1. Introduction
Electrochemical water splitting contributes to producing H2 with high purity, which has aroused intense research interest in recent years.1-3) The reaction mechanisms for hydrogen evolution reaction in acidic solutions are summarized as two steps: (i) The electrochemical hydrogen adsorption (M+ H++ e- → M-H*, Volmer reaction); (ii) The electrochemical desorption (M-H* + H+ + e-/M+ H2, Heyrovsky reaction) or chemical desorption (2M-H* → 2M + H2, Tafel reaction, where M refers to an electrocatalyst species, and H* implies a hydrogen atom chemically adsorbed on an active site of the cathode surface).4)
Monoclinic W18O49 has high carrier density, cost effectiveness and long lasting stability in electrochemical atmosphere.5) However, the use of W18O49 as an efficient electrocatalyst for HER has not been explored because of its large overpotential and few active sites confirmed from density functional theoretical calculations. Atomic doping can modify the electronic structure of the catalyst, promote hydrogen adsorption and evolution, and ultimately improve the intrinsic activity of the catalyst.6) Mo has similar electronic structure to that of W, which could modify the electronic structure of W18O49 and improve electrocatalytic performance. After Mo doping in Zhong’s report,7) the calculated HER overpotential on all oxygen sites is reduced to about 0.23 eV compared with those of the pristine W18O49. Zhao et al.8) fabricated a series of Mo incorporated W18O49 NFs by a solvothermal and templatefree method, in which an overpotential of only 462 mV is required to achieve current density of 50 mA-2 in comparison with 587 mV for pristine W18O49 NFs. Seo et al.9) reported the hydrogen evolution activity of W18O49@WS2 core-shell nanorods was better than that of WS2 nanotubes by controlled sulfurization reaction of W18O49 nanorods, indicating that highly conductive oxide nuclei play a key role in improving the activity of hydrogen evolution. An efficient electrocatalyst for hydrogen evolution made of Pd doped W18O49 nanofibers is reported by Zhao et al.10) The optimal 5 % Pd doped W18O49 nanofibers have an initial overpotential of only 65 mV and an exchange current density of 2.36 × 10-3 mA cm-2.
An n-type Selenium (Se) has been extensively studied as a photoabsorber in thin film devices such as solar cells, photocatalyst, xerography, and rectifiers.11) Moreover, Se has low surface states densities, large carrier lifetime12) and a high conductivity (1 × 10-3 Sm-1)13) which can affect the charge mobility. Yi et al.14) prepared Se-rich CoCH nanoarrays (Se NPs/CoCH NAs/CC) modified by Se nanoparticles on carbon cloth through simple electrodeposition reduction method, which has the characteristics of large surface area, higher charge transfer rate, rich Se sites and low overpotential (η10 = 84.3 mV and η350 = 321.1 mV) for HER. Nasir et al.15) performed comprehensive experiments and periodic density Functional theory (DFT) simulations of BiVO4 heterojunction with selenium (Se-BiVO4). At 1.3 V vs SCE, the Se/BiVO4 heterojunction increases the photocurrent density by 3 times, from 0.7 to 2.2 mA cm−2.
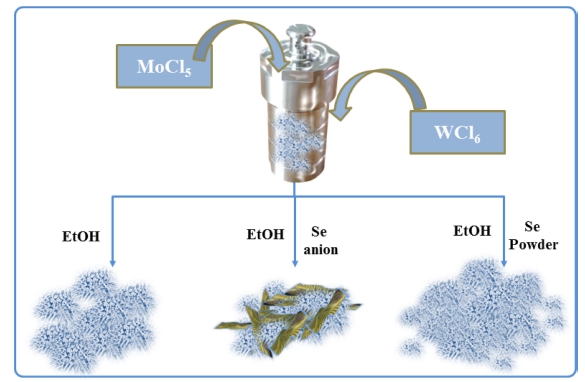
Intrigued by previous studies, this word was firstly focused on the effect of a series of Na2SeSO3 solution and selenium powder which was used to modify Mo- W18O49 as shown in Schematic 1. Furthermore, the resultant Se NPs/Mo-W18O49 nanosheets have relatively stable persistence when suffering long term operation in 0.5 M H2SO4 aqueous solution. The experimental results confirm that these novel Se NPs/Mo-W18O49 nanosheets exhibit much higher photoelectrochemical activities and stabilities than pure Mo-W18O49.
