1. Introduction
Photocatalyst material has a high usability property on the field of environmental protection owing to their performance. On top of that, the unique structured photocatalyst highly recommended for the degradation and adsorption of dye, biological degradation studies.1-3) In recent decades the removal of -organic and inorganic substances over the semiconductorbased photocatalysts in a liquid phase has attracted extensive attention.4) For semiconductor-based photocatalyst, the photon energy is a high potential factor to activate the photocatalyst and improve the quantum efficiency. Considering the above process in detail, the photon energy should be greater or equal to the bandgap energy of the photocatalyst thus to produce the electron-hole pairs. The photoexcited electron on CB and the hole on VB has a role play to oxidize the reductant substance to small molecule matter. However, the high recombination rates of electron and hole reduced the activity of the photocatalyst, further scale-down the quantum efficiency.5) Generally, the best form of the photocatalyst is free radicals which can react with the water or adsorbed reductant substances; however, the free charge tends to react with the water more than reductant substances. Because the water oxidation create the hydroxyl radicals and the produced hydroxyl radicals degrade the reductant dye on the surface of photocatalyst as well as reductant dye in the solution.6) Semiconductor metal oxides such as TiO2, ZnO, ZnS, CuS, and NiO have widely been studied and utilized in photocatalysis. TiO2 nanostructures are wellestablished materials for photocatalytic dye degradation due to their photo-catalytically stable nature, ease of production at low cost, and less risk to environment.7-10) Among these semiconductor metal oxides, nickel oxide is a p-type semicon- ductor with a stable, low-cost, broad band gap and is nontoxic. Likewise, it has excellent optical, electrical, and photocatalytic properties making it a promising material for applications in photocatalysis.11-13)
Several binary semiconductors including CNT/TiO2,14) EG-TiO2,15) ACF/TiO2,16) and ZnS/TiO217) have been used for MB decomposition. The junction of the two semiconductors leads to an effective light response with efficient charge carriers and tunable bandgap energy, thus leading to high catalytic activity nanocomposites. The degradation amounts over those photocatalysts were reached to almost 100 %. Light source and catalysts are two important factors during heterogeneous photocatalysis. TiO2-Based photocatalysis involves irradiation of UV light to enhance the degradation process. Beside the metal-oxide semiconductor, β-SiC fiber has created a lot of attention due to its excellent properties, such as suitable band gap, high thermal conductivity, high mechanical strength and high chemical stability.18-20) β-SiC has strong capacity of reduction oxidation under visible light irradiation due to suitable band gap and redox energy level of its corresponding valence and conduction band. Based on the phenomena, we fabricated the NiO-SiC-TiO2 ternary structure nanocomposite and used it for methylene blue degradation process under the UV-light irradiation. In addition, it has been found that the change of surface of the photocatalyst, the morphology profile with the structure greatly affected to the MB degradation. The close contact the 2D material and semiconducting metal oxides expanded the scope of absorption wavelength and longitude the stability of the photocatalyst. Thus, the unique structure changes offered the high potential for degradation of organic pollutants.
2. Synthesis and Experiment Setup
2.1. Materials
Nickel (II) acetate tetrahydrate (<98.0 %, C4H6NiO4), Titanium (IV) n-butoxide, (<98.0 %, C16H36O4Ti), β-SiC fiber (≥98.0 %) were purchased from Pure Chemical Co. Ltd, and Deionized water was used for the synthesis procedure. Methylene blue trihydrate (C16H24ClN3O3S) was used for the photocatalytic experiment.
2.2. Synthesis of NiO-SiC
1 g of β-SiC and 2.48 g of Ni precursor was dissolved in 100 ml deionized water under continuous magnetic stirring process. After 3 h of stirring process, the mixture was transferred into electric oven and kept under 100 °C for 12 h. The final powder was calcined under 600 °C for 3 h in order to remove the residual substances and improve the crystallinity of the final product. The obtained sample was renamed as a NiO-β-SiC.
