1. Introduction
Most of textile, plastics, paper industries are generating waste effluents everyday containing substantial amount of organic dyes as pollutants including Texbrite MST-L. Discharging them into the water bodies without any prior treatment can cause havoc on the environment.1) Photocatalysis as an advanced oxidation technique, has been developed to reduce he environmental pollution.2,3) Most nanocrystalline semiconductors own nanometer size and large surface/volume ratio thus have the unique physical and chemical properties. Up to now, nanocrystalline semiconductors used as photocatalyst have aroused great interest to initiate interfacial redox reactions.
In last score years, TiO2 has been widely researched by a lot of science researchers due to inexpensiveness and reliable photochemical and biological stability.4,5) However, TiO2 nanoparticles cannot efficiently utilize the solar energy. Because it can be excited only by the ultraviolet light, thus only about 4 ~ 6 % of the solar spectrum can be used. In addition, photocatalytic activity of TiO2 was widely decreased due to the high electrons and holes recombining rate. In order to enhance the photocatalytic activity and extend the light absorption of TiO2 into th e visible region, many methods have been researched. A lot of semiconductors such as CdS,6) CdSe,7) Bi2S3,8) and InAs,9) have been investigated to sensitize TiO2 as a visible light absorber in recent years.
Metal selenides have attracted more attention during the past few years due to their special electronic and optical properties and potential applications.10) Ag2Se exists as a low-temperature phase (β-Ag2Se), and a hightemperature phase (α-Ag2Se) with the phase transition point at 1,331 °C.11) Among these two kinds of Ag2Se, low-temperature phase Ag2Se with narrow-band-gap has been widely used for a photosensitizer. Cao et al.12) reported the preparation of single-crystalline Ag2Se via a solvothermal route and characterization its photocatalytic activity by photodegradation of Rhodamin B dye under ultraviolet (UV) light irradiation. Meng et al.13) recently reported an Ag2Se-graphene/TiO2 composite which showed a red-shifted absorption and better visible light photocatalytic activity than pure TiO2. Mishra et al.14) recently reported a research on synthesis of Ag2Se nanomaterials based TiO2 (P25) with good photocatalytic acitivity. To the best of our knowledge, few reports were studied about the photocatalysis of Ag2Se-TiO2 composites under visible light irradiation although some related researchs were studied as mentioned above.
It is well-known that the physical and chemical properties of materials could be attributed to its particle size and microstructure. Selection of a suitable synthetic route to these nanomaterials is crucial as their properties may evolve during the preparation. According to preliminary literature research, in this work we present the studies on the preparation of Ag2Se nanoparticles modified TiO2 nanocomposite using a facile hydrothermal synthesis method. Comparing with the published work of Mishra et al.,14) this facile synthesis process owns advantage such as a direct-purchased AgNO3 and simply pre-synthesized Na2SeSO3 were used and displaced the precursors of silver(I) reagents AgX (X- = iodide, trifluoroacetate, triflate) and selenoethers R2Se (R = Me, tBu) synthesized in different solvents in that paper. The enhancement of visible light photodegradation of TBA-L and 1,5-diphenyl carbazide (DPCI) for Ag2Se@TiO2 was attributed the photosensitization of Ag2Se and good morphology which leads to efficient separation of electrons and holes.
2. Experimental
2.1. Materials
The titanium (IV) n-butoxide (TNB, C16H36O4Ti), ethylene glycol, anhydrous ethanol, silver nitrate (AgNO3), selenium (Se) powder, ammonium hydroxide (NH3H2O, 28 %) and sodium sulfite (Na2SO3, 95 %) were purchased from Aladdin, China. Texbrite BA-L was purchased from Texchem Co. Ltd, Korea. Titanium oxide nanopowder (P25, Evonik, ex-Degussa) with was purchased from Sigma-Aldrich Chemistry.