2. Experimental Details
2.1. Materials
MoCl5 (99.99 % metals basis), WCl6 (99.99 % metals basis), selenium (Se) powder (99.9 % metals basis, 200 mesh), anhydrous alcohol (EtOH) and sodium sulfite (Na2SO3, 95 %) were purchased from Aladdin, China. All of the reagents used in this experiment were analytical reagent grade and without further purification.
2.2. Preparation of electrocatalysts
A 3D urchin-like Mo-W18O49 nanostructure was prepared according to previous report with modification.7,8) In order to obtain 2 at% Mo doped W18O49 in this work, a MoCl5 solution with concentration of 5mg/mL was prprepared firstly by dissolving 10 mg of MoCl5 in 2 mL of absolute ethyl alcohol. Then 552 μL of MoCl5 solution drained with a pipette gun (100-1,000 uL, DLAB Scientific Co. Ltd.) and 0.2 g of WCl6 were added into 20 mL of absolute ethyl alcohol under stirring for 30 min. Subsequently the formed yellow solution was transferred into a Teflon-lined autoclave and heated at 180 °C for 12 h in air dry oven (DHG-9036A, Shanghai Jinghong Experimental Equipment Co., Ltd.). The resulting solid product was collected by extraction filtration, washed with distilled water and absolute ethyl alcohol, followed by dried in a vacuum freeze drier for 10 h.
According to literature research, a facile solvothermal method was used in this work in order to prevent oxidiation of Mo-W18O49 and easily obtain a series of Se modified samples via same synthesized process. The Se NPs modified Mo-W18O49 was prepared using the same solvothermal method for preparing 2 at% Mo doped W18O49 but adding two kinds of Se precursor materials together in this step. Na2SeSO3 solution was pre-prepared via chemical reaction of Na2SO3 with selenium powder under violently stirring at 90 °C. The detail synthesis process is described as below: 5 g of Na2SO3 and 0.5 g of selenium powder were added in 30 mL distilled water and then refluxed sufficient until a colorless clear liquid was formed. In order to obtain three kinds of electrocatalysts via precursor of Na2SeSO3, the 1 mL, 2 mL and 3 mL of Na2SeSO3 solution was added into the yellow solution respectively, and these formed products were named as 1 mL Na2SeSO3, 2 mL Na2SeSO3 and 3 mL Na2SeSO3. Meanwhile, 25 mg of selenium powder was selected as another Se precursor material replaced of Na2SeSO3 solution and named as 25 mg Se powder. The four kinds of resulting solid products were also collected by extraction filtration, washed with distilled water and absolute ethyl alcohol, followed by dried in a vacuum freeze drier for 10 h.
2.3. Characterization
The crystal structures of the compounds were characterized by X-ray diffraction (XRD) under Cu Kα (X 'pert3, PANalytical) dirradiation, and the morphology and microstructure of the compounds were characterized by scanning electron microscopy (Nova NanoSEM 450, FEI Company). The content of the element in the coating was identified by an energy disperse spectroscope (XFlash 6110, Bruker). The distribution of the element in the coating was characterized by the EDS mapping mode. X-ray photoelectron spectroscopy (XPS) was conducted to distinguish the chemical state (Escalab 250Xi, ThermoScientific).
2.4. Preparation of catalytic electrodes
To prepare the working electrode, 4 mg of catalysts and 30 ml of Nafion solution were dispersed in 1 mL of water-ethanol solution by sonicating for 2 h to form a homogeneous mixture. Then drop 5 mL solution onto a 3 mm diameter glassy carbon electrode (GCE) and dryUnder an infrared light. The electrocatalyst loading amount was controlled at 0.285 mg cm-2 for each catalytic electrode.
2.5. Electrochemical characterizations
The photocurrent and the electrochemical impedance spectra (EIS) were obtained using an electrochemical workstation (CHI 660E, China). A standard three-electrode system was performed in this photochemical study including counter electrode (Pt foil), reference electrode (saturated Ag/AgCl electrode) and working electrodes (compounds on glassy carbon electrode).16) 0.1 M sodium sulfate aqueous solution was acted as electrolyte. According to the Nernst equation, the potential vs. reversible hydrogen electrode (RHE) could be obtained from the measured potential vs. Ag/AgCl. The amplitude of EIS was set to 5 mV and a frequency range of 1,000 kHz to 0.1 Hz was also setup. The HER electrocatalytic activity of Mo- W18O49 and Se NPs modified Mo-W18O49 was examined in the standard three-electrode system (6 mm carbon rod used as counter electrode) with sweeping potential from -0.7 V to 0 V versus Ag/AgCl electrode at a scan rate of 5 mV s-1 in 0.5 M H2SO4 at room temperature. The equation of E(vs RHE)= E(vs Ag/AgCl) + 0.1976 V + 0.0591*pH was used to calibrate all obtained polarization data to the reversible hydrogen electrode and all dates were reported without iR compensation.
