1. Introduction
2. Experimental Procedures
3. Results and Discussion
3.1. Interfacial compound at the Al-Si-Cu/cast iron junction
3.2. Effects of Sr additions
3.3. EBSD analyses of the interfacial compounds
3.4. Hardness of the interfacial compounds
4. Conclusion
1. Introduction
There is a growing industrial need to achieve a wider range of capabilities by combining dissimilar materials, as the performance of a single material has its limitation. For instance, the combination of aluminum alloy, known for its low density and excellent heat conductivity, with cast iron, which exhibits favorable mechanical properties, can be applied in the manufacturing of engine cylinder blocks. This application, in turn, contributes to the overall weight reduction of automobiles. While there are multiple methods available for compounding different metallic materials, compound casting has gained significant attention due to its remarkable economic efficiency and practicality.1-4)
When performing compound casting of cast iron and aluminum alloy, a common approach involves utilizing cast iron as an insert and pouring molten aluminum onto it. Consequently, the surface of the cast iron insert that comes into contact with the molten aluminum dissolves into the aluminum. As a result of iron being introduced into the aluminum melt, iron compounds are generated at the interface, as well as within the aluminum matrix in close proximity to the interface. Over time, the thickness of the interfacial compound grows proportionally to the duration of contact between the two materials.5-7)
Previous studies have reported that the compounds predominantly formed at the interface between steel and Al-Si alloys include Al5Fe2, Al8Fe2Si, Al4.5FeSi, and others. The specific types and growth behavior of these compounds are influenced by the composition of the aluminum alloy.8-14) The formation of interfacial compounds is essential for producing structurally sound compound castings. However, excessive formation of these hard compounds can lead to brittleness and compromise the overall integrity of the casting. Hence, it is crucial to appropriately control the type and thickness of the interfacial compounds layer. While numerous investigations have been conducted on the compounds formed between steel and aluminum melt, there are relatively few studies on the interfacial compounds between cast iron and aluminum. Moreover, the effect of aluminum alloy composition on these interfacial compounds has not been extensively explored.
The growth of the interfacial compounds is influenced by changes in the composition of the aluminum alloy. For instance, studies have indicated that the addition of elements such as silicon, strontium, and titanium can reduce the growth rate of the compound.15,16) Such information is valuable for effectively managing the type and growth rate of the interfacial compound. Specifically, since small amounts of Sr are commonly incorporated into Al-Si casting alloys to enhance their mechanical properties, it is worth investigating the effects of Sr additions on the characteristics of the interfacial layers. Additionally, the hardness of the compound may be altered by the inclusion of minor alloying elements.17,18) Although the effect may be minimal, lowering the hardness of the interfacial compound is expected to decrease the likelihood of cracking at the interface.
2. Experimental Procedures
The growth of the interfacial compounds is influenced by changes in the composition of the aluminum alloy. For instance, studies have indicated that the addition of elements such as silicon, strontium, and titanium can reduce the growth rate of the compound.15,16) Such information is valuable for effectively managing the type and growth rate of the interfacial compound. Specifically, since small amounts of Sr are commonly incorporated into Al-Si casting alloys to enhance their mechanical properties, it is worth investigating the effects of Sr additions on the characteristics of the interfacial layers. Additionally, the hardness of the compound may be altered by the inclusion of minor alloying elements.17,18) Although the effect may be minimal, lowering the hardness of the interfacial compound is expected to decrease the likelihood of cracking at the interface.
The Al-Si-Cu base alloy melt was prepared using commercial A356 alloy and pure copper (99.9 wt%) ingots in a high-frequency induction furnace. To introduce Sr into the base alloy, an Al-10 wt% Sr master alloy was utilized, with Sr concentrations of 0.02 or 0.2 wt%. The chemical compositions of the cast iron and Al-Si-Cu base alloy are presented in Table 1.
Table 1.