2.2. Synthesis of Ag2Se@TiO2 Nanocomposite
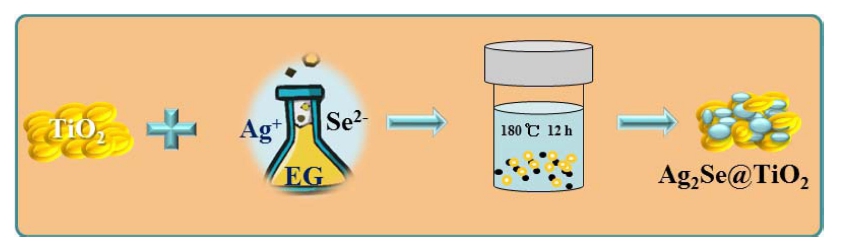
Fig. 1 is the illustration about synthesis of Ag2Se@TiO2 nanocomposite by one-pot hydrothermal synthesis method. Here, nanoscale TiO2 as start material was pre-prepared according to previous published work.11) Na2SeSO3 was pre-prepared via chemical reaction of Na2SO3 with selenium powder under violently stirring at 363 K. The detail synthesis process is described as below: 5 g of Na2SO3 and 0.5 g of selenium powder were added in 30 mL distilled water and then refluxed sufficient until a colorless clear liquid was formed.
In the next step, 4.38 mM as-prepared TiO2 (0.35 g) nanoparticles and 0.5 mM AgNO3 (0.085 g) was added in 30 mL ethylene glycol which contain 2 mL of NH3H2O. And the mixture was followed by stirring for 1 h under ambient temperature for adsorption of with TiO2. Finally, 2.5 mL of Na2SeSO3 solution was added in the /TiO2 mixture mixed following by ultrasonicated for 30 min. The thoroughly mixed solution was transferred into a Teflon-lined stainless steel autoclave that was sealed. The contents were then warmed to 180 °C for 12 h. The obtained sample labeled as Ag2Se@TiO2 with mol ratio of Ag2Se: TiO2 = 1:8.76 was washed several times with water and ethanol, then it was dried in a vacuum oven overnight. The control sample of Ag2Se was synthesized with this similar method without TiO2 was added into the mixture.
2.3. Characterization
To determine the crystal phase and components of the as-prepared samples, XRD characterization was carried out at room temperature using XRD (X'Pert3, PANalytical) with Cu Kα radiation (λ = 1.54056 Å) in the range of 2θ = 10-80° at a scan speed of 1.2° m-1. The morphology and microstructure of the compounds were characterized by Scanning Electron Microscopy (SEM, Nova 450) with an energy dispersive analysis system of X-ray analysis (EDX). Transmission electron microscopy (JEM-2010, Japan) was used to examine the size and distribution of Ag2Se@TiO2. The BET surface area of these photocatalysts was determined through nitrogen adsorption at 77 K by a BET analyzer (Monosorb, USA). The decomposition kinetics for the photocatalytic activity was measured by using a spectrometer (UV-2450, SHIMADZU).
2.4. Evaluation of Reactive Oxygen Species
Firstly, each 10 mL of DPCI stock solutions (1.00 × 10-2 mol/L) were added to four flask (100 mL), then 50 mg of Ag2Se@TiO2 samples was added to these flasks in the order of 1 to 4, respectively. And then it was diluted to 100 mL with water. The final DPCI concentration and used amount of Ag2Se@TiO2 were 1.5 × 10-3 mol/L and 1.5 g/L, respectively. After visible light irradiation for 0 - 120 min, 10 ml of solution was removed from each reactor and extracted with benzene. In the second step, benzene was used to dilute the extracted solutions to 10 mL followed by analyzed UV-Vis spectra of the solutions via a spectrometer.
2.5. Visible Light Photocatalytic Properties
The visible light photocatalytic properties of the control sample P25, TiO2, Ag2Se and Ag2Se@TiO2 were evaluated by photodegradation of Texbrite BA-L under radiation by 25 W LED lamp (PHILIPS, brightness 2700 lm, 6500 K). The intensity of the light source was controlled as 100 mW/cm2 and detected by optical power meter (FZ400, NBeT Company). In the photodegradation process, 30 mg of the photocatalysts were placed in 80 mL of solution of TBA (5 mg/mL). In order to establish adsorption/ desorption equilibrium, the solution was remained in the dark about 2 h before irradiation. At given time intervals, 5 mL of mixture was centrifuged for 5 min to instantly remove the solid material for further analysis. The photocatalytic behavior of the samples was analyzed through the absorbance spectrometry with a UV/Vis spectrophotometer (UV-2450, SHIMADZU).
