1. Introduction
Ophthalmic contact lenses are directly attached to the cornea, which can be found at the front of the eye, for the purpose of vision correction, eye disease treatment, and beauty, and have a direct effect on the eyes.1) In the 1930s, ophthalmic lenses made of PMMA plastic were used instead of glass, while soft lenses made of hydrogel were developed in the 1970s and began to be widely distributed with FDA approval. Since then, RGP lenses with high oxygen transmissibility have been developed, and since the 1990s, silicone has been used as a material for soft contact lenses that can increase oxygen transmissibility. 2) Research on ophthalmic lenses with high oxygen transmissibility using silicon and development of various high-functional materials, as well as high oxygen transmissibility, are currently in progress.3) Contact lenses have different physical properties depending on the mixing ratio of additives during polymerization, and are affected by polymerization method, temperature, and time.4-6) In addition, the lenses with improved functionality are being developed by using various additives. High-functional lenses include lenses with high water content, wettability, and refractive index at the same time.7) Among various additives, a new high-functional lens manufactured by applying nanomaterials with different photo-physical properties and antimicrobial properties according to particle size and color has recently been studied.8-10) Holmium (III) oxide and europium(III) oxide are oxygen compounds with rare-earth elements, and generally have excellent chemical, thermal, and mechanical stability. These are used as colorants, special catalysts, phosphors, and laser materials as phosphors.11-13) Aluminium oxide has a strong affinity for hydroxide ions, and it is used in industrial environments, pharmaceuticals, food additives, and cosmetics.14) Materials for manufacturing lenses with improved water content, wettability, and refractive index are required by an appropriate combination of a silicone monomer that is hydrophobic but has a very high oxygen transmissibility, a hydrophilic monomer, and functional additives.15) In order to optimize the mixing ratio of silicone and hydrophilic monomer, this study evaluated the physical properties of each sample prepared after mixing at various ratios, and tried to shorten the polymerization time via photo polymerization method. The lenses were fabricated by using various nanomaterials as additives to a silicone polymer fabricated with an optimal mixing ratio and shortened polymerization time. In addition, by adding polyvinylpyrrolidone (PVP) in a 1 % ratio, the physical properties according to the degree of dispersion were examined, and the compatibility with hydrophobic silicone and the possibility of application as a functional lens material were confirmed.
2. Experiment
2.1. Reagents and Materials
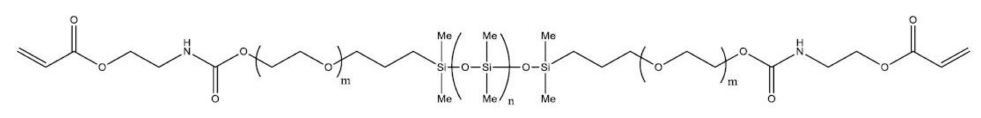
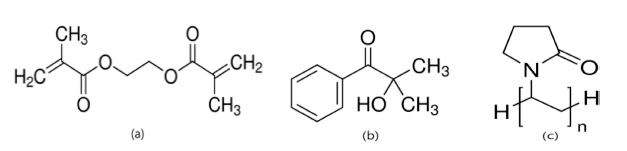
In this experiment, for the basic combination, SIU, a silicone monomer, as the main material of the silicone lens, N, N-dimethylacrylamide (DMA) as a hydrophilic monomer, ethyleneglycol dimethacrylate (EGDMA) as a crosslinking agent, and 2-hydroxy-2-methylpropiphenone (2H2M) as a photoinitiator were used. As additives, holmium(Ⅲ) oxide, europium(Ⅲ) oxide, aluminium oxide, and polyvinylpyrrolidone (PVP) were used. For all reagents, except for the synthesized silicone monomer, products of SIGMA-ADRICH were used without purification. The structural formula of the silicone monomer is shown in Fig. 1, and the structural formulas of ethyleneglycol dimethacrylate (EGDMA) as the crosslinking agent, 2- hydroxy-2-methylpropiphenone (2H2M) as the initiator, and PVP as the dispersant are shown in Fig. 2.