Chemical compositions of cast iron (insert) and Al-Si-Cu (A356 + 0.5 % Cu) alloy (wt%).
| C | Si | Mg | Mn | Cu | Fe | AI | |
| Cast iron | 2.65 | 0.31 | - | 0.15 | - | Balanced | - |
| Al-Si-Cu | - | 6.92 | 0.46 | - | 0.57 | - | Balanced |
The investigation of the interfacial compounds between the cast iron and aluminum alloy melt involved the use of an optical microscope (OM) and a scanning electron microscope (SEM, SU5000, Hitachi, Japan), which was equipped with an energy dispersive X-ray spectrometer (EDS, JEOL, Tokyo, Japan). Additionally, electron backscatter diffraction (EBSD, Hikari Super, TSL, USA) analysis was conducted to determine the types and relative ratios of the interfacial compounds. To evaluate the mechanical properties of the interfacial compounds, nanoindentation tests were performed using a SteP4-NHT3 (Anton Paar TriTec, Switzerland) instrument. The applied load and release rates were set at 100 mN/min, with a pause time of 10 s. Furthermore, micro-Vickers hardness measurements of the intermetallic compounds formed in the Al-Fe-Si specimen were carried out using a MMT-X7 (Matsuzawa, Japan) instrument, applying a load of 50 g.
3. Results and Discussion
3.1. Interfacial compound at the Al-Si-Cu/cast iron junction
When aluminum melt and cast iron come into contact, the iron component is released from the cast iron, entering the liquid and forming AlFe compounds at the junction interface. Since the cast aluminum alloys often include alloying elements such as Si and Cu, leading to a diversified interfacial compound type, including AlFeSi.7,8) In the present study, it was observed that the thickness of the interfacial compound layer slightly increases with prolonged contact time between the Al-Si-Cu base alloy and the cast iron insert. In the specimen with a contact time of zero, the compound forms during the cooling process.
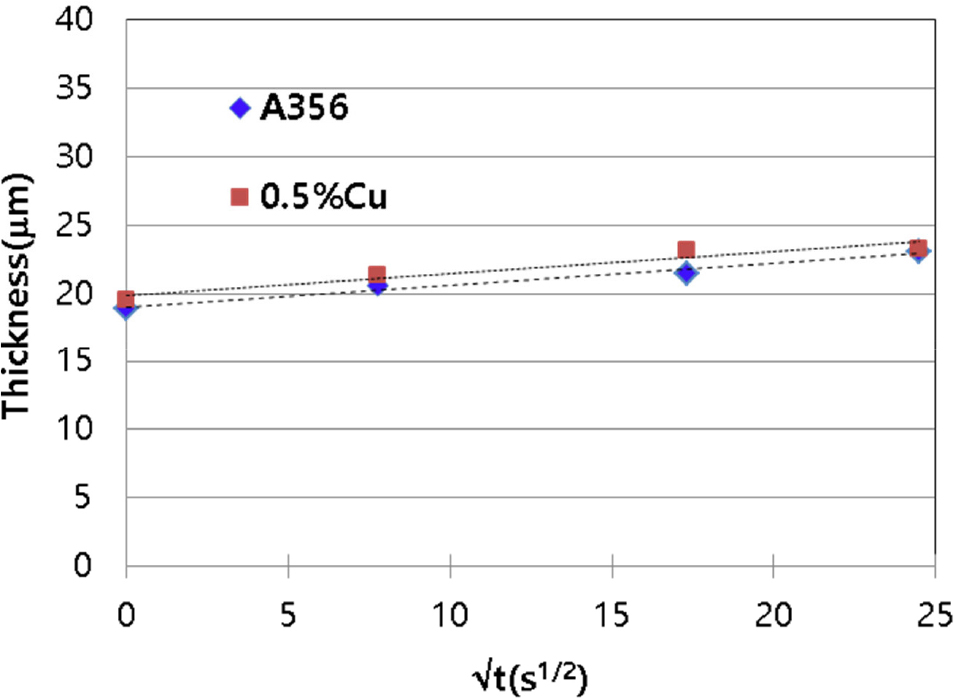
Fig. 1 illustrates that the growth behavior of the Al-Si-Cu base alloy (0.5 wt% Cu-added) is comparable to that of A356, a representative conventional casting alloy in which copper is not added. Although the Cu-added alloy exhibits slightly thicker compounds layer at the same time, it falls within the error range due to slight variations at each interfacial site. Thus, this investigation confirms that the addition of Cu to the Al-Si based alloy does not contribute to reducing the thickness of the interfacial compound layer with cast iron.
