1. Introduction
2. Experimental
2.1. Reagents and materials
2.2. Polymerization
2.3. Experiment equipment
3. Results and Discussion
3.1. Fabrication and analysis of functional silicone hydrogel lenses
3.2. Fabrication and analysis of functional silicone hydrogel lenses containing graphene
4. Conclusion
1. Introduction
Hydrogel lenses are increasingly being used for vision correction and cosmetic effects.1,2) However, unlike glasses, it can result in complications that can cause vision problems, such as corneal opacity, dry eye syndrome, and keratitis. Recently, copolymers developed by many studies are being used in various medical fields.3) They are also used as a material for functional hydrogel contact lenses to compensate for various problems that may occur as a hydrogel lens material. For most hydrogel lenses, higher water content means higher oxygen permeability and better fit. Thus, lenses with high water content and lenses with high oxygen permeability are being continuously studied.4) However, when the water content of the contact lens is high, the refractive index is lowered and the durability of the lens is reduced.5,6) Since the durability of the lens itself is improved when the tensile strength is increased, it is possible to keep the lens from breaking easily. Therefore, physical properties such as tensile strength, which is related to the durability of hydrogel contact lenses, are also important, and studies on materials that increase tensile strength without reducing water content are being actively conducted.7) For several years, an attempt has been made to put graphene, which is called the new material of the future, into practical use in various fields; however, no breakthrough has been made. Graphene is a carbon allotrope that has a two-dimensional plane consisting of carbon atoms. It is attracting attention due to its rapid electron movement, high thermal conductivity and high strength resulting from its two-dimensional structure. In addition, it is currently being used in nano size to polarize the properties of graphene.8,9) The properties of materials may vary depending on the size and shape of the nanoparticles. These nanotechnologies control materials at the atomic or molecular level, which is the smallest unit of material, in order to maximize physical properties by utilizing nanoparticles of 100 nm or less in various fields. Furthermore, graphene can be processed in the form of a ‘nanoplate’, a plate with a thickness of less than 100 nm, by stacking graphene in several layers, and research using graphene nanoplates is being actively conducted.10,11) Graphene oxide obtained by oxidation of graphite is known to have unique functions, such as excellent water solubility, amphiphilicity, and surface functionalization. In particular, it has hydrophilic functional groups (-OH, -COOH) and excellent solubility.12,13) Thus, research is being conducted in a wider range of applications than the typical pure graphene. Based on a silicone hydrogel material using a silicone monomer, including a hydroxyl group (-OH) and a hydrophilic monomer, the silicone monomer, decamethyl cyclopentasiloxane, was used as an additive to improve the fit, and then the physical properties of the lens were evaluated after copolymerization. In addition, graphene was used as an additive to further improve the functionality of the silicone hydrogel material, and after each lens was fabricated using graphene oxide nanoparticles and graphene nanoplates, the physical properties were evaluated to investigate its usability as a functional hydrogel lens material.
2. Experimental
2.1. Reagents and materials
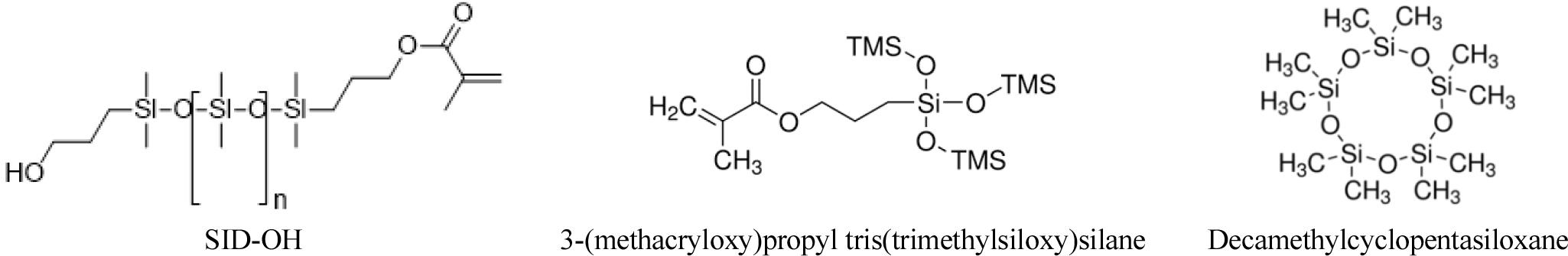
SID-OH, a silicone monomer containing a hydroxyl group in PDMS, and 3-(methacryloxy)propyl tris(trimethylsiloxy)silane (TRISS) were used. Copolymerization was conducted by adding N,N-dimethylacetamide (DMA), ethylene glycol dimethacrylate (EGDMA) as a crosslinking agent, and azobisisobutyronitrile (AIBN) as a thermal initiator. Decamethylcyclopentasiloxane (DCS), graphene oxide nanoparticle (GON), and graphene nanoplate (GNP) were used as additives, polyvinylpyrrolidone (PVP) was used as a dispersant, and the chemical structures of silicon and graphene additives are shown in Figs. 1 and 2, respectively. In addition, all reagents were purchased and used from Sigma-Aldrich (USA).