2.3. Synthesis of TiO2-SiC
1 g of β-SiC and 3.41 ml of Ti precursor was dissolved in 100 ml deionized water under continuous magnetic stirring process. After 3 h of stirring process, the mixture was transferred into electric oven and kept under 100 °C for 12 h. The final powder was calcined under 600 °C for 3 h in order to remove the residual substances and improve the crystallinity of the final product. The obtained sample was renamed as a NiO-SiC. The ternary NiO-SiC-TiO2 nanocomposite was prepared by following the same procedure as-above mentioned.
2.4. Characterization
The phases of samples were examined using X-ray diffraction measurements (SHIMADZU XRD-6000) equipped with a Cu Ka X-ray source (1.5406 Å). The chemical composition was analyzed using energy dispersive X-ray (EDX) analysis incorporated into the SEM. Transmission electron microscopy (TEM, Hitachi HT7700, operated at 100 kV) was used to analyze the morphologies of the samples. Highresolution transmission electron microscopic (HRTEM) images of the samples were obtained using a Hitachi H9500 operated at 300 kV. UV-vis diffuse reflectance spectra (DRS) analysis was performed using ultraviolet-visible (UV-vis) spectrophotometer (SHIMADZU UV-2600) ranging (200 to 800) nm. The bandgap energies of the photocatalysts were calculated through application of a modified Kubelka-Munk function obtained from the UV-vis DRS data. The resistivity of the samples was examined by EIS (electrochemical impedance spectra) using an electrochemical analyzer (Zahner, Germany). The photocurrent value was tested using a SLS 301-stabilized benchtop Tungsten-Halogen light source with a 150 W Halogen bulb. The wavelength range is 360~2,700 nm. Output optical power is >1.6 W.
2.5. The experiment setup for the MB degradation
0.00184 g of methylene blue was dissolved in 100 ml deionized water and stirred for 2 h. The test of degradation was carried out in a glass reactor with magnetic stirrer under the ambient temperature and pressure environs. The UV light has been chosen as a light source for the degradation of the MB experiment. The distance between the light source and the glass reactor was controlled at 6 cm. The total time of the experiment was 4 h, and every 1 h, 10 ml of liquid sample was withdrawn from the glass reactor. Removing the photocatalyst from the withdrawn liquid is very important to examine the real absorbance of the liquid sample. A centrifugation process was carried out to remove particles from the test solution. The concentration of the acquired solution was analyzed by UV-spectrophotometry (Optizen POP, Korea) for use in a quartz cell (1 cm × 4.5 cm) and scanned from (200 to 950) nm wavelength region.