Similar to the A356 alloy, the Cu-added alloy exhibited a tendency for the compounds layer to grow proportionally toe the square root of time, and the addition of Cu did not result in any significant change in the growth behavior. The growth of interfacial layer thickness is influenced by a certain degree of dissolution occurring at the interface. Nonetheless, it is believed that the current experimental setup maintains a minimal dissolution rate, with the growth rate predominantly controlled by diffusion.5,7)
3.2. Effects of Sr additions
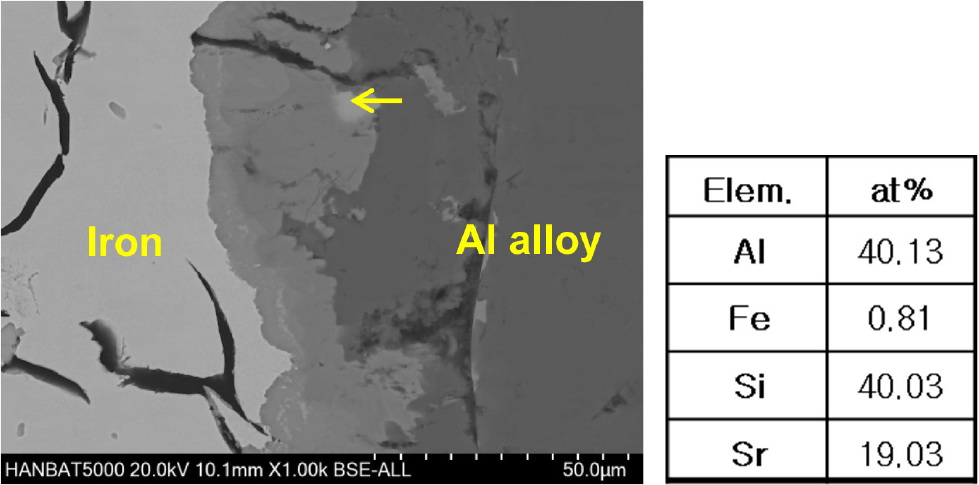
In order to modify the microstructure of Al-Si casting alloys, particularly the eutectic silicon phase, and enhance their mechanical properties, Sr is often added.19) When minor alloying elements are added to the liquid aluminum alloy, it can lead to changes in the characteristics of the interfacial compounds. As shown in Fig. 2, AlFeSi phases are predominantly formed at the interface regardless of the Sr content. However it is evident that there is not a single type of interfacial compound, and a more detailed analysis is needed. Furthermore, it is worth noting that the interfacial layer exhibited a comparatively high concentration of Sr, despite the small amount added. Nevertheless, no notable trend was observed indicating a significant increase in the concentration of segregated Sr within the interfacial layer, even with a further increase in the added Sr content to 0.2 wt%. As shown in Fig. 3, a Sr-rich phase within a portion of the interfacial layer is identified as Al2Si2Sr.20,21)
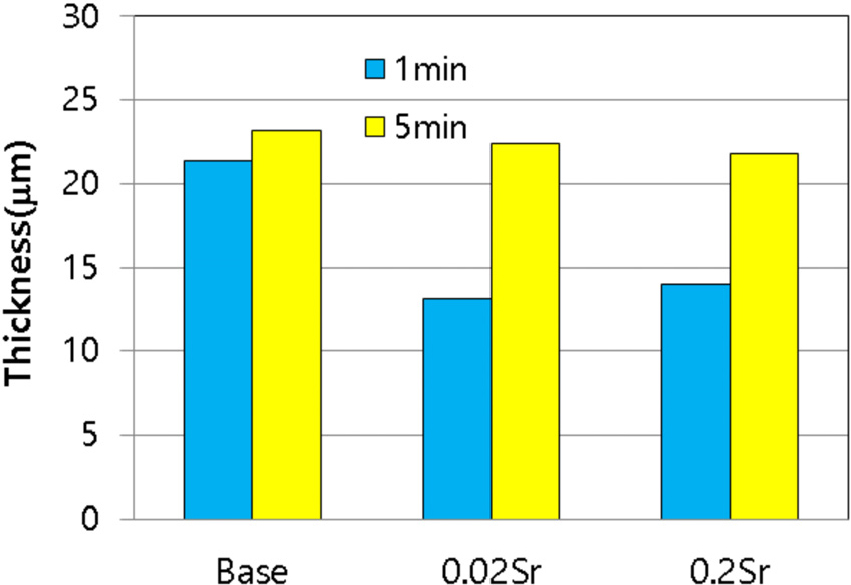
The presence of enriched Sr within the interfacial layer indicates its influence on the growth of the interfacial compounds. Fig. 4 clearly demonstrates that the addition of Sr resulted in a noticeable decrease in the growth rate at a contact time of 1 min. Previous studies have reported that Sr in the aluminum melt reduces the chemical potential of iron atoms, affecting the formation of interfacial compounds.15) Additionally, it is known that Sr additions lead to a reduction in the self-diffusion coefficient of Al and Si atoms in liquid Al-Si alloy.22) However, when a higher amount of Sr (0.2 wt%) was added, no further suppression of interfacial compound growth was observed. Moreover, exceeding a concentration of 0.02 wt% Sr is impractical as it leads to over- modification of the eutectic Si phase, thereby impairing the mechanical properties of aluminum alloy. Therefore, this study focused on the case of adding 0.02 wt% Sr for further investigation.
