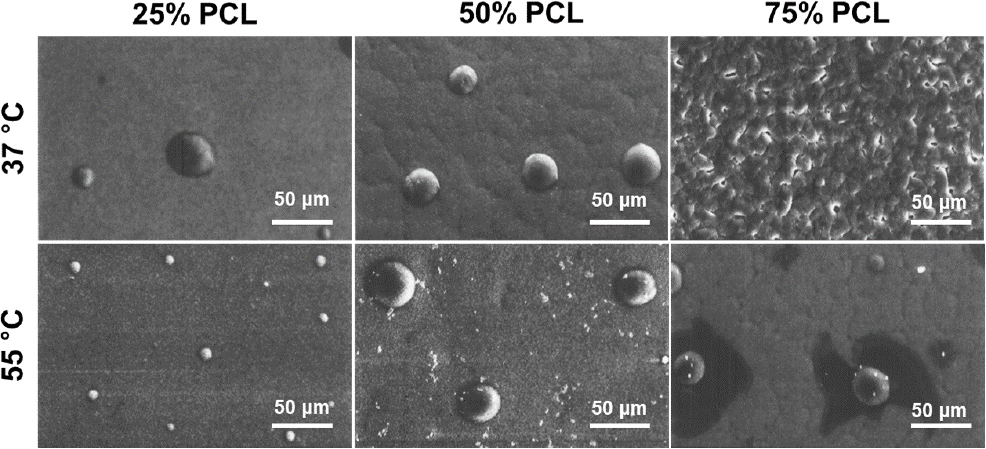
Fig. 3 presents FE-SEM micrographs illustrating the temperature-dependent morphological changes in PCL/chitosan films at 37 °C and 55 °C. Films containing 25 %, 50 %, and 75 % PCL exhibited similar structural patterns, with distinct chitosan-rich regions and continuous PCL regions. At 37 °C, only the 75 % PCL blend showed relatively uniform mixing, while the other compositions still displayed prominent chitosan-rich domains. The semi-crystalline nature of PCL led to the formation of wrinkle-like boundaries in films with more than 50 % PCL at both temperatures. These findings highlight the crucial role of both mixing and drying temperatures in achieving homogeneous composite films, which are necessary for effective and consistent drug delivery systems. The immiscible precipitation observed in films dried at room temperature further emphasizes the need for controlled drying conditions to ensure uniformity (in-house data).
3.3. Molecular and hydrophilicity changes
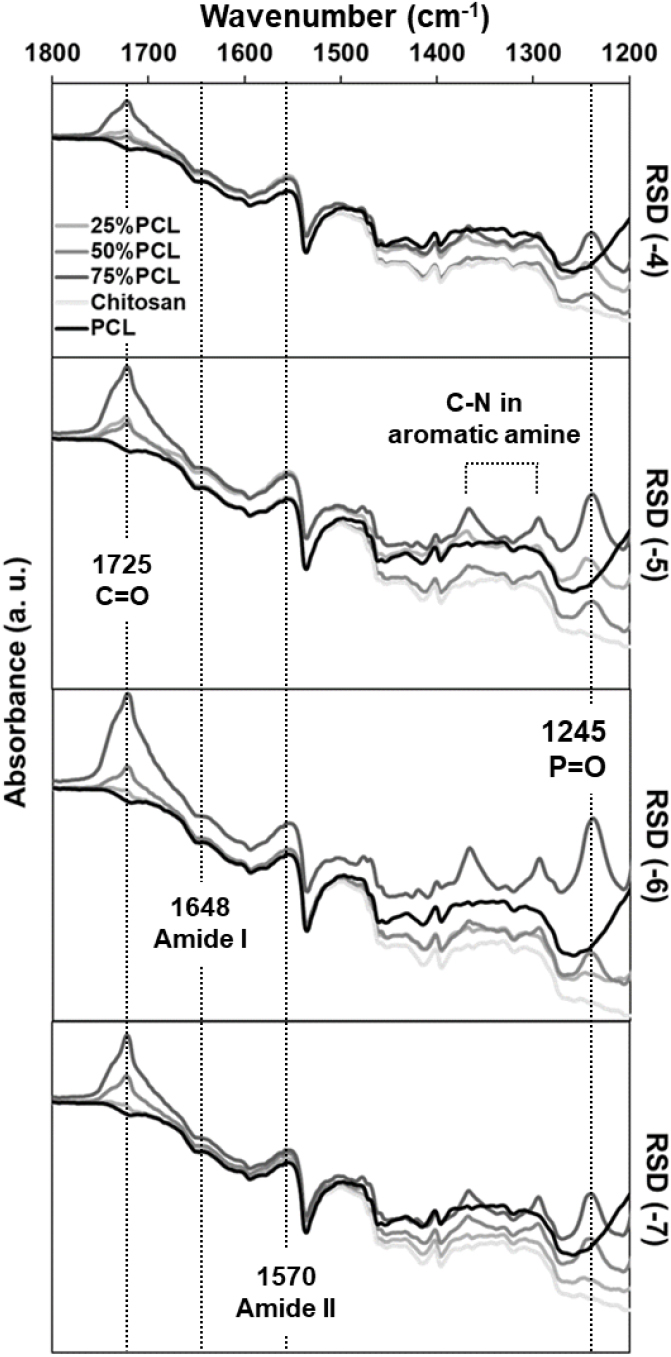
Molecular interactions among PCL, chitosan, and RSD were confirmed using FT-IR spectra as shown in Fig. 4. The characteristic peak of PCL at 1,725 cm-1 represents C=O stretching, while the peaks at 1,648 cm-1 and 1,570 