3. Result and Discussion
3.1. XRD, SEM, TEM and EDX analysis
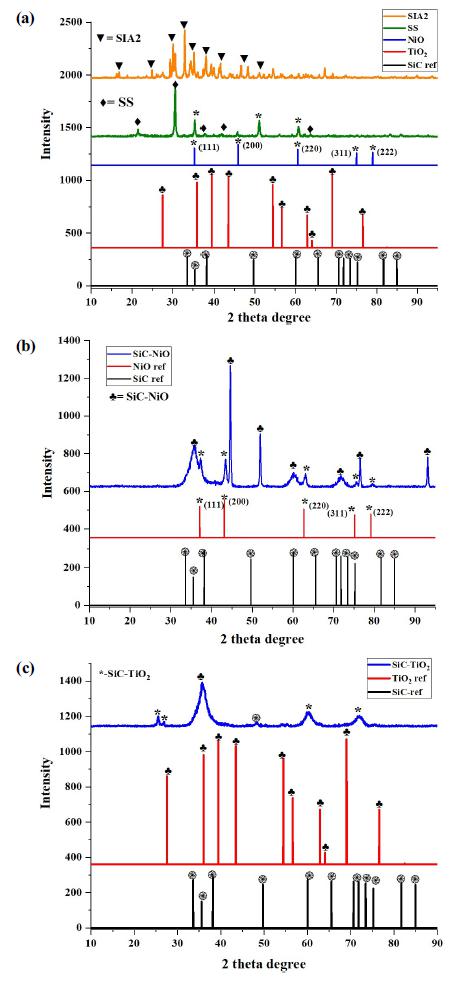
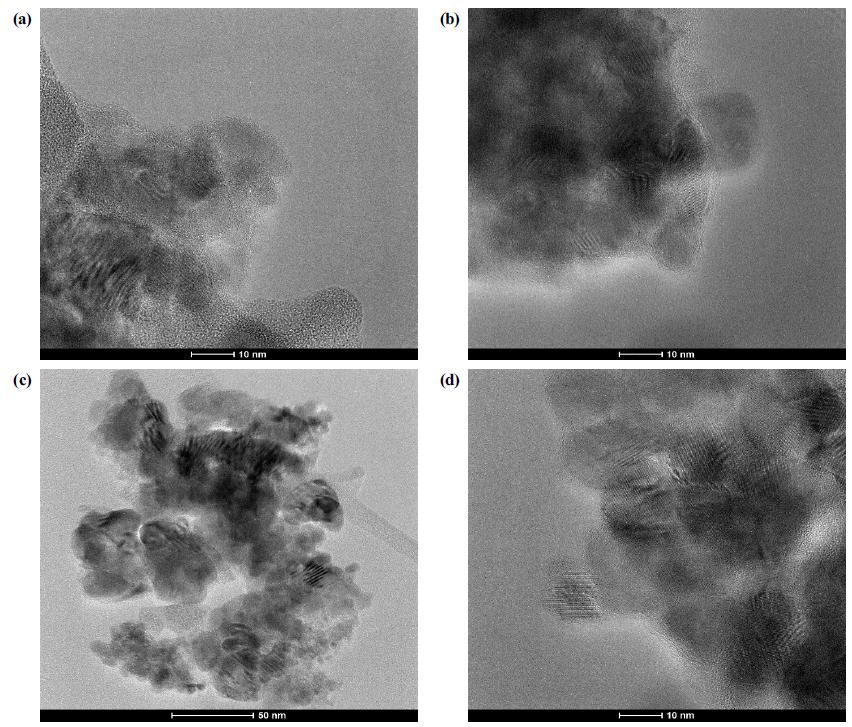
The crystal structures of the nanocomposites were analyzed by XRD. As expressed in Fig. 1, the intensity and sharpness of the XRD peaks indicated that the pure semiconductor and the nanocomposite materials were successfully synthesized with high crystallinity. The characteristic peaks of TiO2, SiC fiber and NiO were well found and indexed in Fig. 1. The scanned profile of the sample with a high-energy beam of electrons were expressed by the SEM, and the images were summarized in Fig. 2. The SEM images of NiO-SiC were expressed in Fig. 2(a). The bustled-irregular shaped NiO has been found in the SEM images of NiO-SiC [in Fig. 2(a)], thus this morphology profile still found in the SEM image of NiO-SiC-TiO2. The NiO is well-coated on the surface of the SiC fiber because the SiC fiber has been offered the abundant and lustrous surface area where MO can be located fiber TiO2-SiC nanocomposites. The TiO2 had a big particle with a dissimilar size, and the TiO2 NPs were well-spread. The ternary NiO-SiC-TiO2 nanocomposites had a clear SEM profile as shown in Fig. 2(c). The spherical shaped NiO and TiO2 were well-distributed on the abundant surface of SiC fiber. The SiC fiber had a smooth surface with a width of 11.3 nm, and all physical properties of the SiC fiber offered the best location for the metal oxide semiconductors to fabricate the best SEM profiles. The elemental analysis is the important analysis to examine the impurity of the final nanocomposites. The datas were summarized and attached in Fig. 3. The main elements were well-detected, however, only tungsten element was found from the EDX analysis, it may be derived from the laboratory instruments that used for synthesis of NiO-SiCTiO2 ternary nanocomposite. The high magnified images with the internal structure of the samples were detected by TEM, and the images were expressed in Fig. 4. It has been found that the NiO and TiO2 metal-oxide semiconductor were welllocated on the SiC-fiber to form the ternary structured nanocomposites. Fig. 4(a-c) expressed the TEM images of the NiO-SiC, TiO2-SiC, and NiO-SiC-TiO2 and Fig. 4(d) expressed the HRTEM images of ternary NiO-SiC-TiO2 in order to evaluate the crystal structure and lattice imperfections.