3. Results and Discussion
3.1. Characterization
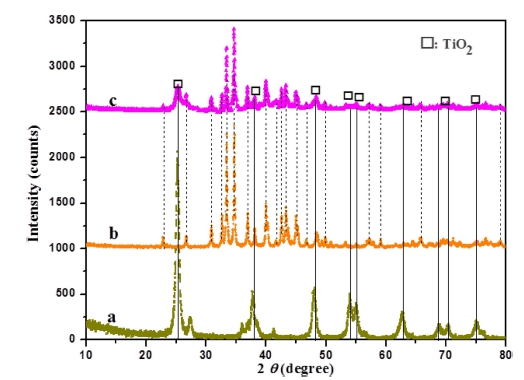
The crystal phase structures of as-prepared TiO2, Ag2Se and Ag2Se@TiO2 composites were characterized by XRD as shown in Fig. 1. It can be found that the special crystal planes of (101), (004), (200), (105), (211), and (204) are assigned to the anatase TiO2 phase (JCPDS file, No. 21- 1272).15) The main sharp peaks were assigned to (002), (111), (102), (120), (112), (121), (013), (031), (113), (032), (123) crystal planes originated from the orthorhombic Ag2Se phase, which were in accordance with the results reported by Zhang et al.16) with the lattice parameters were close to a = 4.333 Å, b = 7.062 Å, and c = 7.764 Å (JCPDS card no. 24-1041). No impurity phase is detected. The broadening of these diffraction peaks indicates that the sample is nanosized. Comparing with pure Ag2Se, after introduced of TiO2, it can be clearly seen that the intensity of the Ag2Se peaks was decreased which confirms the development of synthesis homogeneously dispersed Ag2Se and TiO2 nanocrystals.
The micro-surface structures and morphologies of the as-prepared TiO2, Ag2Se and Ag2Se@TiO2 were characterized by FE-SEM [Fig. 3(a)]. Scanning electron microscopy (SEM) is used to examine the morphology of samples at very high magnification. In Fig. 2(a) and (b), pure TiO2 has a small particle size with a good dispersion state, also there is found a little tendency to agglomerate in some parts. Fig. 2(c) and (d), shows us a favorable morphology but a little tendency to agglomerate of Ag2Se nanoparticles which constitutes of small and bulk sized particles. As shown in Fig. 2(e) and (f), it is worth noting that after introduction of TiO2 nanoparticle, the morphology of Ag2Se was changed which shows us a smaller sized flake-like shape (~ 1 - 2 μm in width) combined with small sized TiO2 nanoparticles. The favorable morphology of Ag2Se@TiO2 composite could be observed from the high magnification image in Fig. 2(d) and (f), the transformational morphology of Ag2Se can be ascribed to the introduction of TiO2 powder which may change the reaction condition among the reaction process, the similar morphology of Ag2Se also can be found in Mishra’s report.14)
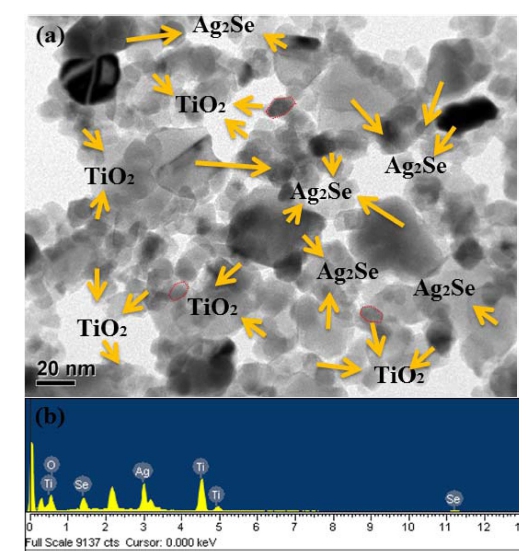
Fig. 3
(a) TEM micrographs and (b) EDX elemental microanalysis of as-prepared Ag2Se@TiO2 nanocomposite.