2.2. Photo Polymerization of Silicon Polymer
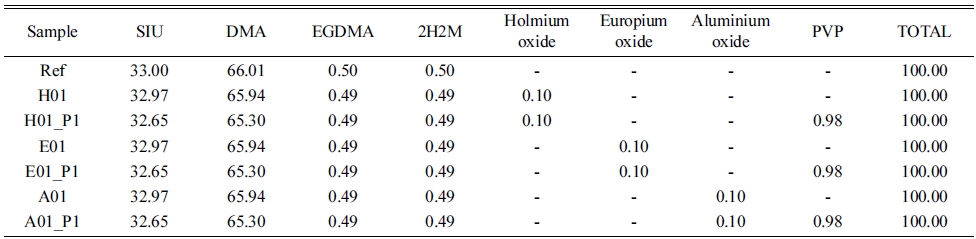
In order to prepare a silicone polymer to be used as the basic mixing condition, a silicone monomer and N, Ndimethylacrylamide (DMA) were used as the main ingredients and mixed with ethyleneglycol dimethacrylate (EGDMA) and 2-hydroxy-2-methylpropiphenone (2H2M) for photo polymerization. The optimum mixing ratio and polymerization time were confirmed by varying the mixing ratio and polymerization time of silicone and N, Ndimethylacrylamide (DMA). Silicone and N, N-dimethylacrylamide (DMA) were mixed in the ratio of 1:1, 1:2, and 2:1, and stirred for about 30 minutes. The stirred samples were named SY1, SY2, and SY3, respectively. In addition, the samples were polymerized for 40 and 70 seconds at a wavelength of 365 nm by using a UV curing system (FJ100, Phoseon Technology), respectively. According to the radiation time, they were named SY1-40, SY1-70, SY2-40, SY2-70, SY3-40, and SY3-70, respectively. The prepared polymer was hydrated in 0.9 % physiological saline for 1 day, and then the physical properties were measured and compared. The mixing ratio of each sample used in the experiment is presented in Table 1.
2.3. Fabrication of Lenses Containing Nano-Oxide and PVP
The optimum ratio obtained through the experimental results in 2.2 was determined as Ref. Nanoparticles of holmium(III) oxide, europium(III) oxide, and aluminium oxide were added at 0.1 % ratio to the Ref sample and dispersed for about 30 minutes with an ultrasonic disperser (Branson 2510), and then stirred for about 30 minutes at room temperature to fabricate each lens. They were named H01, E01, and A01, respectively. Additionally, the samples made by adding PVP at 1 % ratio to the samples, where each of the three types of nano-oxide was added and stirred at room temperature for about 40 minutes, were named H01_P1, E01_P1, and A01_P1, respectively. All of the mixed samples were polymerized for 40 seconds at a wavelength of 365 nm by using a photo polymerization device under the same conditions as Ref, and then subjected to hydration in 0.9 % physiological saline for 24 hours in order to measure and compare the physical properties of the lenses. The mixing ratios of the samples used in each experiment are presented in Table 2.
2.4. Measuring Device and Methods
The optical transmittance of the prepared polymer was measured in the range of 280 to 780 nm by using a spectral transmittance meter (Agilent, Cary 60 UV-Vis), and average values were presented. The refractive index was measured with an ABBE Refractometer (ATAGO DR-A1, Japan), and water content was calculated by measuring the hydrated and dried weight of the lens via gravimetric method according to ISO 18396-4:2006. In addition, values measured by using a contact angle instrument (Kruss GMBH, DSA30) were used to evaluate wettability. The oxygen transmissibility of fabricated lenses was measured by using the polarographic method, and lens surface observation and roughness analysis were conducted by using AFM (XE-100, Park Systems).
3. Results and Discussion
3.1. Physical Properties of Polymerized Silicone Lenses
3.1.1. Physical properties according to mixing ratio and radiation time
3.1.1.1. Optical transmittance
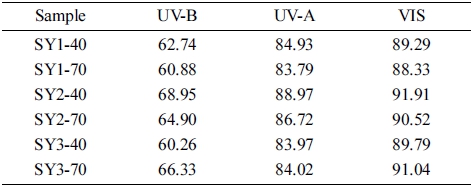
The results of optical transmittance to evaluate the optical properties of the fabricated lenses are shown in Table 3. The results showed generally high transmittance and confirmed that transparent lenses were fabricated. However, there was a difference in the spectral transmittance value according to the mixing ratio, and for the same ratio, there was a slight difference depending on the radiation time. Therefore, through the spectral transmittance measurement, it is determined that the SY2-40 has high transparency, and the spectral transmittance of each lens are presented in Fig. 3.
