1. Introduction
2. Experimental
2.1. Materials
2.2. Plasma polymerization and oxidation
2.3. Wetting measurement
2.4. Morphology studies
3. Results and Discussion
3.1. Water contact angle and wetting
3.2. Morphology
4. Conclusions
1. Introduction
Polyester is one of the popular synthetic textiles due to its light and soft properties, strong durability and low price. In particular, it is widely used as a material for linings, shirts, blouses, etc. because it does not easily wrinkle and has good wrinkle recovery. However, polyester clothing is inconvenient to wear due to its high hydrophobicity, which accumulates static electricity in dry air, and has low hygroscopicity and permeability to sweat in hot and humid air. In addition, polyester fibers are difficult to dye, coat and clean due to their hydrophobicity.1,2) For the comfort and performance of polyester as a garment, a hydrophilic treatment is essential. The alkaline soda process has been used for a long time as a hydrophilic treatment of synthetic textiles, but it consumes a lot of energy in the final drying step and has many environmental problems because it uses a lot of water and chemicals.3,4) Various studies have been conducted on textile surface treatment using plasma as an eco-friendly method.5,6) Theoretically, plasma treatment is useful for effective fiber hydrophilization because it affects only the uppermost surface of the fiber without changing the internal structure or bulk properties of the fiber.6-9) During plasma treatment, the surface of the fiber undergoes chemical changes, changes in the surface structure, and changes in the physical properties of the surface. Plasma treatment creates new chemical species by disrupting chemical bonds on the fiber surface by ions, electrons, photons, and radicals. In particular, it is possible to make the fibers hydrophilic by making hydrophilic functional groups such as -OH and -COOH on the surface.5,6,10)
Most of the early studies on the plasma surface treatment of textiles were conducted at low pressure, but low-pressure equipment is expensive and has a size limit, so atmospheric pressure plasma processes that do not require vacuum equipment are continuously attracting attention. Dielectric barrier discharge (DBD) among atmospheric pressure plasmas is widely used in textile surface treatment studies because of their easy discharge formation at atmospheric pressure, large area availability, and low equipment cost.11-17) Leroux et al. demonstrated that wettability of PET textile was improved by air DBD treatment because the oxidation treatment increased the hydroxy and carboxy groups on the fiber surface.13,17)
However, the hydrophilicity rapidly decreased within a few hours after plasma treatment and maintained at a constant value after decreasing for about 3 days.8,18) In particular, the hydrophilicity decreased rapidly after washing treatment.17) The decrease in hydrophilicity was assumed to be loss of hydrophilic functional groups by surface restructuring or removal. Therefore, it is difficult to maintain hydrophilicity for a long time only by forming a hydrophilic functional group on the surface by plasma treatment. Long-term hydrophilicity is not required to improve dyeability or adhesion, but long-term hydrophilicity maintenance is required to use polyester for clothing.
Silicon oxide is very hydrophilic.19-21) Therefore, the polyester textile can have semi-permanently hydrophilic by being coated with the silicon oxide. Generally, high-quality silicon oxide is coated by thermal CVD or plasma-enhanced CVD using silane gas in a vacuum at high temperature.22-25) However, the silicon oxide should be coated at a low temperature of about 200 degrees or less because of the low softening point of polyester. Also, high-quality silicon oxide is brittle, so it is not suitable for coating textiles. Kim et al. reported a hydrophilic surface by forming a SiO2-like thin layer only on the upper surface of the film through oxygen plasma treatment after synthesizing a siloxane polymer film using APDBD (atmospheric pressure dielectric barrier discharge) at room temperature.26) Since the siloxane polymer is soft and the uppermost SiO2-like layer is very thin (several nanometers), it is thought that it can be easily maintained on the fiber surface without breaking.
In this study, using APDBD, a siloxane polymer film was coated on a polyester fabric and then plasma oxidized to form a SiO2-like layer to make the outermost surface hydrophilic. The main purpose of this study was to evaluate the wettability of polyester textile coated with plasma oxidation-treated siloxane polymer and the change in wettability following repeated washing treatment.