2.2. Polymerization
SID-OH, TRISS, and DMA were mixed as a basic combination, and DCS was added in a ratio of 1 % to 20 %, respectively. In addition, after forming a new basic combination by selecting an appropriate ratio of DCS, GON and GNP was added by using a ratio of 0.05 % to 0.2 %, respectively. According to the addition of nanoparticles and nanoplates, PVP as a dispersant was added respectively, followed by stirring using an ultrasonic disperser for 1 h, and thermal polymerization at 120 °C for 2 h using a casting molding method. The samples fabricated with the basic combination was named Ref, and the samples were named D1, D2, D3, and D4 depending on the amount of DCS added. According to the addition of graphene to improve functionality, D2 was newly named D_Ref, and the samples were named D_GO1, D_GO2, and D_GO3 according to the amount of GON added, and D_GP1, D_GP2, and D_GP3 according to the amount of GNP added, respectively. Table 1 shows the mixing ratios of the fabricated lenses.
Table 1.
Percent compositions of samples (unit: wt%).
2.3. Experiment equipment
Each of the prepared lenses was hydrated in 0.9 % sodium chloride physiological saline for 24 h, and physical properties were evaluated. The refractive index, water content and oxygen permeability were measured based on ISO 18369-4:2006, the refractive index was measured using an ABBE Refractormeter (ATAGO NAR IT, Japan), and the water content was measured using a gravimetric method. The tensile strength was measured using Universal Testing Machine (AGS-X 20N, Japan) and evaluated by measuring the highest value at which the lens breaks at the time a force of 0 to 2.00 kgf is applied to both sides of the sample. In addition, polymerization stability was evaluated through elution test (absorbance, potassium permanganate reduction test, pH test), and antibacterial properties against Escherichia coli and Staphylococcus aureus was evaluated using 3M PetrifilmTM, a dry film medium. All experimental values were measured at least 5 times to increase accuracy and expressed as average values. In addition, the significance of the experimental values were confirmed by one-way ANOVA, and when p < 0.05 in the analysis, it was judged to be statistically significant.
3. Results and Discussion
3.1. Fabrication and analysis of functional silicone hydrogel lenses
DCS was used as an additive to fabricate functional silicone hydrogel lenses, and oxygen permeability, water content, and tensile strength of the lenses fabricated with the addition ratios of 1 % to 20 % were measured. Based on this result, the ratio of functional silicone hydrogel material was optimized.
3.1.1. Oxygen permeability
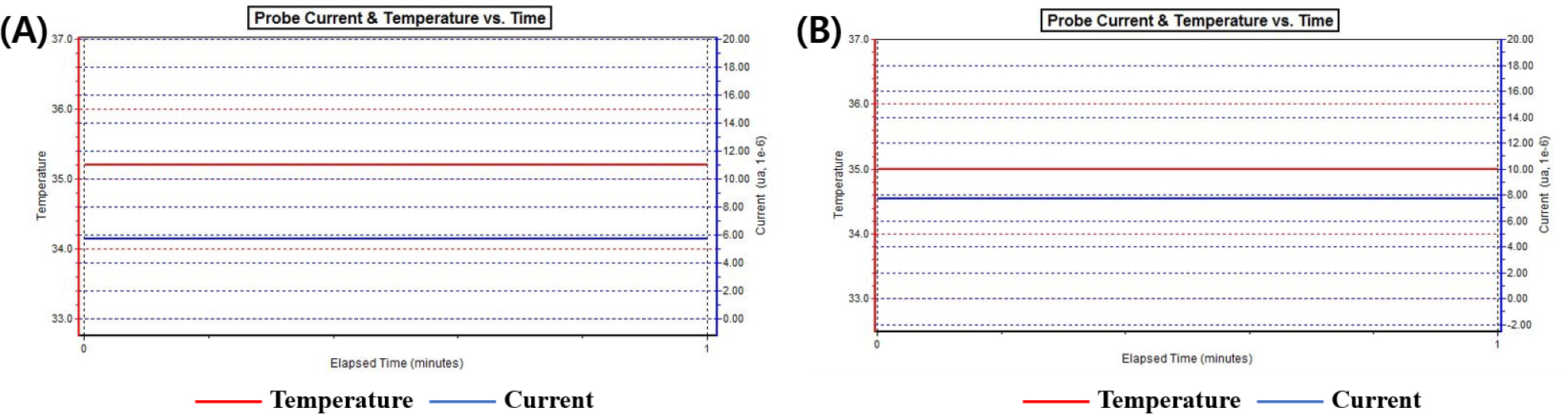
As a result of Dk to evaluate the oxygen permeability of the fabricated lens, the Dk of Ref without using additives was 20.06 × 10-11 (cm2/sec) (mlO2/ml × mmHg), and the samples with DCS added at a ratio of 1 % to 20 % in Ref showed a distribution of 21.85 to 24.84 × 10-11 (cm2/sec) (mlO2/ml × mmHg). In general, the oxygen permeability of the lens to which 2-hydroxyethyl methacrylate (HEMA) is added is about 10 × 10-11 (cm2/sec) (mlO2/ml × mmHg), thereby showing low oxygen permeability.14,15) In this study, oxygen permeability was improved due to the influence of various silicone monomers and DCS, a silicone monomer used as an additive. In the current measurement graph of each lens, the higher the current value, the higher the oxygen permeability. The Dk of the Ref and D4 groups are compared in Fig. 3 as a representative example.