3.1.1.2. Refractive index and water content
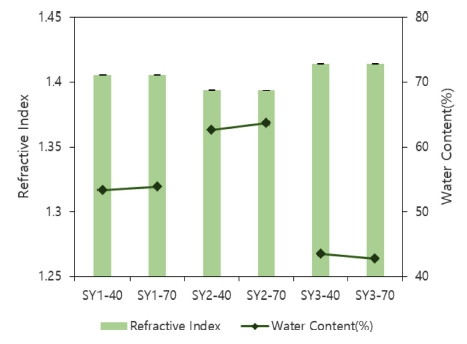
As a result of the refractive index of the lenses according to various mixing ratios of silicone and hydrophilic monomers and radiation time, it was 1.4057 for SY1-40, 1.4058 for SY1-70, 1.3937 for SY2-40, 1.3937 for SY2- 70, 1.4142 for SY3-40, and 1.4141 for SY3-70. The results showed that as the ratio of the hydrophobic silicone monomer increased, the refractive index tended to increase. However, water content was 53.29 % for SY1- 40, 53.91 % for SY1-70, 62.64 % for SY2-40, 63.69 % for SY2-70, 43.45 % for SY3-40, and 42.72 % for SY3- 70. As the proportion of the hydrophilic monomer, N, Ndimethylacrylamide (DMA), increased, the water content tended to increase, which is inversely proportional to the result of the refractive index. The results of the refractive index and water content of each sample are compared and presented in Fig. 4.
3.2. Properties of Lenses containing Nano-Oxide and PVP
3.2.1. Holmium(Ⅲ) oxide
3.2.1.1. Surface analysis
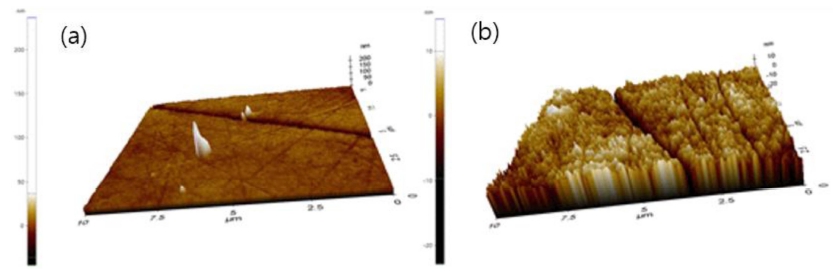
Using SY2-40 with excellent physical and optical properties as Ref, three types of oxide and PVP at 1% ratio were added respectively, and the physical properties of fabricated lenses were measured in order to compare and analyze the physical properties according to oxide and the addition of PVP. As a result of AFM analysis of the lenses to which nano-oxide and PVP were added for surface analysis of the fabricated lenses, it was found that the nanoparticles were evenly dispersed in the surface of the fabricated polymer. The AFM images of the lenses are presented in Fig. 5.
3.2.1.2. Optical transmittance
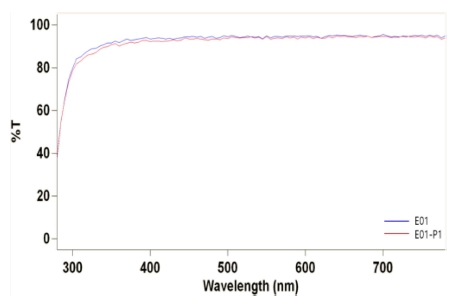
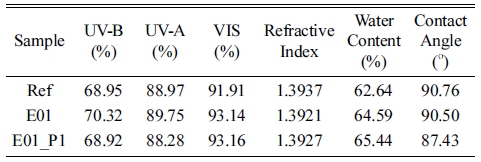
As a result of the spectral transmittance to analysis the optical properties of the lenses, the transmittance of H01 added with holmium(III) oxide was 68.28 %, 87.50 %, and 92.04 % in the UV-B, UV-A, and VIS regions, respectively. The transmittance of H01_P1 added with 1 % PVP was 68.89 %, 88.82 %, and 93.79 %, respectively. The spectral transmittances are presented in Fig. 6.