3. Results and Discussion
3.1. Phase and morphology
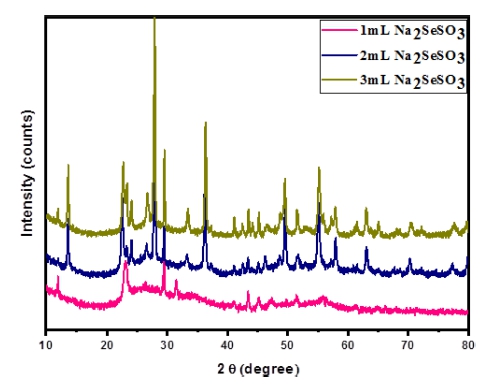
The 2 at% Mo-doped W18O49 and Se NPs modified Mo- W18O49 samples were investigated by X-ray diffraction (XRD) as shown in Fig. 1. Here, the peaks named Se anion and Se powder are related to two kinds of electrocatalysts synthesized by 1 mL Na2SeSO3 solution and 25 mg of selenium powder. Comparing with these three peaks, the main peaks at 2θ = 47.7° and 23.3° match best with the (020) and (010) reflections of the monoclinic phase W18O49, while all other broad diffraction peaks are much weaker, suggesting that the W18O49 nanostructure with preferential growth occurs along the (010) direction.17) Intense diffraction peaks were observed at 13.8°, 27.7°, and 42.0° (red clubs marking) which are corresponding respectively to (002), (004), and (006) diffraction planes of two-hexagonal (2H) MoSe2 (JCPDS #872416).18) And other peaks marked with organge diamond are belonged to elemental Se.19)
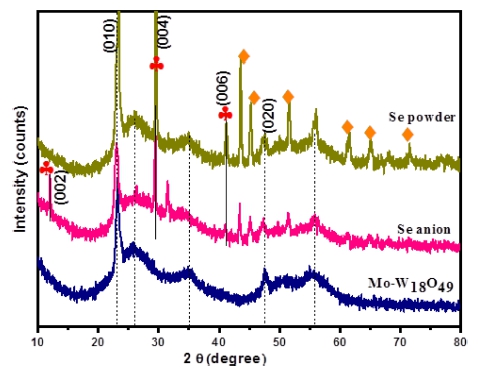
Fig. 1
XRD patterns of as-prepared Mo-W18O49, 1 mL of Na2SeSO3 solution treated Mo-W18O49; 25mg Se powder treated Mo-W18O49.
Fig. 2 shows the X-ray diffraction patterns of different amount of Na2SeSO3 solution treated Mo-W18O49. Comparing with these three peaks, it is not difficult to find that peaks position was changed, with increasing the solution amount to 2 and 3 mL, the main peaks of W18O49 were disappeared. And some by-products such as WO3-x· H2O were generated, which can be ascribed to the hydrolyzation of WCl6 with some amount of H2O, similar result can be seen in the published work which was prepared under open air.20)
The difference of micro-surface structures and morphologies between 2 at% Mo-doped W18O49 and Se NPs modified Mo-W18O49 were characterized by FE-SEM (Fig. 3). Scanning electron microscopy (SEM) is used to examine the morphology of samples at very high magnification with same scale length. Fig. 3(a, b) shows us the morphology of individual 2 at% Mo-W18O49 nanostructure is urchin-like, and the uniform average diameter of such hierarchical formation material is typically 1 μm. Fig. 3(a, b) shows us a different morphology which related to 1 mL of Na2SeSO3 solution treated Mo-W18O49. The average size of nanosheet shown in Fig. 3(c), can be visual estimation about 0.5 ~ 1 mm. These nanosheets are composed of different sized nanosheets with superimposed and interwoven station. And Fig. 3(d) shows us high quality scanning electron microscopy with 10,000-magnification, it can be clearly seen that most of nanosheets were interwoven and superimposed with some spherical particles. Based on its related result of XRD analysis in Fig. 1, a little amount of H2O exist in 1 mL of Na2SeSO3 solution used can easily changes the synthesized conditions, thus different morphology was observed. Fig. 3(e and f) shows a morphology which related to 25 mg Se powder treated Mo-W18O49, a similar but little different morphology was observed comparing with Fig. 3(a-b), such as the hollow part in the middle of the urchin structure is reduced and the size of urchin structure is increased.