2. Experimental
2.1. Materials
The polyester woven fabric was made from the yarn of Huvis (Korea). The warp yarn was 75 denier with 36 filaments and the weft yarn was 100 denier with 36 filaments. Ends/inch, picks/inch and weight of the fabric were taken as 70, 120, 120 g/m2 respectively. The discharge gases used were 99.999 % pure helium and oxygen. Hexamethylcyclotrisiloxane (HMCTSO, Sigma-Aldrich, ≥98 %) as a siloxane monomer was used to coat siloxane polymer film. Although HMCTSO is solid at room temperature, it has a sufficiently high vapor pressure to achieve rapid growth of the film. In addition, HMCTSO is non-toxic and non-explosive, making it easy to handle.
2.2. Plasma polymerization and oxidation
The atmospheric pressure DBD system used in this study was the same as that of the previous study.26) The APDBD system consisted of a flat ground electrode and two cylindrical high-voltage electrodes covered with alumina as a dielectric. The diameter, length and thickness of the alumina tube are 15 mm, 150 mm and 1 mm respectively. The reaction gases were introduced by installing an alumina tube with a 1 mm slit at the top between two cylindrical electrodes. The gap between the high voltage electrode and the polyester fabric (100 × 150 mm) on alumina plate covering ground electrode was maintained at 0.5 mm. The fabric was repeatedly passed through the 30 mm discharge zone at a speed of 1 mm/s.
A stable APDBD was formed at a voltage of 4 kV using a 30 kHz AC power supply while supplying helium at a rate of 5 l/min. Siloxane polymers, SiOxCy(-H), were synthesized with or without additional oxygen using 0.05 l/min of helium as the MHCTSO carrier gas. The SiOxCy(-H) thin layer coated on polyester was oxidized for 160 seconds while supplying 5 l/min of helium and 0.1 l/min of oxygen. The details of process parameters of SiOxCy(-H) coating and oxidation is shown in Table 1.
Table 1.
Experimental parameters of SiOxCy(-H) coating and post-oxidation.
2.3. Wetting measurement
Polyester textile wettability was studied by a sessile drop method using a commercial PSM (Plasma Systems and Materials, Korea) NPT-CAM10 system. Since the hydrophilized textile absorbs water droplet quickly through capillary action, the contact angle was measured immediately after dropping the water droplet. The contact angle was measured at least 5 times and averaged. In addition, a strip wicking test was performed to evaluate the capillary effect.27) The strip wicking was tested according to KS K ISO 9073-6:2007 standard.
The polyester fabric (50 × 70 mm) was washed using a solution of 35 ml of liquid detergent in 20 liters of tab water. In this solution, the fabric was folded in half and washed by rubbing 20 times repeatedly, followed by rinsing in running water for 30 seconds. After washing, it was dried naturally at room temperature for 24 hours. After washing and drying the plasma-treated fabric repeatedly, the contact angle was measured to evaluate the change in hydrophilicity according to washing.
2.4. Morphology studies
The surface of polyester fabric was observed with scanning electron microscope (SEM, JEOL: JSM-6390) at 3 kV to study the change in the surface morphology of the fabric after washing. It was evaluated whether the coating layer was peeled off after washing by detecting a silicon peak using energy dispersive spectroscopy (EDS, Oxford: ISIS). EDS peaks were obtained at ×500 magnification by area scan mode.
3. Results and Discussion
In a previous study, we reported the formation of SiO2-like layer by plasma oxidation treatment on the surface of SiOxCy(-H) thin film synthesized by APDBD using HMCTSO as a precursor.26) It was also reported that the increase in the amount of oxygen gas added during the synthesis of the SiOxCy(-H) film increased the film density, increased the silicon content of the synthetic film, and lowered the carbon content.28) In addition, increasing the amount of HMCTSO carrier gas improved the deposition rate of the thin film, but increased the carbon content of the synthesized film due to insufficient decomposition of HMCTSO.28-30) For semi-permanent hydrophilicity (SPH) of polyester fabric, the adhesion is more important than the thickness of the SiOxCy(-H) thin film formed on the fabric surface. And the upper layer of the thin film must be effectively converted into SiO2-like layer by plasma oxidation. Therefore, in this study, the flow rate of HMCTSO carrier gas was selected as 0.05 l/min.