3.3. EBSD analyses of the interfacial compounds
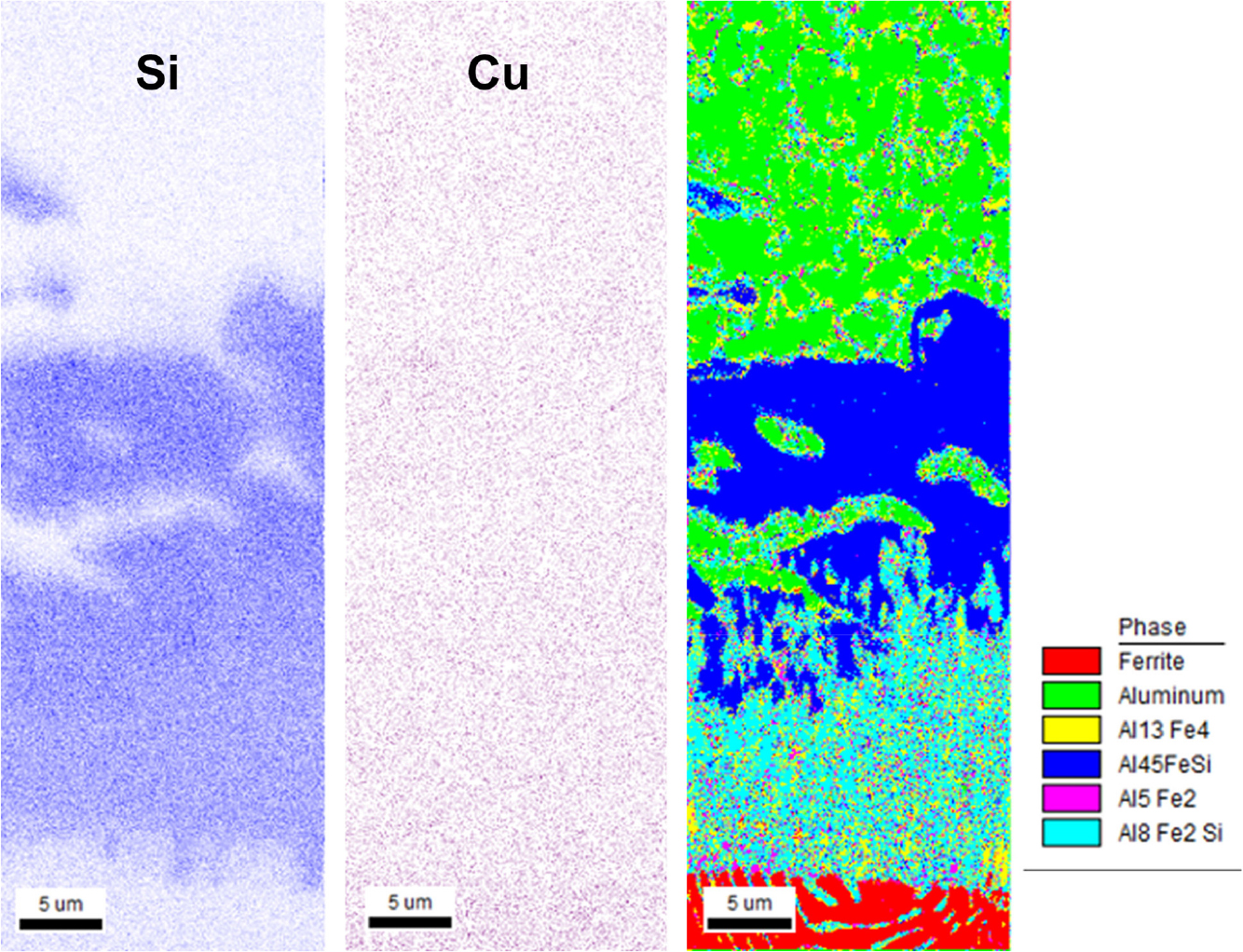
As shown in Fig. 5, various compounds are displayed at the interface between the Al-Si-Cu base alloy and cast iron. The majority of these compounds consist of Al8Fe2Si and Al4.5FeSi phases. Additionally, a small amount of AlFe phases, including Al13Fe4, can be found in the Al8Fe2Si. The characteristics of the interfacial compounds in the Al-Si-Cu alloy are believed to be similar to those found in the A356 alloy, which does not contain copper.7,8) It is also known that specimens with a shorter contact time exhibit fewer AlFe phases.7) In regards to the presence of copper, it does not appear to be significantly concentrated in the interfacial compounds, suggesting that it is unlikely to have a significant impact on compound growth.