For the obtaining of more detailed and higher magnification, the surface nanostructures and particle sizes of Ag2Se@TiO2 nanocomposite were studied by TEM analysis depicted in Fig. 3(a). TEM images revealed that, Ag2Se@TiO2 sample displayed well-dispersed Ag2Se owns special morphology of small sized plate which have average size around 20 - 40 nm. Meanwhile some welldispersed small sized nanoparticles with an average size of 15 nm can be clearly seen, it can be assigned to TiO2 nanoparticles due to it has unique crystal morphology. The presence of distinct juncture between two crystal phases further confirmed the strong interaction between Ag2Se and TiO2 in Ag2Se@TiO2. According to the favorable, it can be speculated that a good dispersion of small particles could provide more reactive sites than aggregated particles.17) Chemical composition analysis and element weight % of prepared Ag2Se@TiO2 naonocomposite were examined by EDX. The main elements such as Ti, O, Ag and Se are existed in the Fig. 4[(b) and (d)]. The oxygen and Ti element mainly comes from the TiO2 nanopowder. And the strong Kα and Kβ peaks from Ti element appear at 4.51 and 4.92 keV, while a moderate Kα peak of the element O appears at 0.52 keV.18) The strong Kα and Kβ peaks appear at 11.2 and 12.4 KeV can be attributed Se element while the moderate Kα peak appears at 2.98 KeV can be attributed Ag element. 19,20)
3.2. VL Photocatalysis Performances
3.2.1 Adsorption Properties
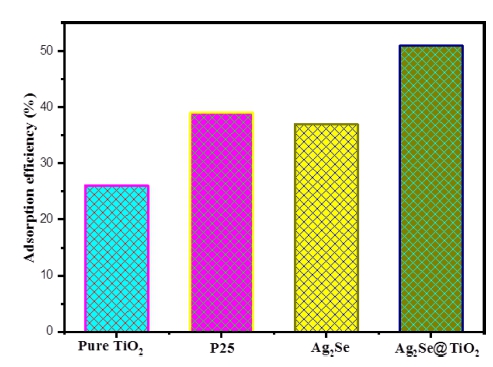
In the first stage of photocatalytic experiment, the adsorption-desorption equilibrium should be achieved via the adsorption process which was carried out by stirring for 120 min in the darkness box. From the result shown in Fig. 4, the level of Texbrite BA-L adsorption by Ag2Se@TiO2 was higher than that of the TiO2, P25 and Ag2Se control samples. This can be attributed to the big surface area of the Ag2Se@TiO2 catalysts about 38.72 m2/g, this could be correlated to strong adsorption ability. The surface area of Ag2Se is about 21.26 m2/g which is higher than that of pure TiO2 sample about 16.41 m2/g. Surface area of P25 is similar with TiO2 catalysts about 20.14 m2/g. After establishing an adsorption–desorption equilibrium, for Ag2Se@TiO2 catalyst the Texbrite BA-L dye solution was removed about 51% while only 26 % for pure TiO2.
3.2.2. Degradation effects of Texbrite BA-L (TBA)
The visible light (VL) photocatalytic activity was investigated by degradation of Texbrite BA-L used as an industrial dye. Fig. 5(a), shows us that the relative concentration of Texbrite BA-L for anatase TiO2 is lower than that of P25 due to pure TiO2 cannot be actively excited by visible light irradiation, similar results have also been observed by Yu et al.21) The photocatalytic degradation of TBA solution with Ag2Se@TiO2 composite was better than any other composites. These results can be explained by the main reason: the small particles with good dispersion could provide more reactive sites for the reactants than aggregated particles.22) As mentioned above in surface characteristics, Ag2Se@TiO2 composite has a favorable morphology for adsorption more organic pollutants. In addition, the favorable morphology also played an important role in shuttling visible light photo-induced electrons generated from Ag2Se into the conduction band of TiO2 efficiently.