cm-1 correspond to the amide I and II vibrational modes of chitosan. Peaks between 1,300 cm-1 and 1,400 cm-1 represent the C-N vibrations of the pyridine moiety in RSD. These peaks demonstrate that 50 % and 75 % PCL films efficiently incorporate RSD, while 25 % PCL films show less effective RSD loading. Additionally, a peak at 1,245 cm-1 indicates P=O stretching in RSD, confirming effective drug incorporation in the 75 % PCL films.
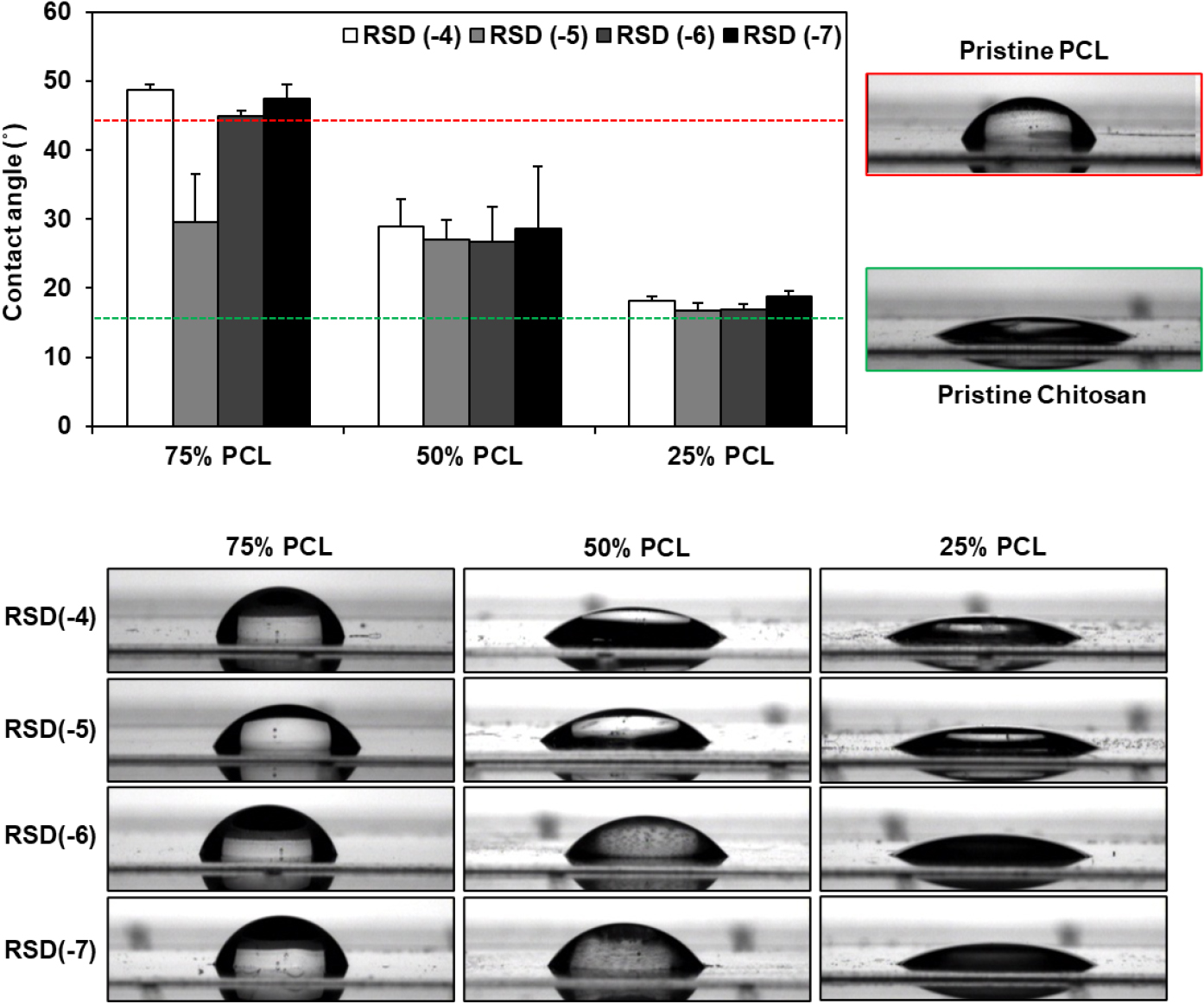
Fig. 5 presents the water contact angle measurements of the composite films. Pristine PCL and chitosan films exhibited contact angles of 44° and 16°, indicating their respective hydrophobic and hydrophilic natures. As expected, the PCL/chitosan composite films showed varying hydrophobicity depending on the PCL content, with lower PCL contents resulting in lower contact angles: 43° for 75 % PCL, 28° for 50 % PCL, and 17° for 25 % PCL. Furthermore, it was confirmed that the contact angle is influenced by the PCL content, regardless of the RSD concentration. In the case of the 75 % PCL film loaded with RSD(-5), the instability of the surface is thought to have led to a decrease in the contact angle. This phenomenon arose because half of the measurements were lower than those of other 75 % PCL films. As shown in Fig. 2, the 75 % PCL film containing RSD(-5) exhibited a mixed surface morphology with partial homogeneity, although chitosan aggregates were observed in certain areas. While a specific concentration of RSD can affect the stability of the composite film, its low concentration indicates that the surface characteristics determining hydrophobicity are primarily governed by the PCL content. Hydrophobicity and hydrophilicity play crucial roles in influencing osteoblast cell proliferation and differentiation. Hydrophilic surfaces tend to promote better cell adhesion, spreading, and proliferation, as they allow proteins in the extracellular matrix to interact more efficiently with the cell membrane. Hydrophilic materials, such as chitosan, enhance osteoblast proliferation by providing a favorable environment for cell attachment and signaling pathways.27) In contrast, hydrophobic surfaces, like those of PCL, are less conducive to cell adhesion but can promote osteoblast differentiation, particularly in the later stages. This is due to their ability to reduce nonspecific protein adsorption, allowing specific signaling molecules to better influence differentiation processes.28,29) Therefore, balancing hydrophobicity and hydrophilicity is essential to optimize both proliferation and differentiation for effective bone regeneration.
3.4. Cellular responses
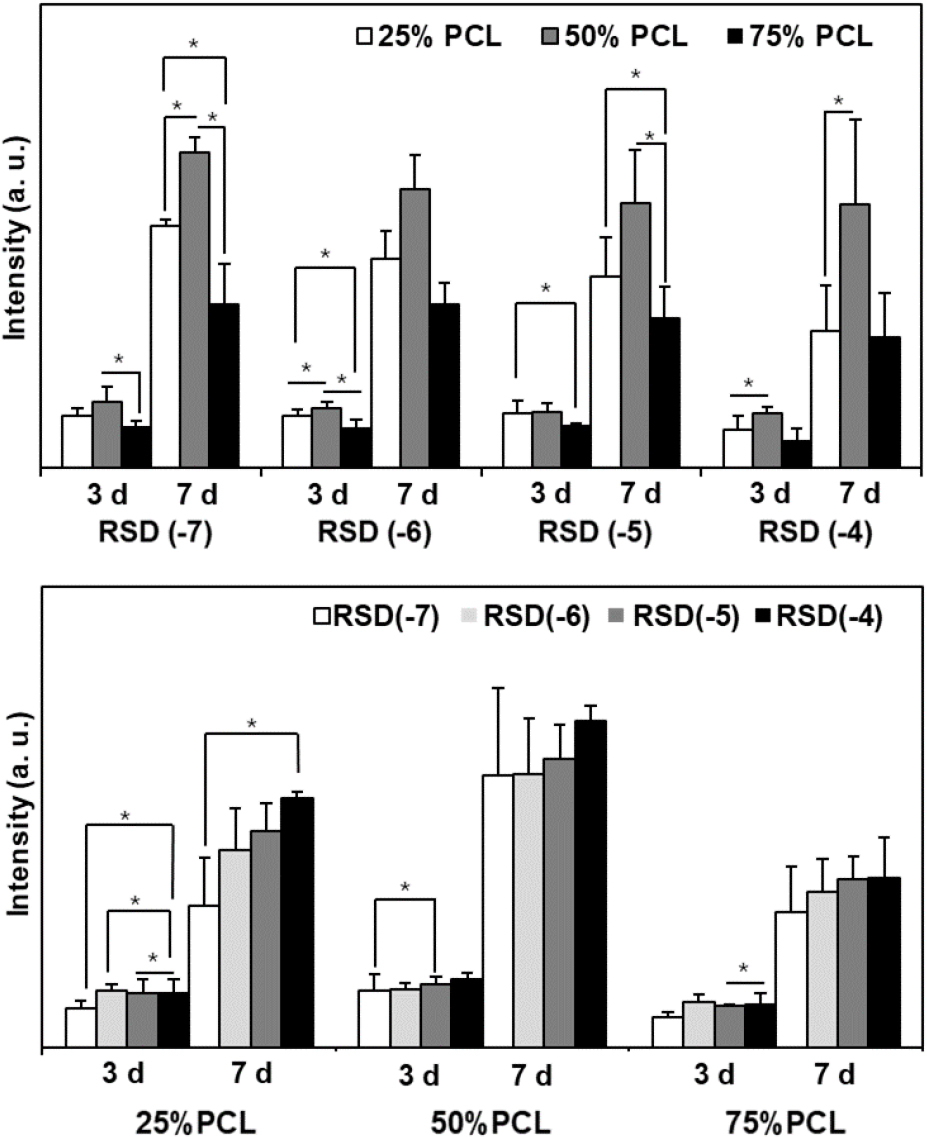
Fig. 6 depicts the proliferation of MG63 cells over 7 days on the composite films. Notably, 50 % PCL exhibited the most significant cell proliferation at both 3 and 7 days. This enhanced proliferation is likely due to moderate hydrophilicity and cell attachment condition. On the other hand, in the case of 75 % PCL, it showed the lowest proliferation regardless of the concentration of RSD. Additionally, RSD(-7) promoted more vigorous proliferation, whereas RSD(-4) inhibited it, suggesting that higher RSD concentrations may negatively impact MG63 proliferation. In the case of 25 % PCL, the differences in proliferation due to varying RSD concentrations were more pronounced. While RSD is known to suppress osteoclast activity and positively influence osteoblast differentiation, its role in reducing proliferation during differentiation may explain these observations.
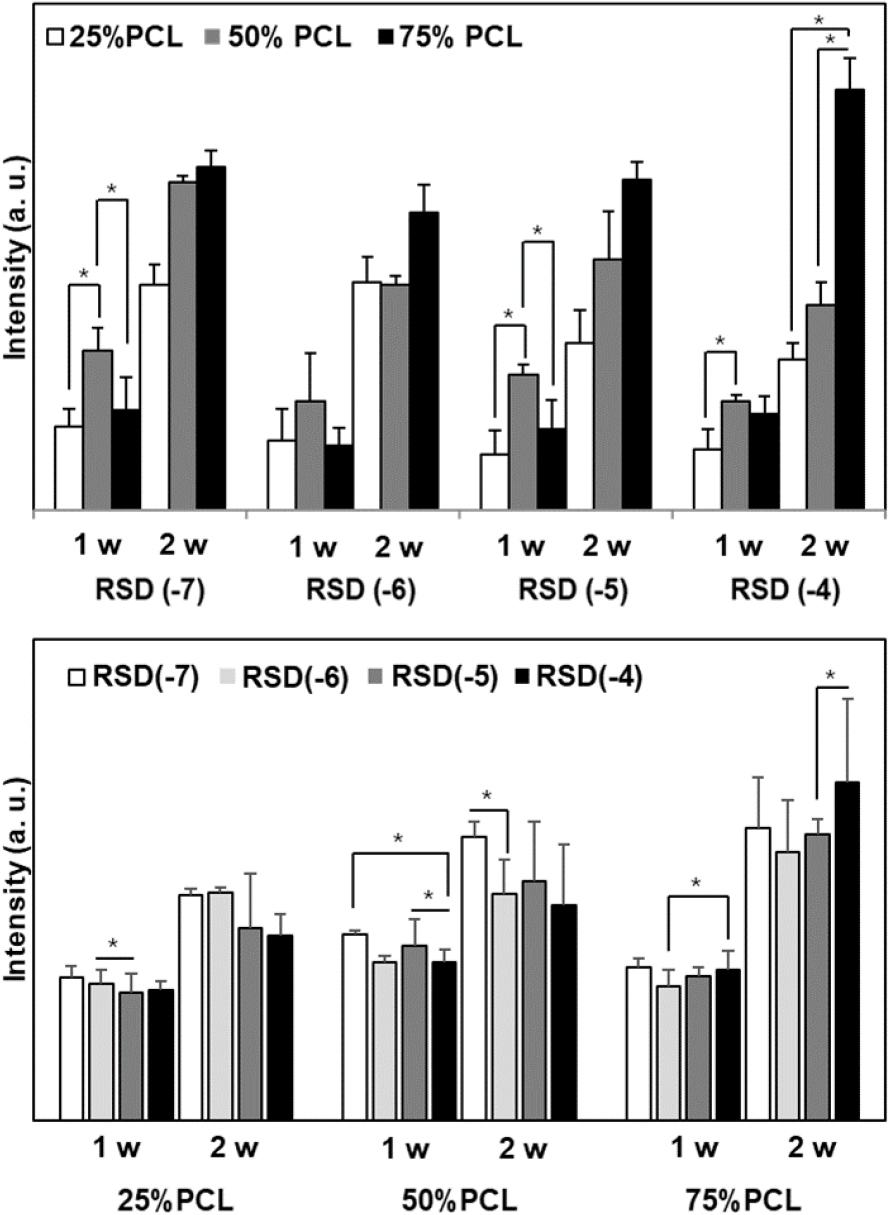
Fig. 7 illustrates ALP activity in MG63 cells cultured on the composite films over 2 weeks. ALP activity, an early marker of osteoblast differentiation, was minimally affected by RSD during the first week. However, 50 % PCL demonstrated higher ALP activity than the 25 % and 75 % PCL films; the 25 % PCL film exhibited the lowest differentiation ability regardless of the RSD concentration. By the second week, 75 % PCL films, particularly those with RSD(-4), showed significantly increased ALP activity. This suggests that rapid cell proliferation during the first week in the 50 % PCL films led to higher ALP activity, while the slower release of RSD in the 75 % PCL films stimulated cell differentiation. These results highlight the importance of the PCL/chitosan ratio to collaborate with RSD release and hydrophobicity influencing cellular responses. For instance, RSD enhances the expression of osteogenic markers like Runx2, Osterix, and bone sialoprotein, and promotes osteoblast activity through connexin43, a protein involved in gap junctions that facilitate osteoblast communication.30,31,32) Furthermore, RSD increases osteoblastic differentiation by stimulating the activity of key transcription factors involved in osteogenesis, such as Dlx5, and boosts osteoblast-specific gene expression.33) These effects suggest that RSD not only prevents bone loss but also supports bone formation, which may offer therapeutic advantages in enhancing bone repair and regeneration.