3.1.2. Water content
Water content refers to the proportion of moisture contained in the lens itself. It is an important indicator among the physical properties of hydrogel lenses. As a result of the measurement, the water content of Ref was 61.34 %, and the distribution was 61.54 % to 62.60 % depending on the amount of DCS added. All of the fabricated lenses showed a water content of 50 % or more. In addition, the water content gradually increased according to the amount of additives.
3.1.3. Tensile strength
The tensile strength of the lens was measured in order to confirm its durability. As a result of the measurement, the tensile strength of Ref was 0.93 MPa, and it was found to be 0.86~0.62 MPa depending on the ratio of additives. As the water content of the lens increased, the durability decreased. In the case of tensile strength, the result was inversely proportional to the tendency of the water content.
3.2. Fabrication and analysis of functional silicone hydrogel lenses containing graphene
In the case of silicone hydrogel lens fabricated by adding DCS at each ratio, the water content and oxygen permeability were increased, while the tensile strength was very low, thereby indicating that the durability was very weak. Therefore, a new basic combination was formed by selecting SID-OH, DMA, 3-TRISS, and DCS of 5 % (D_Ref). In addition, graphene oxide nanoparticles and graphene nanoplates were added at a ratio of 0.05 % to 0.2 %, respectively, to the basic combination. After that, the physical properties of the fabricated lenses were evaluated.
3.2.1. Surface analysis
D_GO3 and D_GP3 containing 0.2 % of GON and GNP, respectively, were selected, and the surface of the hydrogel lens according to the type of additive was measured by SEM to confirm the surface distribution shape. As a result of the measurement, it was confirmed that D_GO3 with GON had a size of 30 nm to 60 nm, and D_GP3 with GNP had a size of 40 nm to 100 nm. The surface analysis images by SEM are shown in Fig. 4.
3.2.2. Physical properties
3.2.2.1. Water content and refractive index
The water content and refractive index of the hydrogel lens, including graphene nanoparticles and nanoplates, respectively, were measured. The water content of D_Ref without graphene nanoparticles was 62.36 %, and the refractive index was 1.3830. In addition, the combination including GON showed a water content of 62.57 % to 63.69 % and a refractive index of 1.3839 to 1.3901 depending on the amount of additives. The combination including GNP showed a water content of 62.30 % to 62.35 % and a refractive index of 1.3843 to 1.3887 depending on the amount of additives. Regardless of the type of graphene, the refractive index slightly increased as the amount of additive increased, and water content slightly increased for GON, but did not change for GNP. It is judged that the hydrophilic functional groups (-OH, -COOH) of GON affected the water content of the lens.
3.2.2.2. Oxygen permeability
As a result of Dk to evaluate oxygen permeability of the fabricated lenses, Dk of D_Ref was 23.45 × 10-11 (cm2/sec) (mlO2/ml × mmHg), and Dk of the combination with GON was 23.69 to 25.72 × 10-11 (cm2/sec) (mlO2/ml × mmHg) depending on the amount of GON added. On the other hand, there was no significant change in GNP according to the amount added. Therefore, it was confirmed that the addition of GON affects the oxygen permeability of hydrogel lenses, and it is thought that this was influenced by the water content closely related to the oxygen permeability.
3.2.2.3. Tensile strength
The tensile strength of D_Ref was 0.77 MPa, and the tensile strength of the combination containing GON and GNP increased as the added amount of graphene increased, from 0.82 MPa to 1.66 MPa and 0.91 MPa to 1.81 MPa, respectively. Regardless of the type of graphene, the tensile strength gradually increased as the amount added increased, thereby indicating excellent durability. This was found to have an effect on the tensile strength of the lens due to the high mechanical properties of graphene itself, especially in the sample containing GNP. In addition, the physical properties of contact lenses including graphene are shown in Table 2.