3.1. Water contact angle and wetting
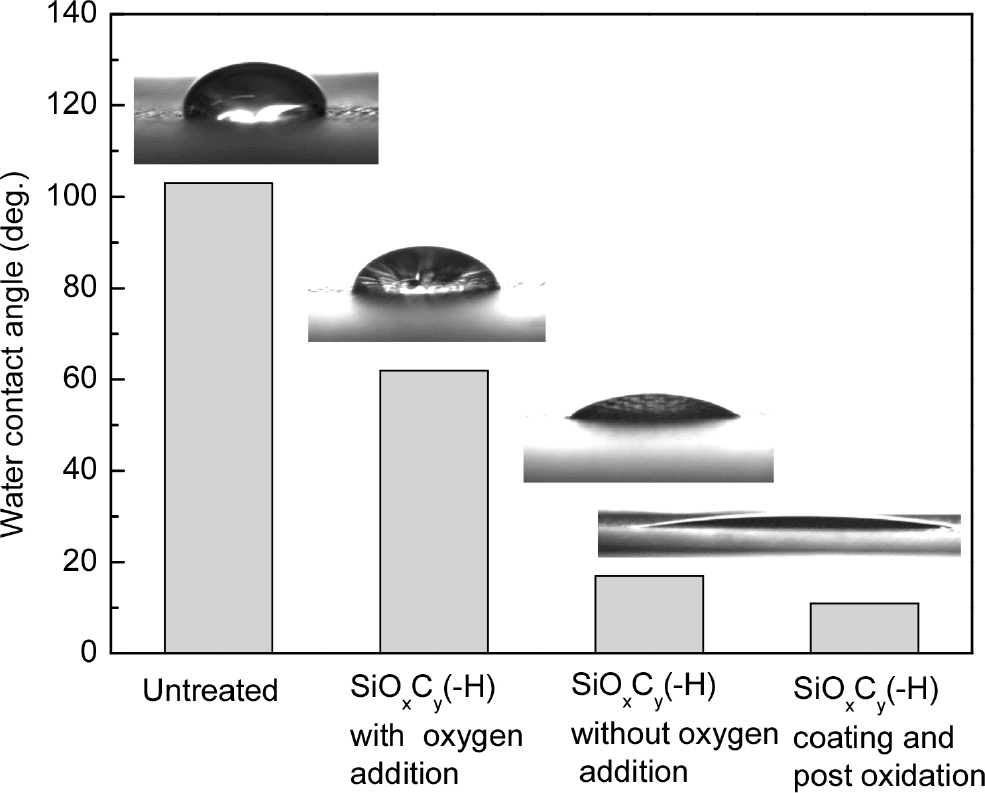
Fig. 1 shows water droplets and the water contact angle (WCA) of untreated, SiOxCy(-H) coated, and coated and plasma oxidized polyester fabrics. The polyester fabric was hydrophobic and the WCA was measured to be greater than 100 degrees. The WCA of SiOxCy(-H) deposited with the addition of oxygen was approximately 60 degrees which was similar to the previous work deposited on Si wafers.26) On the other hand, the polyester fabric coated with SiOxCy(-H) deposited without the addition of oxygen had very good hydrophilicity, so the contact angle could not be measured due to the immediate absorption of water droplets. The WCA of plasma-oxidized polyester fabrics after coating with SiOxCy(-H) films could not also be measured due to instantaneous water drop absorption. The two WCA and water droplets on the right side of Fig. 1 were the results measured after one wash for comparison with other samples.