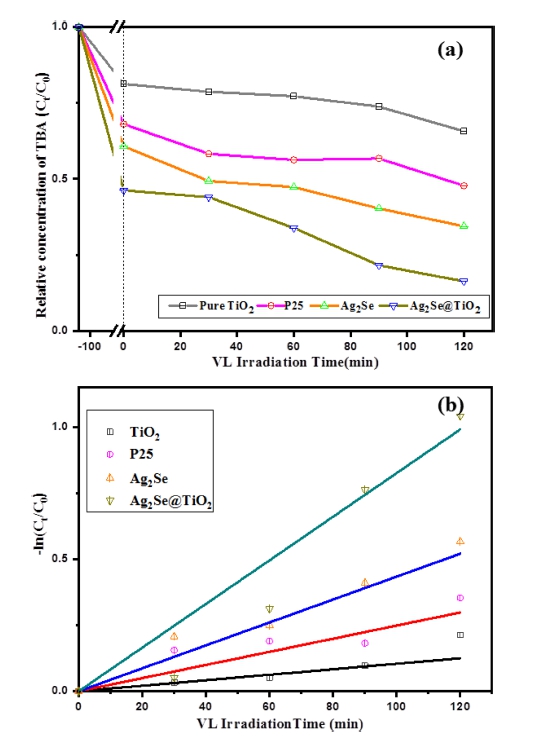
Fig. 5
(a) Relative concentration of TBA (C/C0) for adsorption and degradation; (b) Apparent first order kinetics of TBA photocatalysis degradation against P25, TiO2, Ag2Se and Ag2Se@TiO2 nanocomposite.
The photocatalytic degradation effect for different photocatalysts under visible light obeys pseudo-first-order kinetics with respect to the concentration of Texbrite BA-L:
Integrating this equation (with the restriction of c = c0 at t = 0, with c0 being the initial concentration in the bulk solution after dark adsorption and t the reaction time) will lead to the following expected relation:
where ct and c0 are the reactant concentrations at times t = t and t = 0, respectively, and kapp and t are the apparent reaction rate constant and time, respectively. According to this equation, a plot of -ln(ct/c0) versus t will yield a slope of kapp. According to the results displayed in Fig. 5(b), for Ag2Se@TiO2 composites the photo-discolourization rate constant of Texbrite BA-L (TBA) reaches about 3.8 × 10-4 min-1 which is much higher than the corresponding values for pure Ag2Se (8 × 10-5 min-1), P25 (1.9 × 10-4 min-1), and 3.2 × 10-5 min-1 of TiO2.
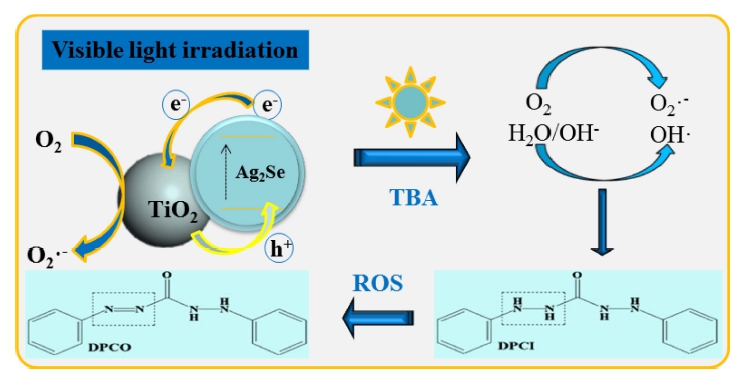
The visible light photocatalytic activity of Ag2Se@TiO2 nanocomposite is much better than the other samples. In our nanosized Ag2Se coupled TiO2 nanocomposite system, Ag2Se semiconductor has a beneficial role which can improves photoexcited charge separation rate and extends the response acitivity of TiO2 into visible range. Based on previous researchs,23,24) according to the results of pollutant degradation and ROS generation a mechanism was speculated shown in Fig. 6. The visible light photogenerated electrons were transferred to the CB of Ag2Se, thereby increasing the number of electrons as well as the rate of electron-induced redox reactions. The Ag2Se@TiO2 shows enhanced catalytic activity due to the high charge separation induced by the synergetic effects of TiO2 and Ag2Se. While the generated electrons (e-) react with the dissolved oxygen molecules in water, there will generate some oxygen peroxide radicals in the mixture solution. The positive charge hole (h+) can react with OH- derived from H2O to form hydroxyl radicals OH•. The Texbrite BA-L dye may be degraded by oxygen peroxide radicals and hydroxyl radicals OH• to CO2, H2O, a nd o th er mineralization products.25)
3.2.3. Generation of ROS
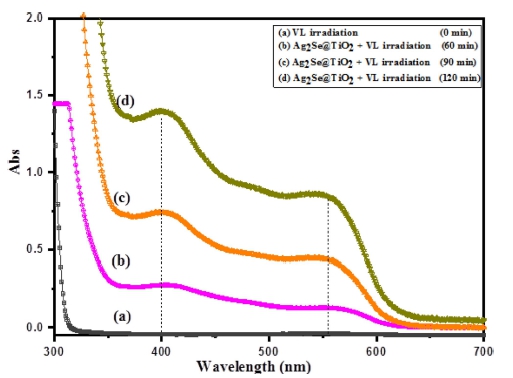