When the contact time between the cast iron and the aluminum melt is short, the presence of an adequate amount of Si in the aluminum near the interface facilitates the formation of AlFeSi phases. However, as the contact time extends, the Si concentration in the vicinity of the interface diminishes, leading to a decline in diffusion through the interfacial layer. Consequently, a substantial quantity of AlFe phases is generated. Therefore, when the contact time is relatively short, i.e., 5 min. or less, it is expected that the interfacial compounds layer will mainly consist of AlFeSi phases.
Fig. 6, when 0.02 wt% Sr is introduced, demonstrates that AlFe phases are apparently present at the interface, despite the relatively short contact time of about 5 min. Surprisingly, it was observed that a significant amount of AlFe phases formed at the interface even with a contact time as short as 1 min. As previously mentioned (Fig. 2), EBSD analysis results also indicate a concentration of Sr element within the interfacial layer. The formation of AlFeSi phases requires the diffusion of aluminum, iron and silicon, however, it appears that the addition of Sr delays the diffusion of these elements, particularly silicon, as evidenced by previous studies.15,22) Consequently, this situation creates favorable conditions for the formation of AlFe phases rather than AlFeSi phases. It is worth noting that the dominant compound typically formed at the interface between aluminum alloy with a low silicon content and iron is the Al5Fe2 phase.2)
3.4. Hardness of the interfacial compounds
The intermetallic compounds that form between cast iron and aluminum alloy melt exhibit inherent high hardness and lack of ductility, making them prone to crack formation. In Fig. 7, the nano-indentation hardness of these interfacial compounds is presented, with hardness measurement conducted in two regions: the portion near the cast iron and the portion near the aluminum alloy. Among the interfacial compounds, the region adjacent to the cast iron mainly consists of Al8Fe2Si along with some binary AlFe compounds. On the other hand, the region close to the aluminum alloy is composed of Al4.5FeSi phase. Consequently, it is inferred that the hardness of the Al4.5FeSi phase is relatively lower compared to other intermetallic compounds. This result in hardness can be attributed to the fact that the Al8Fe2Si phase, predominantly found in areas adjacent to the cast iron, exhibits higher hardness than the Al4.5FeSi phase.23)
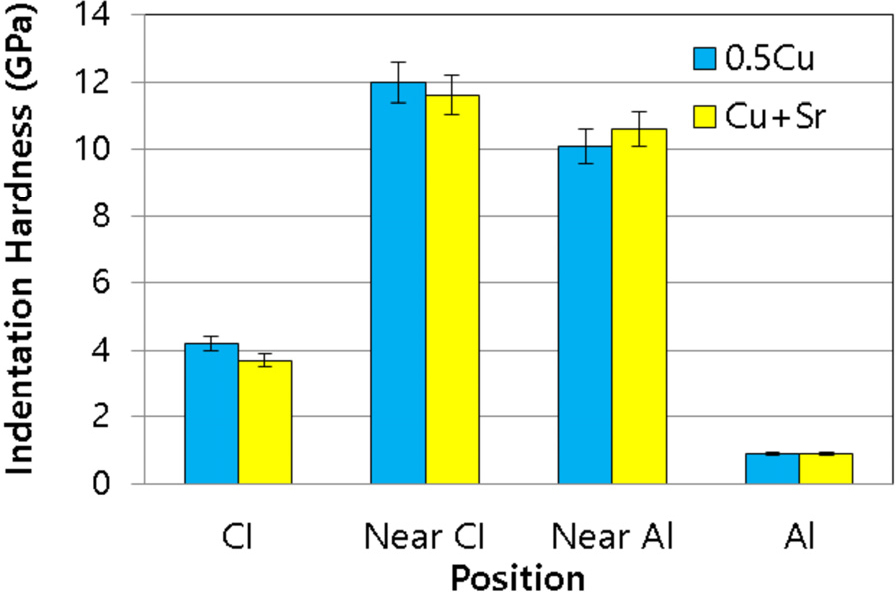
Fig. 7
Nano-indentation hardness of the interfacial compounds formed at the interface between cast iron and liquid Al-Si-Cu alloy with and without 0.02 wt% Sr heated at 720°C for 5min.: CI (cast iron part), near CI (intermetallic layer near cast iron), near Al (intermetallic layer near aluminum), Al (aluminum part).