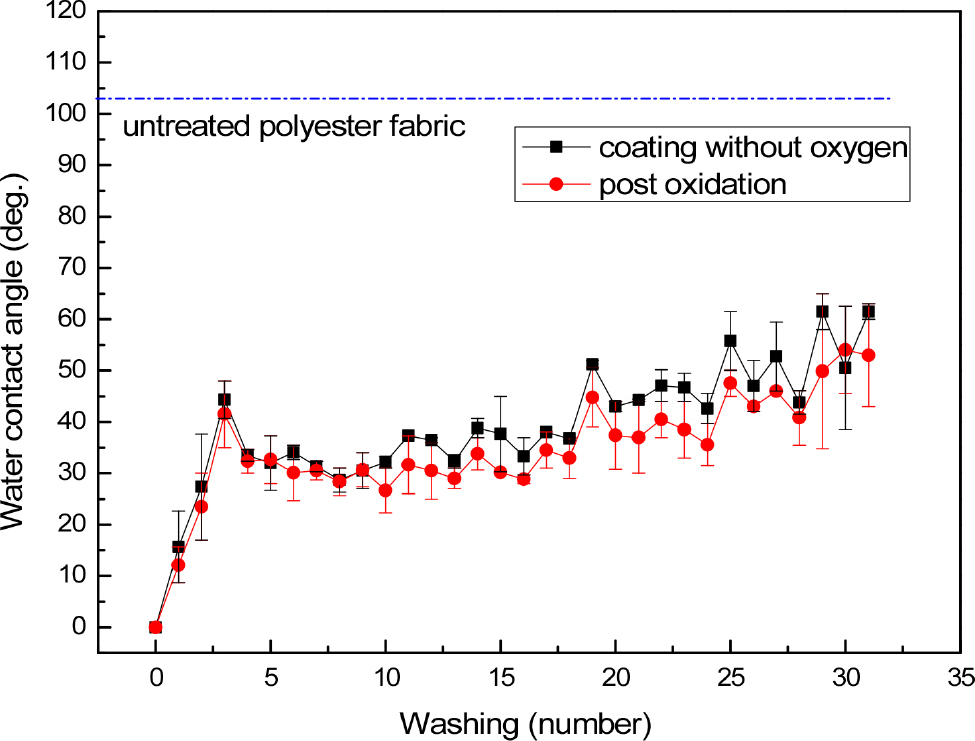
Fig. 2 shows the WCA variation of polyester fabric coated with SiOxCy(-H) film without adding oxygen as a function of the number of washing. Fig. 2 also shows the change in WCA of the plasma-oxidized fabric after SiOxCy(-H) film coating. In the case of the fabric before washing, whether or not post-treatment with oxygen plasma, water droplets were absorbed immediately upon falling on the fabric, so the WCA could not be measured. It can be seen that the SiOxCy(-H) film synthesized without adding oxygen gas exhibits hydrophilicity. In both cases, the WCA increased rapidly during the initial washing, and increased to about 30~40 degrees after washing 3 times as shown in Fig. 2 The WCA was maintained at 30~40 degrees until approximately 20 washes. It can be seen that the WCA of the fabric post-treated with oxygen plasma was about 10 degrees lower than that of the case without post-treatment within the error range, but in both cases, excellent hydrophilicity was maintained even after washing. After washing about 20 times, the WCA gradually increased as the number of washings increases, but both fabrics maintained a contact angle of about 50 degrees after washing 30 times.
