1. Introduction
Recently, numerous works have been reported on the titanium aluminide, TiAl.1-3) There are no doubt that these efforts are motivated by the potential of the compound as a heat resisting structural material in the near future. Intermetallic compound TiAl is equi-atomic composition at stoichiometry and has L10 type ordered structure based on face centered tetragonal with a c/a ratio of 1.02.4) It has a low specific gravity, being approximately 3.8, and maintains its high specific modulus and strength at elevated temperatures which are superior to the conventional nickel-base superalloys.5) In addition, the Ti-Al alloy is shown to have good oxidation resistance as well as creep resistance.6,7) With such favorable properties, the practical application of the compound is urgently expected and the effect has been made to improve poor room temperature ductility which is the major draw back as a structural material. For the alloy designing of the compound, such fundamental information is necessary as the effect of deviation from stoichiometry and that of ternary additions. Unfortunately, we have to admit that even the binary titanium-aluminum equilibrium phase diagram to define the phase relation involving γ and α2 has yet been established.
In the present work, the Ti-Al alloys are rapidly solidified using hammer anvil method and investigated microscopically in order to understand the phase relations between the TiAl(γ) phase and neighboring phases. The information obtained in this study has been discussed in terms of solidification process including solid state reactions and phase stability.
2. Experimental Procedure
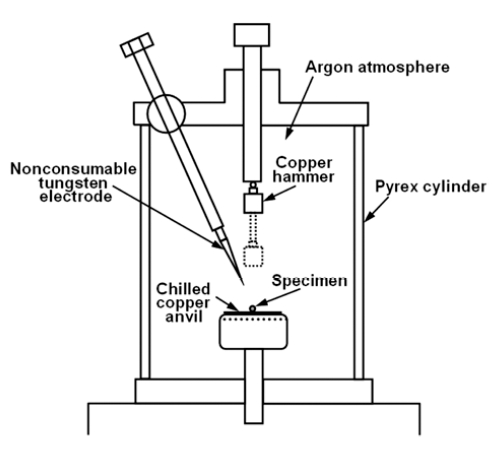
Master ingots of binary Ti-Al specimens, containing 50, 52, 54, 56, 58 and 60 atomic percent aluminum, were prepared by arc melting using a non-consumable tungsten electrode under an argon atmosphere. Each ingot obtained was approximately 20 g in weight. Specimens for rapid solidification, being 0.1 g in weight, were cut out from the ingots using an abrasive wheel. Rapid solidification was carried out by hammer anvil method under an argon atmosphere. Remelting before rapid solidification was attained by arc discharge to the specimen placed on water-chilled copper anvil. Rapid solidification to foils, being about 20 mm in radius and several tens of μm in thickness, were achieved by dropping a hammer made of copper with a flat end onto remelted specimen. Fig. 1 shows the schematics of the experimental device for the rapid solidification.
Quenching rate attained by this method is presumed to be 107 or 108 K/s. X-ray diffractometry was carried out on the each rapidly solidified foils to identify the phases present. Microstructures of the foils were investigated using JEOL JEM-3010 transmission electron microscope with an accelerating voltage of 300 kV.
3. Results and Discussion
3.1 Quenching rate
In this study, the hammer anvil technique was chosen for the rapid solidification, because it is known to have the highest quenching rate among the conventional quenching methods such as melt spun and single or twin roll method. It has been reported by Aboki et al. that an unusual microstructure involving non-equilibrium phases is found in Ni3Al rapidly quenched by a hammer-anvil technique which has never been observed in foils prepared by the twin roll method.8) They estimated that their hammer anvil method provided a magnitude of order faster than twin-roll method. The hammer anvil method in the present work is presumed to provide the same order of quenching rate because the results shown by Aboki et al. have been reproduced with the apparatus shown in Fig. l.
3.2 Microstructures of the rapidly solidified alloys
Microstructures of rapidly solidified alloys containing 50 and 54 percent aluminum are shown in Fig. 2. They in common give a structure consisting of TiAl(γ) and Ti3Al(α2) phases. In both alloys, TiAl phase with grain size of a few μm is the matrix. To be noted is that the morphology of α2 phase differs along with aluminum content. In alloys with 50 percent aluminum shows the Ti3Al particle with similar size to TiAl grains. A similar microstructure is observed in alloys containing 52 percent aluminum.