The experimental results indicate that the addition of Sr does not have a significant effect on the hardness of the interfacial compounds. However, as mentioned earlier, when Sr is introduced, a certain amount of binary AlFe phases coexist with the Al8Fe2Si phase in the interfacial region near the cast iron. Thus, the hardness value of the region may partly reflect the hardness of the binary AlFe phases. Previous studies have reported that the nano-indentation hardness of the Al5Fe2 phase is approximately 8 GPa, which is lower than the hardness of the Al8Fe2Si phase measured in this study.17) Consequently, if the fraction of Al5Fe2 phase in the interfacial region near the cast iron increases, the average hardness of that region is expected to decrease. On the other hand, the hardness of the Al13Fe4 phase appears to be similar to or slightly lower than that of Al5Fe2 phase,24) although some uncertainty remains and further investigation is warranted.
In order to compare the hardness values of the θ-Al13Fe4 and α-Al8Fe2Si phases, an aluminum alloy with high iron and high silicon content (20 at% Fe-10 at% Si) was prepared by vacuum arc remelting. The cast alloy underwent isothermal heating at 590 °C for 1 h, followed by water quenching, to examine micro-hardness.25) As indicated in Fig. 8, the microstructure primarily consists of three regions: light, dark, and very dark areas. Based on SEM-EDS analysis, it is inferred that the light phase corresponds to Al8Fe2Si, while the dark phase corresponds to Al13Fe4. Additionally, the very dark phase with a relatively small area fraction is presumed to be γ-Al3FeSi. Table 2 displays the micro-hardness values of the two main phases: Al8Fe2Si and Al13Fe4. It can be observed that the hardness of the θ-Al13Fe4 phase is apparently lower compared to that of the α-Al8Fe2Si phase. Hence, if the addition of Sr results in the formation of more binary AlFe compounds in the interfacial layer, it is expected that the average hardness of the interfacial compounds near the cast iron would decrease.
Table 2.
Micro-hardness of the two main intermetallic compounds formed in the Al-20 at% Fe-10 at% Si alloy ingot.
| Al13Fe4 | Al8Fe2Si | |
| Hardness (Hv) | 882.6 | 979.5 |
While this study does not encompass experiments on bonding characteristics, it is essential to create an interfacial layer for achieving strong bonding between cast iron and Al-Si alloy. However, the formation of a thick high-hardness interfacial layer would increase the risk of crack formation near the interface. Adding Sr to the Al-Si alloy has the effect of impeding the growth of the compound layer’s thickness and slightly reducing the hardness of the compound layer around cast iron. As a result, it is anticipated that the interfacial bonding stability will be somewhat positively influenced.
4. Conclusion
The interaction between the liquid Al-Si-Cu alloy and cast iron leads to the formation of diverse interfacial compounds, and the thickness of the intermediate layer grows with prolonged contact time. This study focused on the effects of Sr additions on the structure and rate of thickness increase in the interfacial compounds layer. The key findings can be summarized as follows:
(1) At the interface between the liquid Al-Si-Cu base alloy and the solid cast iron, a compound layer primarily consisting of α-Al8Fe2Si and β-Al4.5FeSi phases is formed. The thickness of this layer exhibits a tendency to increase proportionally with the contact time.
(2) When Sr is added, a distinct reduction in the rate of thickness increase of the interfacial layer is observed. However, even with higher amounts of Sr added, there is no observed trend of further decreasing the rate of thickness increase in the layer.
(3) The addition of Sr to the Al-Si-Cu alloy results in its preferential accumulation within the interfacial layer. Consequently, binary AlFe phases, specifically Al5Fe2 and Al13Fe4, exhibit a more pronounced formation in the vicinity of the α-Al8Fe2Si phase.
(4) Through nano-indentation hardness testing conducted on various phases present in the interfacial layer, it was consistently observed that the α-Al8Fe2Si phase showed a higher hardness value compared to the β-Al4.5FeSi phase, regardless of the inclusion of Sr.
(5) Furthermore, it was verified that the α-Al8Fe2Si phase possesses a higher hardness value than the θ-Al13Fe4 phase.