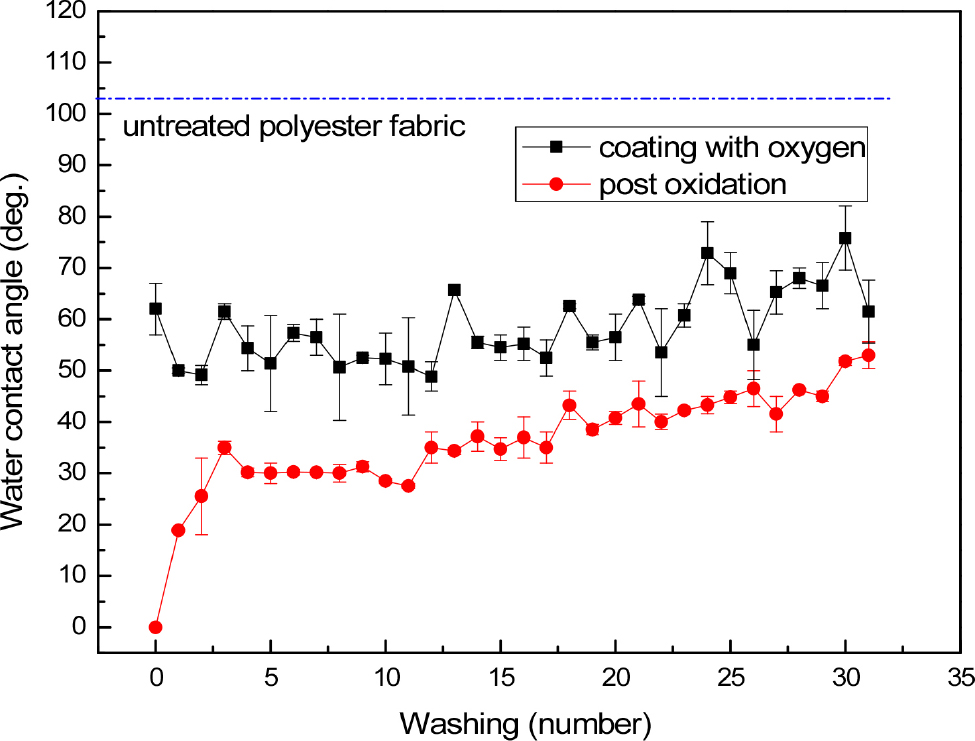
Fig. 3 shows the WCA variation of polyester fabric coated with SiOxCy(-H) film synthesized while supplying 0.04 l/min of oxygen as a function of the number of washing. The WCA of the polyester fabric coated with the SiOxCy(-H) film before washing was about 55~65 degrees. Until about 15 washes, the WCA was between 40 and 60 degrees, and there was no increase in the contact angle with washing. The WCA gradually increased after 15 washes, and increased to about 60~70 degrees after 30 washes.
On the other hand, water droplets falling on the oxygen plasma post-treatment fabric before washing were immediately absorbed, so the WCA could not be measured. As shown in Fig. 3, the WCA of oxygen plasma post-treatment fabric increased to about 20 degrees after one wash and to about 30 degrees after two washes, and was maintained at about 30 degrees until the 13 washes. As the number of washings increased, the WCA showed a tendency to gradually increase, and the contact angle was in the range of 30~40 degrees up to 20 times and 35~45 degrees up to 30 times. When the SiOxCy(-H) films were post-treated with oxygen plasma, a very thin layer of SiO2-like was formed on the surface.26) Therefore, as shown in Figs. 2 and 3, the hydrophilicity of the polyester fabrics whose surface of the coating layer was changed to SiO2-like with excellent hydrophilicity is maintained even after washing more than 30 times with WCA of about 50 degrees. If only hydrophilic functional groups were formed on the surface by oxidation, the hydrophilicity would have been lost in one or two washing.17) In addition, although the properties of the SiOxCy(-H) film were different, the coated fabrics showed similar hydrophilicity even after oxidation treatment because a SiO2-like layer was formed on the film surface by post-oxidation.
Table 2 shows the wicking time to reach 50 mm of polyester fabrics. The wicking time of the fabric before washing was 366 seconds for the untreated fabric, but more than doubled to 733 and 884 seconds for the SiOxCy(-H) coated fabric. The increase in wicking time is thought to be due to a decrease in the capillary effect of the fabric with the coating. The rapid decrease in wicking time to 133 and 142, respectively, after one washing is thought to be due to the improvement of the capillary effect as the coating film blocking among the fiber filaments was broken during the washing process. Of course, the wicking time was shorter than that of untreated fabric due to the hydrophilization effect by coating.