Fig. 2
Transmission electron micrographs of rapidly solidified Ti-Al alloys containing (a) 50 % and (b) 54 % aluminum. Both alloys show duplex structures consisting of TiAl and Ti3Al. Morphologies of Ti3Al vary with aluminum content.
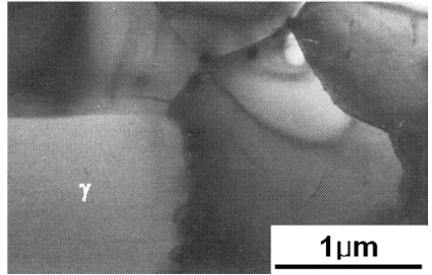
Alloys with 54 percent aluminum show more complex structures, in which α2 exists not as a particle, but in a fine lamellar form in a particle with around 1 μm in size consisting of alternating γ and α2. To be noted is that such two phase structures are observed in these aluminum-rich alloys although the γ/γ+α2 phase boundary is believed to lay on about 50 percent aluminum according to some equilibrium phase diagrams. It is considered that these metastable structures must be caused by the high quenching rate in this method. These results are consistent with previous works by some investigators. Vujic et al. show that the γ/γ+α2 phase boundary is on approximately 55 percent aluminum in their alloys rapidly solidified through the same method.9) Hall and Huang have observed the two phase structures for alloys containing 54 percent aluminum although their experiment is by melt spun method.10)
Alloys with over 56 percent aluminum content show virtually single phase γ structures nevertheless quite small amount of particles with the lamellar structure are present. An example of the microstructure is shown in Fig. 3 for the alloy containing 60 percent aluminum. The grain size of γ matrix is also 3 to 4 μm whereas the particles are smaller than 0.1 μm.
3.3 High temperature phase of alloys with titaniumrich compositions
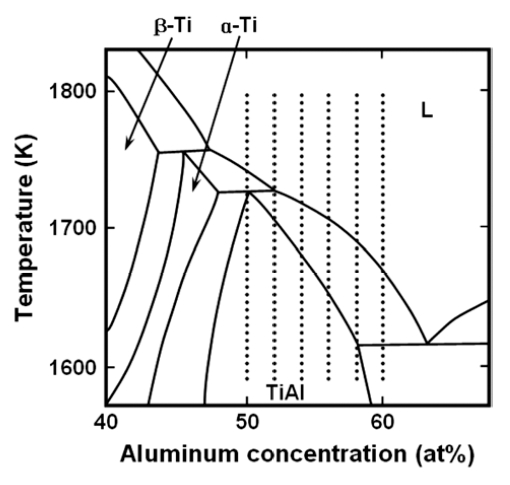
On the Ti-Al phase diagram, plenty of versions are reported as stated above. One of the controversies is on the formation process of α-Ti (titanium solid solution with hexagonal structure). The two different reaction schemes are reported as follows;
or
where, β-Ti is titanium solid solution with bcc structure.
Fig. 4 shows a portion of equilibrium phase diagram quoted by Mishurda et al.11) This phase diagram adopted the peritectic reaction as the scheme (1). In the figure, compositions of the alloys used in the present study are indicated. Present work has shown that the two phase microstructures consisting of α2 and γ were observed at 50 to 54 atomic percent aluminum. It is better understood by the phase diagram in Fig. 4 in which supercooling by rapid solidification would yield the formation of α2 from α formed through the scheme (1). These results would be an indirect evidence for formation process of α-Ti to be of peritectic, although the investigation for the alloys with Ti-rich compositions is necessary for more detailed discussion.
4. Conclusions
In the present study, the following conclusions are drawn through the microstructural observation in rapidly solidified Ti-Al alloys.
1) Quenching rate attained by a hammer anvil method used in the present study is estimated to be as fast as 107 to 108 K/s.
2) Rapidly solidified Ti-Al alloys containing 50 to 54 percent aluminum give two phase structures consisting of γ and α2. The morphology of α2 phase varies with aluminum content.
3) In rapidly solidified alloys containing 56 to 60 percent aluminum, γ single phase structure is observed.
4) From the observations in the present study, the existence of the peritectic reaction as β-Ti + L→α-Ti is suggested.