3.2. Chemical state
Inder to distinguish the chemical state of pure Mo- W18O49 and Se NPs modified Mo-W18O49 composite, XPS measurement was performed as shown in Fig. 4. The full XPS spectrum indicates that the Se NPs modified Mo-W18O49 (1mL Na2SeSO3) composite is mainly composed of W, O, Se and Mo elements. Before Se precursor treated, the obtained XPS result was similar Zhang et al.21) published work. Comparing with the result of pure Mo- W18O49 with 1 mL Na2SeSO3, it was easily the peaks of Se at the range of 0 to 100 eV.
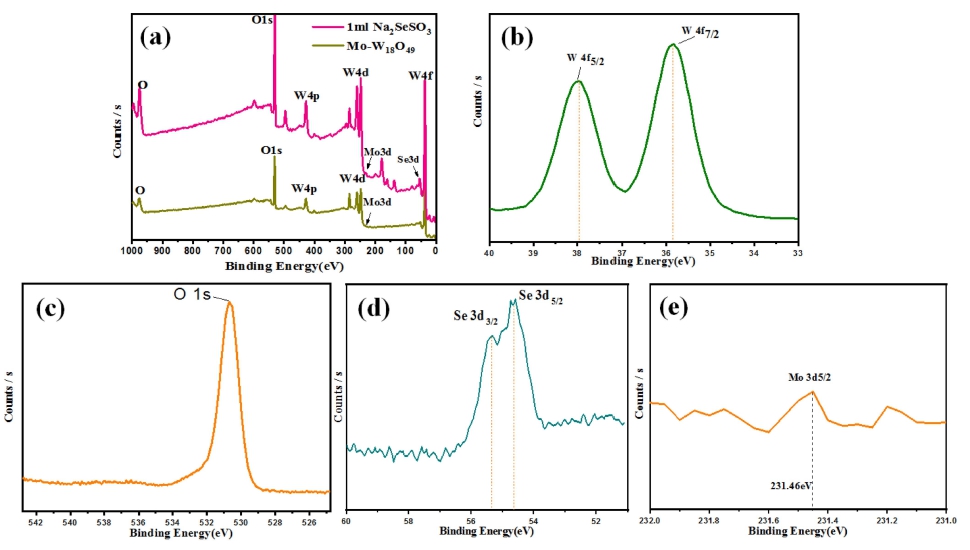
Fig. 4
(a) XPS survey spectrum of pure Mo-W18O49 and 1 mL of Na2SeSO3 solution treated Mo-W18O49; XPS spectra of (b) W 4f, (c) O 1s, (d) Se 3d and (f) Mo 3d for 1 mL of Na2SeSO3 solution treated.
XPS characterizations are performed to determine the chemical valence of W species, yielding a doublet peak for W4f5/2 and W4f7/2. The XPS peak at binding energy of 37.98 eV refers to W6+, and the other peak at 35.78 eV is for W5+ (Fig. 4b), and its total percentages of W6+ and W5+ species are determined to be 39.56 % and 60.44 %.22) For the O 1s XPS spectra shown in Fig. 4c, the binding energy of 530.8 eV was attributed to the O2− in the W18O49, while the peak at 532 eV probably corresponded to the hydroxyl group.23) As shown in Fig. 4d, Se 3d orbit shows two small peaks at 54.7 and 55.3 eV which are due to Se 3d5/2 and Se 3d3/2.24) The states of Mo species in the Mo-W18O49 samples are determined from the highresolution XPS spectra [Fig. 4(e)]. The Mo 3d binding energy values appear at 231.46, which correspond to Mo 3d5/2 and attributed to Mo4+.7)
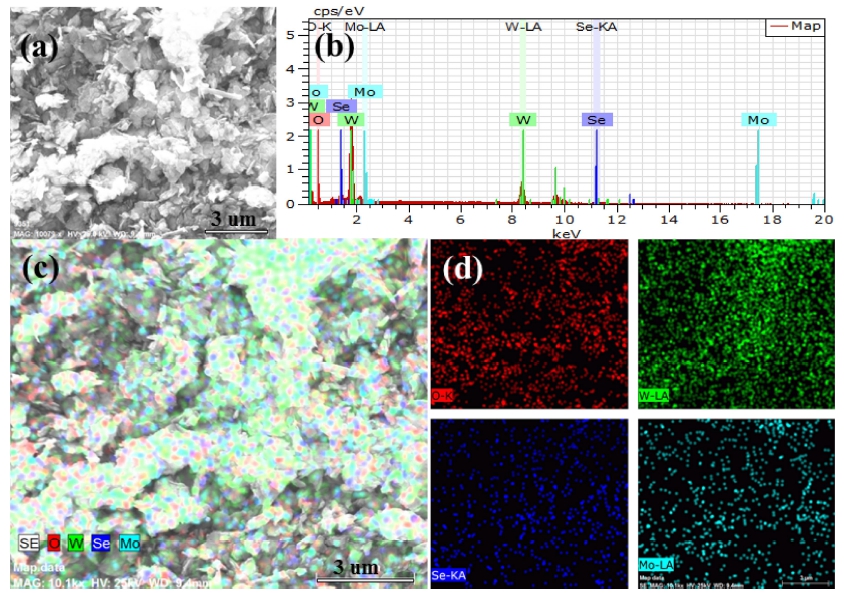
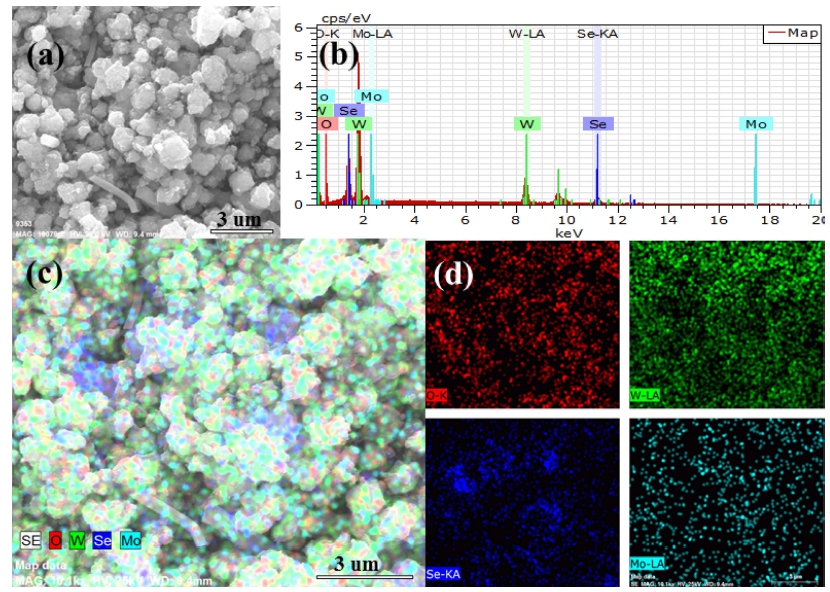
The energy dispersive X-ray (EDX) spectrum and Energy dispersive spectroscopy (EDS) corresponding element mapping are shown in Fig. 5 and Fig. 6, which related to the Se NPs modified Mo-W18O49 composite treated via 1 mL of Na2SeSO3 solution and 25 mg of Se powder, respectively. Fig. 5 and Fig. 6 demonstrate the elemental distribution