On the other hand, the wicking time of the oxygen plasma-treated fabrics decreased to 140 and 169 seconds, respectively, less than twice that of the untreated fabric. It is believed that there was no problem in the capillary effect of the oxygen plasma-treated fabrics because the coating film blocking among the fiber filaments was removed during the oxygen plasma treatment. In addition, it is considered that the wicking time was fast due to the excellent hydrophilicity of the surface because the surface of the coating became SiO2-like by oxidation. The wicking times were faster for the oxygen plasma-treated fabrics up to 5 washes compared to the coated only fabrics, but similar or slower after 10 washes. However, the oxygen plasma treated fabrics still showed about half the wicking time compared to the untreated fabric.
Table 2.
Wicking times reached to 5 cm for fabrics as a function of washing numbers.
3.2. Morphology
Fig. 4 shows SEM micrographs before and after washing of untreated, coated only, and coating and plasma oxidized fabrics. Untreated fabric [Fig. 4(a-0)] shows a smooth surface as in the picture of general polyester yarn, and it can be seen that the surface [Fig. 4(a-30)] has little change even after washing 30 times. There was nearly no change in the surface before and after washing.
Fig. 4(b-0) is a SiOxCy(-H) film coated without oxygen addition, and particles due to partial peeling of the coating film were observed, but it can be seen that the coating was relatively uniform and smooth. Fig. 4(c-0) shows the surface of the coated yarns treated with oxygen plasma for 120 seconds. Although there were some particles, the surface was smoother than fib. 4(b-0). The surface of the yarns coated with SiOxCy(-H) film synthesized by supplying oxygen was also relatively uniform and smooth as shown in Fig. 4(d-0). Fig. 4(e-0) shows the yarns of the fabric, which was coated at the same condition as shown in Fig. 4(d-0), treated with oxygen plasma for 120 seconds. There were cracks on the surface and some peeling of coating layer as shown in Fig. 4(e-0). It is well known that the localized surface area exposed to filamentary discharge of APDBD was easily damaged.31,32) Filaments in APDBD were generated more easily and stronger with high oxygen concentration.31,32) Therefore, cracking and delamination of the SiOxCy(-H) film is considered to be damage caused by APDBD filaments during oxygen plasma treatment. Depending on the characteristics of the coated SiOxCy(-H) film, the cracking and peeling behavior of the film may be different during the oxygen plasma treatment. When the film [Fig. 4(d-0)] was synthesized with the addition of oxygen, the carbon content was lower than that of the film [Fig. 4(b-0)] synthesized without oxygen gas.28) In this case, cracks and peeling of the film occurred during oxygen plasma treatment.
Fig. 4(b-5) and Fig. 4(c-5) show the fabric surfaces after washing 5 times, where the rapid increase in WCA according to washing was stable as shown in Fig. 2. In the case of Fig. 4 (b-5), it can be seen that several cracks occurred in the coating layer. However, Fig. 4(c-5) shows no significant difference from before washing. This trend is similar in the photos after washing 20 and 30 times, and it is seen that a lot of peeling of the film occurred in Fig. 4(b-20) and Fig. 4(b-30). On the other hand, a few cracks and delamination can be seen in Fig. 4(c-20) and Fig. 4(c-30). During the washing process, the fabric was folded in half and the surfaces of the fabric were rubbed against each other 20 times. It is believed that parts of the coating film were damaged during the rubbing, resulting in cracks and peeling. The damage of the coating film was large within the initial 5 washes and the damage thereafter was relatively small, indicating that the WCA was similar or slightly increased as shown in Fig. 2.