Table 2.
Physical properties of contact lens samples containing graphene.
| D_Ref | D_GO1 | D_GO2 | D_GO3 | D_GP1 | D_GP2 | D_GP3 | |
|
Refractive index* | 1.3830 ± 0.0001 | 1.3839 ± 0.0002 | 1.3872 ± 0.0001 | 1.3901 ± 0.0000 | 1.3843 ± 0.0003 | 1.3858 ± 0.0002 | 1.3887 ± 0.0003 |
|
Water content (%)* | 62.36 ± 0.0913 | 62.57 ± 0.1165 | 62.92 ± 0.1037 | 63.39 ± 0.1052 | 62.30 ± 0.1116 | 62.30 ± 0.1006 | 62.35 ± 0.1076 |
|
Oxygen permeability (Dk)* | 23.45 ± 0.1229 | 23.69 ± 0.1158 | 24.56 ± 0.1461 | 25.72 ± 0.1539 | 23.56 ± 0.1823 | 23.25 ± 0.1402 | 23.31 ± 0.1704 |
|
Tensile strength (MPa)* | 0.77 ± 0.097 | 0.82 ± 0.029 | 0.90 ± 0.056 | 1.66 ± 0.056 | 0.91 ± 0.073 | 1.13 ± 0.029 | 1.81 ± 0.024 |
3.2.2.4. Stability test
Extractable test was performed in order to confirm the stability characteristics following the addition of GON and GNP. For the extractable test, potassium permanganate reduction test and pH test were performed, and the amount of nano materials added was selected to be 0.2 % for the experiment.
3.2.2.4.1. Potassium permanganate reduction test
The extractable value of the control group consisting of distilled water was 18.5, that of the graphene-free group (D_Ref) was 17.86, that of GON (D_GO3) was 17.29, and that of GNP (D_GP3) was 17.75. In all groups, the difference in the extractable value was 2.0 or less, thereby indicating that it was stable to organic matter regardless of the presence or absence of nano materials.
3.2.2.4.2. pH test
The results of the pH test are shown in Table 3, and in all samples, the difference in pH between the control group and the experimental group was measured to be less than 1.5. Therefore, it was confirmed that the extractable value was very small overall, and the stability was excellent regardless of the presence or absence of nano materials.
Table 3.
pH test results of samples.
| Control | Experimental | |
| D_Ref* | 5.26 ± 0.002 | 5.34 ± 0.003 |
| D_GO3* | 5.00 ± 0.002 | 5.00 ± 0.002 |
| D_GP3* | 5.05 ± 0.003 | 5.02 ± 0.002 |
3.2.2.5. Antibacterial test
In order to evaluate the antibacterial activity, the antibacterial properties against microorganisms were confirmed by using the dry film method. In order to check the antibacterial activity of the lenses with GON and GNP added, 1 ml of the sample solution was injected into the dried medium, and then incubated for about 24 h in a thermostat at 35 ± 1 °C to check antibacterial properties against Staphylococcus aureus and Escherichia coli. As a result of the measurement, it was confirmed that in D_Ref without graphene, a rather large number of microorganisms were found in both Staphylococcus aureus and Escherichia coli media. On the other hand, the groups to which GON and GNP were added showed excellent antibacterial properties as the graphene content increased. It was also confirmed that the group to which GNP was added was particularly excellent in antibacterial properties. Figs. 5 and 6 shows the measurement results of the antibacterial properties.
4. Conclusion
In this study, decamethylcyclopentasiloxane (DCS) was used to improve the oxygen permeability of the lens, and two types of graphene, graphene oxide nanoparticle (GON) and graphene nanoplate (GNP), were used to further improve the physical properties. As a result of measuring the physical properties of the hydrogel lenses according to the use of additives, it was found that the water content and oxygen permeability of the silicone hydrogel material added with DCS gradually increased according to the amount added, but the tensile strength gradually decreased. Furthermore, the addition of GON and GNP improved the durability of the lens by gradually increasing the tensile strength of the contact lens, and showed excellent stability and antibacterial properties. Combinations with GON showed that oxygen permeability and refractive index were gradually improved according to the amount added. In the case of GNP, tensile strength and antibacterial properties were superior to those of GON without changing most of the basic physical properties. Therefore, the silicone hydrogel lens material containing DCS, GON, and GNP has high oxygen permeability and antibacterial properties. At the same time, it can complement the durability of the high water content lens material. Thus, it is determined that they can be used in various ways as a material for ophthalmic functional hydrogel lenses.