3.2.1.3. Physical Properties of polymers with holmium (Ⅲ) oxide
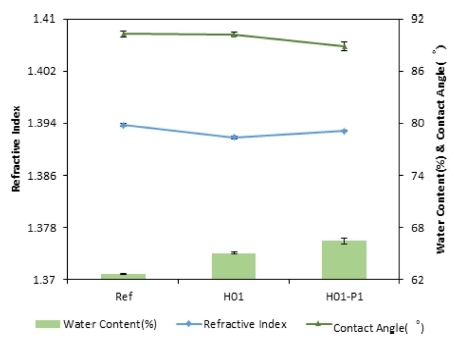
As a result of analyzing the physical properties of fabricated lenses, a slight change in the average refractive index appeared for each lens. Compared to the average refractive index of the Ref sample, the refractive index of the sample to which nano-oxide was added was slightly decreased, and when PVP was added at the same time, the refractive index was slightly increased. And also, the average water content of the fabricated lenses was 62.64 % for Ref, 65.03 % for H01, and 66.43 % for H01_P1, which tended to increase gradually with the addition of PVP. In addition, as a result of measuring the contact angle to evaluate the wettability, it was 90.29° for Ref, 90.26° for H01 with holmium(III) oxide, and 88.89° for H01_P1 with 1 % PVP, thereby indicating that wettability slightly improves as the contact angle decreases overall. As a result of measuring oxygen transmissibility of the fabricated lenses, the average oxygen permeability (Dk) was 35.4262×10-9 (cm/sec) (mlO2/ml×mmHg) for Ref, 34.5433×10-9 (cm/sec) (mlO2/ml×mmHg) for H01, and 29.5971×10-9 (cm/sec) (mlO2/ml×mmHg) for H01-P1, thereby indicating that it decreases with the addition of an additive and a dispersant. As a result, the increase in water content and decrease in contact angle were attributable to the influence of PVP, which is a hydrophilic group, and the results are presented in Fig. 7. The reason why the average oxygen permeability tended to decrease was judged to be attributable to the reduced silicon content by the addition of additives rather than the influence of the hydrophilic group, PVP.
3.2.1.4. Surface analysis by AFM
As a result of measuring AFM to analyze the surface roughness of the samples to which holmium(III) oxide and PVP were added, the surface roughness (Ra) was 6.131 nm for Ref, 3.928 nm for H01 with holmium(III) oxide, and 2.797 nm for H01_P1 with PVP. It was found that the surface roughness of the lenses decreased as an additive and a dispersant were added. The consequent slight increase in the refractive index of H01_P1 compared to that of H01 is judged as a result of an increase in optical density as nanoparticles are more evenly dispersed on the lens surface due to the action of PVP, a dispersant. In the case of the H01 sample, the degree of the surface smoothness was increased due to the addition of holmium oxide, thereby making the contact angle smaller than that of the Ref, so that wettability was increased. At the same time, water content increased due to the increased wettability. The AFM images for the measurement of surface roughness of fabricated lenses are presented in Fig. 8.
3.2.2. Europium(Ⅲ) oxide
The optical transmittance of the lens to which europium(III) oxide was added, the transmittance was almost similar to that of the Ref, and the result is shown in Fig. 9. Additionally, as a result of measuring the refractive index, water content, and contact angle, the refractive index slightly decreased when nanoparticles were added compared to the Ref, and slightly increased compared to the E01 lens when PVP was added as a dispersant. The water content of the E01 and E01_P1 lenses gradually increased compared to the Ref, and as the water content increased, the contact angle decreased, thereby resulting in an increase in wettability. The optical and physical properties of each fabricated lens are summarized in Table 4.
3.2.3. Aluminium oxide
3.2.3.1. Surface analysis
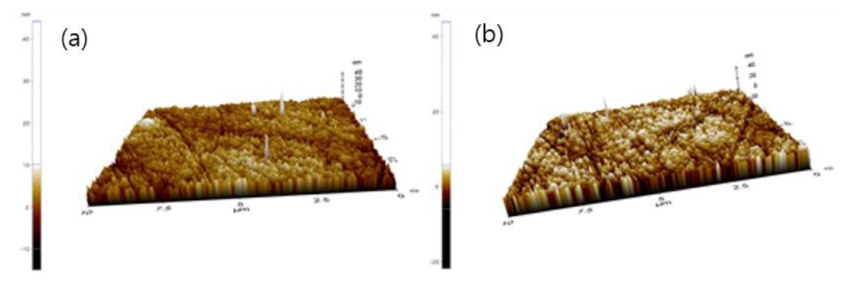
As a result of AFM analysis of the lens to which aluminium oxide and PVP were added, it was confirmed that the nanoparticles were evenly dispersed on the surface of the fabricated lens. The AFM result images for each lens are presented in Fig. 10.
3.2.3.2. Optical transmittance
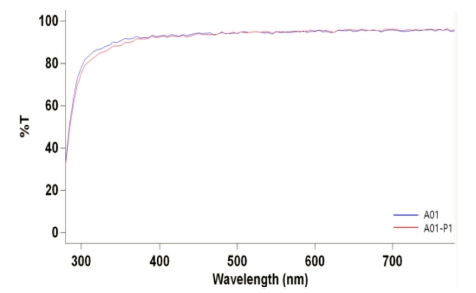
As a result of measuring the optical transmittance of each fabricated lens, the transmittance of the A01 group with aluminium oxide added based on the Ref in UV-B, UV-A, and VIS regions was 67.80 %, 87.78 %, and 92.95 %, respectively. The transmittance of the A01-P1 group with PVP added based on the A01 was found to be 66.27 %, 86.94 %, and 93.02 %, respectively. As a result, when aluminium oxide was added, the transmittance of the A01 lens was higher than that of the Ref lens in the VIS region. It is judged that aluminium oxide was more evenly dispersed on the lens surface by adding a dispersant, PVP, thereby affecting the transmission of light. The spectral transmittance results of A01 and A01_P1 are presented in Fig. 11.