of O, W, Se and Mo are distributed of nanostructure, and the depth of distribution is approximately consistent with the specific gravity of EDX analysis. Comparing with Fig 5(c) and Fig 6(c) (scale length is 3 μm), Se element was uniformly distributed in the nanostructure which is related to Se anion treated sample, while Se powder treated Mo-W18O49 sample exhibits serious agglomeration state of Se. According to the favorable, it can be speculated that a good dispersion of small particles could provide more reactive sites than aggregated particles.25)
3.3. Electrochemical performance
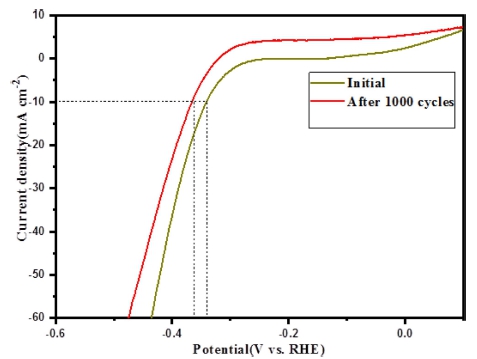
The electrocatalytic HER activities of Mo-W18O49, Se anion and Se powder treated Mo-W18O49 deposited on a glassy carbon electrode (GCE) were evaluated by linear sweep voltammetry in 0.5 M H2SO4 (Fig. 7). For comparison, the catalytic HER activity of these five kinds of samples was studied under the same conditions with similar loading amount of ≈0.285 mg cm−2. Comparing with Pure Mo-W18O49, Se powder treated Mo-W18O49 exhibit similar results but slight lower current density at RHE range from -0.5 to 0.2 V. Particularly, 1 mL of Na2SeSO3 solution treated Mo-W18O49 catalysts exhibit overpotential as 340 mV at cathodic current densities of 10 mA cm−2, while the overpotential is 488 mV for pristine Mo-W18O49. Based on the results of FE-SEM and Energy dispersive spectroscopy (EDS) corresponding element mapping, 1 mL of Na2SeSO3 solution treated Mo-W18O49 catalysts shows us most of nanosheets were interwoven and superimposed with some spherical particles. And most of Se element was uniformly distributed in the nanostructure which is related to Selenium treated sample. The enhancement of HER activity can be contributed to the special morphology which can provide more reactive sites and electron ion transport channels. With increasing the amount of Na2SeSO3 solution its realted overpotential was increased. This result is agree with the XRD results, the by-products own low water splitting ability, thus it is not suitable to use as electrocatalyst. The electrochemical stability of Se NPs/Mo-W18O49 was also studied in 0.5 M H2SO4 solution. As shown in Fig. 8, after 1,000 cycles of CV measurement, a little decrease can be observed.