In order to detect the ROS generation of Ag2Se@TiO2 nanocomposite, UV–vis spectra of DPCO extract liquors was analyzed via a spectrometer. The four reaction solutions as discussed earlier were placed in an ultrasonic chamber in a 200 mL borosilicate glass container. Under visible light irradiation, 1, 5-diphenyl carbazide (DPCI) can be oxidized by oxidizing substances into 1, 5-diphenyl carbazone (DPCO). Under visible light irradiation, Ag2Se@TiO2 nanocomposites are excited to higher energy state. That is, some electrons are transmitted from a valence band (VB) to a conduction band (CB). Meanwhile, the generated electron hole pair is formed by the TiO2. Simultaneously, the excited electron transfer from the conduction band of Ag2Se to the TiO2 surface. The obtained •OH radicals can oxidize 1,5-diphenyl carbazide (DPCI) into 1,5-diphenyl carbazone (DPCO). The DPCO was extracted by the benzene solvent. As shown in Fig. 7, absorbance spectra and the observed absorbance peaks around 400 nm of Ag2Se@TiO2 nanocomposite, which are in agreement with the results reported in the literature.26,27)
3.2.4. Stability of Ag2Se@TiO2 and P25
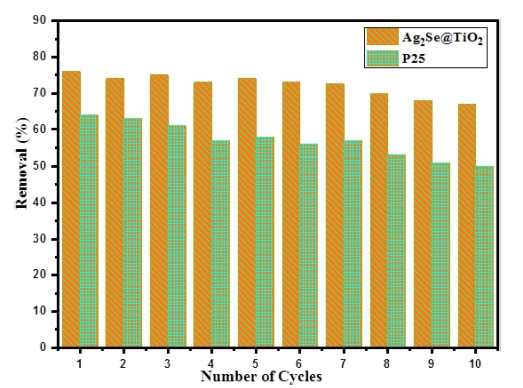
It is well known that photocatalysts possess the advantages of high stability and rapid recovery without secondary contamination of water after treatment, however the reusability of most titanium dioxide-based hybrid catalysts is affected by mechanical or chemical instability modifiers.28) In order to investigate the long-term stability of Ag2Se@TiO2 nanocomposite and P25 under visible light irradiation, 10 times recycling experiments are carried out by repeating the Texbrite BA-L degradation process. As shown in Fig. 8, after ten cycles the removal efficiency of P25 decreased from 64 to 50 % (14 % drop). By contrast, Ag2Se@TiO2 photocatalyst maintained as high Texbrite BA-L removal efficiency as 67 % after 10 runs (only 9% drop). The reduction in the degradation efficiency among the cycles may be explained by the formation of by-products and their accumulation in the cavities and on the active surface sites of the as-prepared photocatalysts. These results further confirm that Ag2Se@TiO2 nanocomposite can be used as a catalytic material in an environmental purication process or in waste water treatment in the future.
4. Conclusions
A novel Ag2Se@TiO2 nanocomposite photocatalyst was successfully prepared through a facile hydrothermal synthesis method. The adsorption and surface properties as structural and chemical composition of the Ag2Se@TiO2 were investigated. It shows that the TiO2 particles homogenously coated by the small sized Ag2Se plates with uniform particle which indicated that morphology of Ag2Se was effectively ameliorated after introduction of TiO2 nanoparticles. In this work, the Texbrite BA-L degradation rate constant of as-prepared Ag2Se@TiO2 nanocomposite is two times as much as that of P25 which can be attributed to well-dispersed TiO2 coupled with Ag2Se.