The change in surface state due to washing was more pronounced in Fig. 4(d) and Fig. 4(e) where oxygen was added during the SiOxCy(-H) film synthesis. When oxygen plasma treatment was not performed, there was no significant difference between the surface of the yarns [Fig. 4(d-5)] washed 5 times and the surface of the yarns [Fig. 4(d-0)] before washing. Therefore, as shown in Fig. 3, the change in WCA after washing was insignificant. On the other hand, the slight increase in the contact angle after 15 washes, as shown in Fig. 3, is due to the increased damage to the coating layer during repeated washing. Fig. 4(d-20) and Fig. 4(d-30) show multiple cracks and peeling on the surface of the yarns. In the case of oxygen plasma treatment, the surface of the yarns [Fig. 4(e-5)] washed 5 times showed a relatively smooth compared to the yarns before washing [Fig. 4(e-0)]. This means that the cracked coating peeled off easily during the first few washes. As a result, as shown in Fig. 3, the WCA was initially increased with washing and stabilized at about 30 degrees before washing 5 times. The gradual increase in the WCA after 15 washes was also due to the increased damage to the coating layer during repeated washing as shown in Fig. 4(e-15) and Fig. 4(e-30).

Fig. 4
SEM micrographs before and after washing of (a) untreated, (b) coated without adding oxygen only, (c) coated without adding oxygen and plasma oxidized, (d) coated with adding oxygen only, (e) coated with adding oxygen and plasma oxidized polyester fabrics. The digits in parentheses is the number of washes.
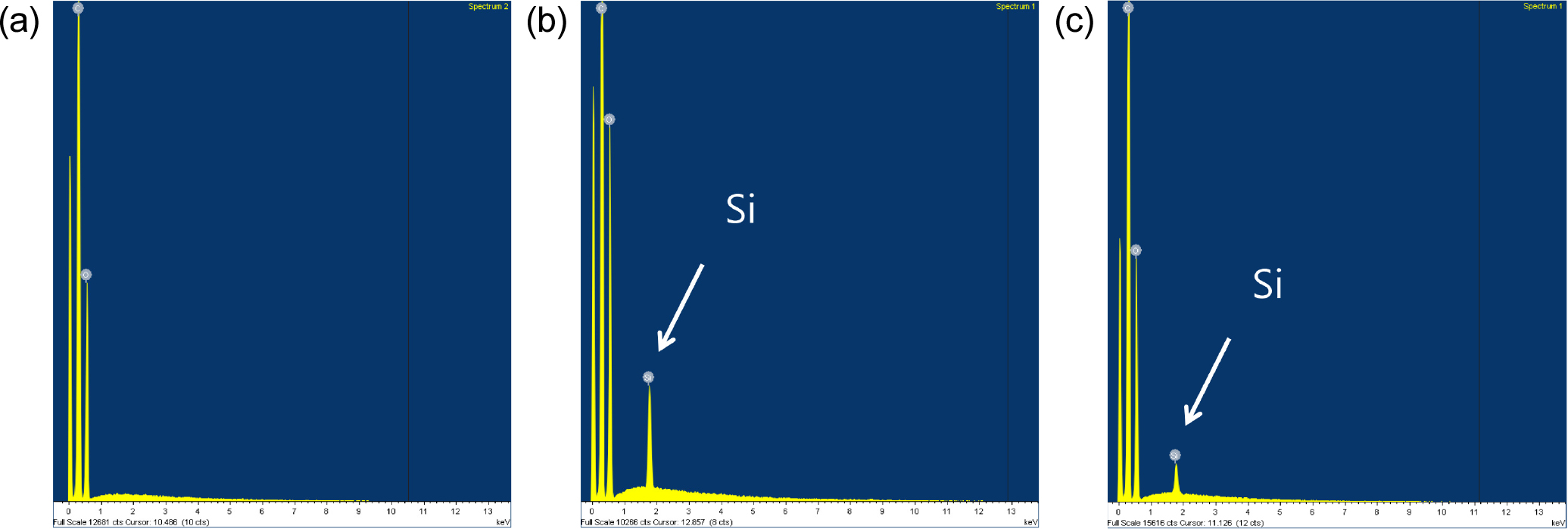
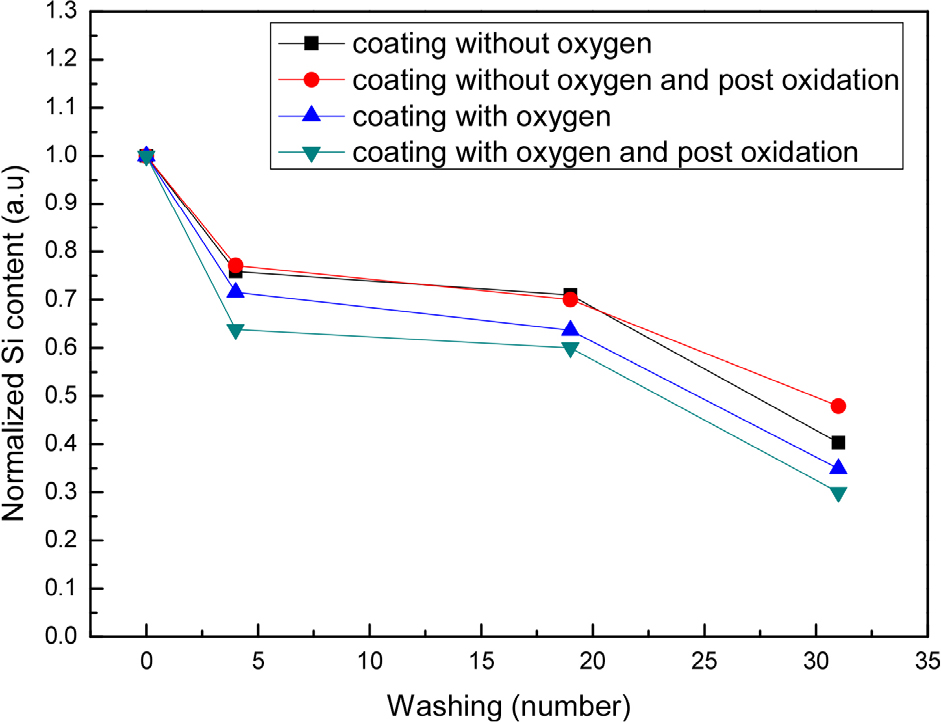