3.2.3.3. Physical properties of lenses with aluminium oxide
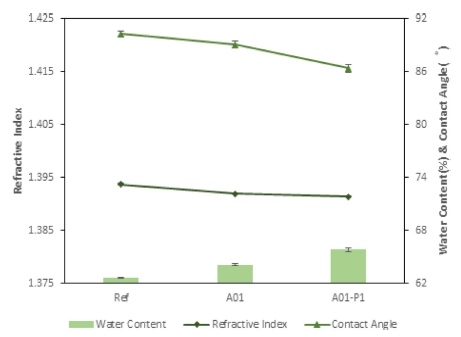
As a result of the refractive index of each fabricated lenses, it was 1.3937 for Ref, 1.3919 for A01, and 1.3914 for A01-P1, slightly decreasing but showing little change. On the other hand, as a result of measuring the water content of the fabricated lenses, it was 62.64 % for Ref, 64.11 % for A01, and 65.82 % for A01_P1, thereby showing a gradually increasing trend. Nano-oxide and PVP seemed to have influenced the basic physical properties of the lenses. In addition, as a result of measuring the contact angle, it was 90.29° for Ref, 89.04° for A01, and 86.37° for A01_P1, and wettability is increased. As a result of measuring oxygen transmissibility, oxygen permeability (Dk) of the lenses was 35.4262 × 10-9 (cm/sec) (mlO2/mL×mmHg) for Ref, 30.8041 × 10-9 (cm/sec) (mlO2/mL×mmHg) for A01, and 29.2623 × 10-9 (cm/sec) (mlO2/mL×mmHg) for A01_P1. It was found that the water content in the polymer increased due to the slightly reduced refractive index of the fabricated lenses, thereby increasing the water content, and it was judged that the wettability improved due to the increased number of water molecules. In addition, the decrease in average oxygen permeability (Dk) compared to the Dk of Ref seemed to be attributable to the effect of reducing the proportion of silicon in the polymer due to the addition of aluminium oxide and PVP. The changes in water content and wettability for each sample are presented in Fig. 12.
3.2.3.4. Surface analysis by AFM
As a result of the AFM analysis of the fabricated lenses, surface roughness (Ra) was 6.131 nm for Ref, 2.433 nm for A01, and 2.387 nm for A01-P1. When aluminium oxide and PVP were added, the surface roughness was reduced than that of Ref. Consequently, it is judged that the roughness of the lens surface decreased, the wettability of the lens increased, and the water content increased. It was thought that the refractive index did not change as there was no change in the surface roughness of the lens in the A01 and A01_P1 groups. The AFM images for each lens are presented in Fig. 13.
4. Conclusion
This study analyzed the optimal mixing ratio with N, N-dimethylacrylamide (DMA), a hydrophilic monomer, by using the silicone monomer, and appropriate polymerization conditions to increase the efficiency of photo polymerization. For the fabrication of each sample, 0.1 % of various nano-oxides were added to the basic combination Ref, and PVP was added in a ratio of 0 to 1 %. Furthermore, the physical properties of lenses, including various types of nano-oxides and a dispersant, were compared and analyzed. Optical transmittance, refractive index, water content, contact angle, oxygen transmissibility, and AFM were measured in order to analyze the physical properties and surface of each fabricated lens. As a result, different physical properties were shown according to the mixing ratio of the silicone monomer and N, Ndimethylacrylamide (DMA), and transmittance and water content were found to be excellent at a mixing ratio of 1:2. As a result of the experiment by varying the irradiation time for photo polymerization, it was found that there was little difference in the physical properties between the light irradiation results for 40 seconds and 70 seconds, so the polymerization time was determined to be 40 seconds. When holmium(III) oxide and europium (III) oxide were added based on the Ref sample, the physical properties of the lens showed a similar tendency overall, and aluminium oxide showed a slightly different tendency from the previous two oxides. It is judged that various nano-oxides and PVP, a dispersant, can be used as high-functional silicone hydrogel lens materials.