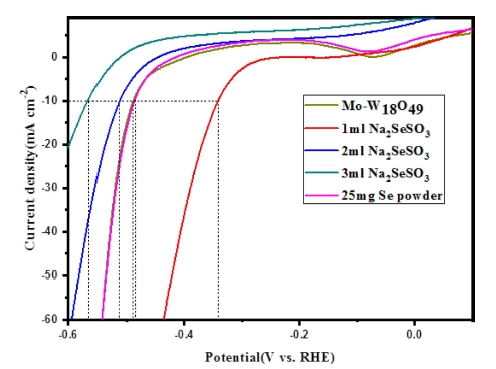
Fig. 7
Polarization curves of Mo-W18O49, Se anion and Se powder treated Mo-W18O49 in 0.5 M H2SO4 f or HER .
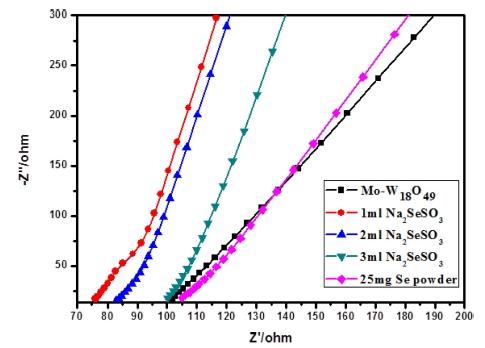
Electrochemical impedance spectroscopy (EIS) is investigated at various HER overpotentials to demonstrate the relative enhancement of the catalytic activity. As shown in Fig. 9, the minimum electrochemical impedance of the sample treated with 1 mL sodium thioselenate is about 75 ohms, which is about 30 ohms lower than that of the original Mo doped W18O49 sample after selenization. The decrease of the electrochemical impedance point means that the charge transfer rate at the interface of the material is the fastest, and the faster electron mobility may further improve its conductivity. On the basis of the above results, we attribute the best electrochemical performance for Se NPs/Mo-W18O49 composite mainly to better electron transport and efficient electron-hole separation. Above all, it can be clearly seen that the Se NPs could serve as excellent co-catalyst for Mo-W18O49 to enhance its electrochemical performance.
4. Conclusion
In summary, a series of Se NPs/Mo-W18O49 composite with different Se precursor treated were successfully prepared for the improved HER performance. The surface properties as structural and chemical composition were investigated. The as-prepared Se NPs/Mo-W18O49 exhibits most of nanosheets were interwoven and superimposed with some spherical particles., an overpotential as 340 mV is required to attain current density of 10 mAcm-2 in 0.5 M H2SO4 aqueous solution. In addition, the special morphology for Se NPs/Mo-W18O49 promotes electron transfer, ensuring fast interfacial charge transfer to improve electrocatalytic reactions for practical application in hydrogen evolution.