Figs. 5 and 6 shows the results of energy dispersive spectrums of the fabrics shown in Fig. 4. The Si peak is clearly observed all fabrics coated with the SiOxCy(-H) film. It can be seen that the Si content decreased as the number of washings increased in all fabric samples. The decrease in the Si content of the fabrics explains well that the coating layer was peeled off during repeated washing, as shown in Fig. 4. After washing 30 times, the Si content was reduced to less than half compared to the initial stage, but the hydrophilicity of the fabric treated with oxygen plasma was maintained as shown in Fig. 3.
4. Conclusions
We demonstrated that hydrophobic polyester fabrics can be made semi-permanently hydrophilic (SPH) by a combination of coating and oxidation using atmospheric pressure dielectric barrier discharge (APDBD). First, a SiOxCy(-H) thin film was coated on a polyester fabric using APDBD. Then, the upper part of the SiOxCy(-H) coating was treated by APDBD oxygen plasma to form a SiO2-like thin layer to make the fabric semi-permanently hydrophilic. The SPH polyester fabric was so hydrophilic that the water contact angle (WCA) could not be measured due to rapid wetting. The wicking time of the SPH fabric was also shortened to about half that of untreated fabric. Although the hydrophilicity of the SPH fabric decreased as washing was repeated, hydrophilicity was maintained at a WCA of about 50 degrees even after 30 cycles. The decrease in hydrophilicity of the SPH fabric due to repeated washing was due to partial peeling of the SiOxCy(-H) thin film coated on polyester yarns, but the coating film was sufficient to maintain hydrophilicity even after washing 30 times. Therefore, the continuous process of SiOxCy(-H) coating and post-oxidation treatment using APDBD is expected to be applied as an eco-friendly semi-permanent hydrophilic treatment method for polyester fabrics.