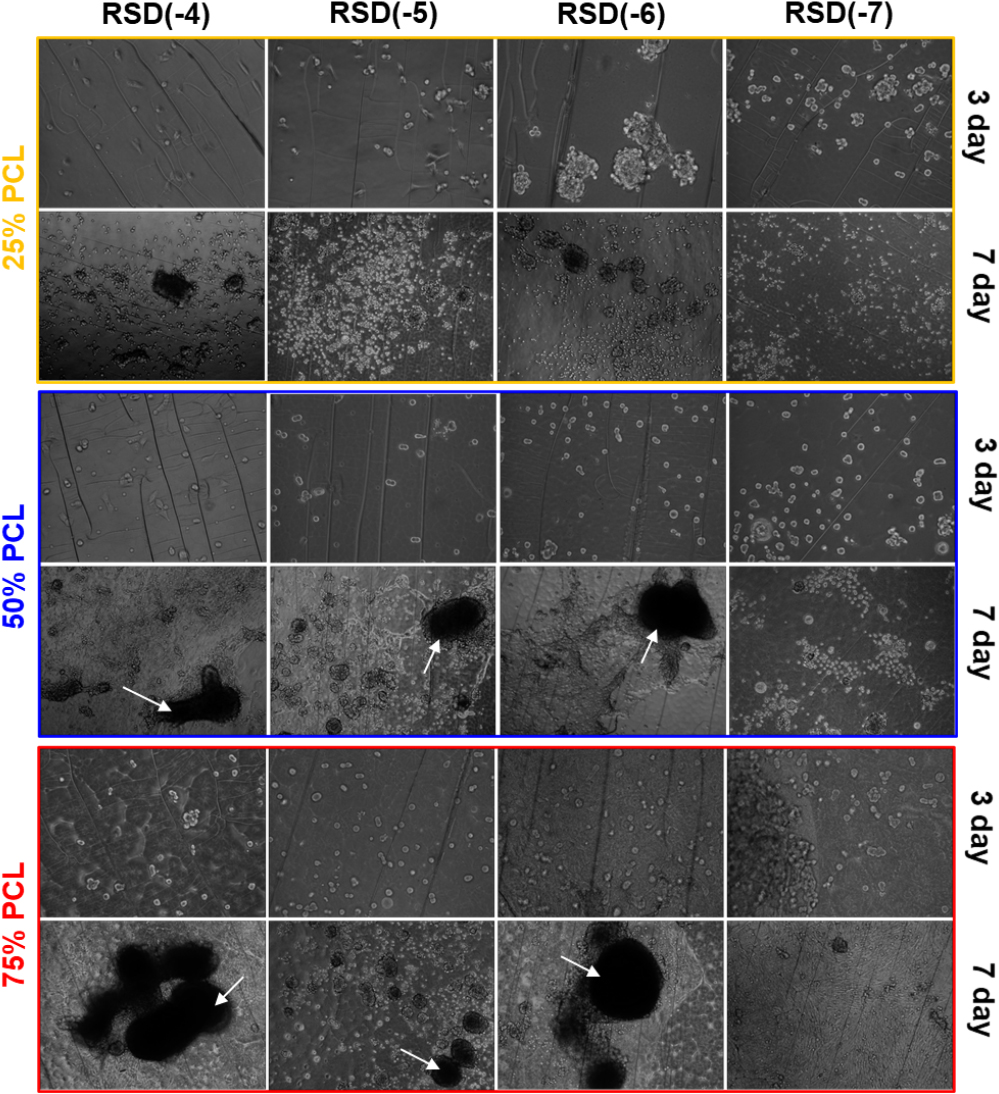
Fig. 8 shows optical microscopy images of MG63 cells after 7 days. In the 25 % PCL films, cells displayed relatively poor adhesion, proliferation, and spreading, likely due to inadequate cell adhesion condition and the suboptimal mechanical properties of the films, despite the known biocompatibility of chitosan. In the 50 % PCL films, cells exhibited extensive spreading and formed bone-nodule-like structures (see arrow) in the RSD(-6) and RSD(-4) films. In contrast, the RSD(-7) films showed limited morphological changes and assembly. The 75 % PCL films, particularly those with RSD(-4), supported the most vigorous formation of multilayer cell assemblies after 7 days. Interestingly, all RSD(-7) films lacked nodule formation but exhibited relatively robust proliferation, as seen in Fig. 6, indicating that RSD may have a stronger effect on osteoblast differentiation than proliferation.
4. Conclusion
The PCL/chitosan composite films demonstrated significant potential as drug delivery systems for bone repair, with controlled miscibility, morphology, and hydrophilicity influenced by the PCL content and drying conditions. The blend of PCL and chitosan effectively modulated the drug release profiles and supported osteoblast cellular responses. RSD incorporation at optimized concentration, i.e., RSD(-4), was shown to enhance osteoblast differentiation, as evidenced by increased ALP activity and osteogenic marker expression, particularly in films with higher PCL content. The balance of hydrophilicity and hydrophobicity in the films influenced both osteoblast proliferation and differentiation, with the 50 % PCL films supporting the best cell proliferation and the 75 % PCL films facilitating enhanced osteoblast differentiation. The results highlight the critical role of PCL/chitosan ratio in tailoring both drug release and cellular responses, demonstrating that RSD not only prevents bone loss but also promotes bone formation. These findings suggest that PCL/chitosan composite films, particularly those optimized for RSD release, could serve as effective drug delivery system for bone tissue regeneration, offering a promising strategy for future therapeutics.