3.2. Electrochemical analysis
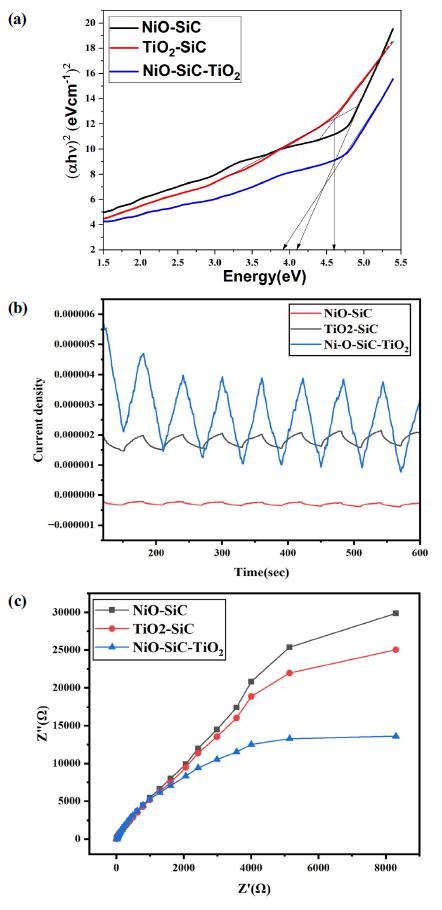
The charge carrier and resistivity analysis are the essential examination for the photocatalysts material. The UV-Vis DRS, EIS and Photocurrent response analysis were conducted to evaluate the charge carrier of the NiO-SiC, TiO2-SiC, and NiO-SiC-TiO2 nanocomposites. As expressed in Fig. 5(a), the lowest bandgap energy value obtained on NiO-SiC-TiO2, the value is 2.81 eV. The highest bandgap energy obtained on TiO2-SiC which is 3.72 eV; this value may be derived from the combination of two high bandgap energy semiconductors. The TiO2 nominated as a high bandgap energy metal-oxide semiconductor which has and 3.20 eV, and the SiC fiber had a 3.26 eV. The NiO-SiC had a 3.17 eV. In conclude from the evaluated bandgap energy, the junction of TiO2, NiO and SiC fiber offered the greater band adjustment to reduce the bandgap energy and expanded the absorption wavelength of light. Fig. 5(b) expressed the photocurrent response of the nanocomposites that coated on the FTO-glass. Under 7-times light on and off cycles, the ternary nanocomposites showed the greater response to the light. The lowest photocurrent response obtained on NiO-SiC, although it has a lower bandgap energy than TiO2-SiC; it may be due to the surface profile of the nanocomposite. The EIS Nyquist analysis was further conducted in order to confirm the correctness of the Transient photocurrent responses of the nanocomposites. The semicircle profile and size are the main factor that expressed the resistance between the electrode and electrolyte. The smallest semicircle profile obtained on NiO-SiC-TiO2 nanocomposites that indicated the low charge-transfer resistance with the better charge carrier and separation process. Besides, the highest semicircle profile obtained on NiO-SiC, and the EIS data was well-matched with the Transient photocurrent response of it. In conclude from the electrochemical analysis, the introduction of TiO2 and NiO on 2D SiC fiber created the better effective separation of charges and created the enhanced catalytic activity for the degradation of methylene blue over the ternary nanocomposites.
3.3. Degradation of Methylene blue
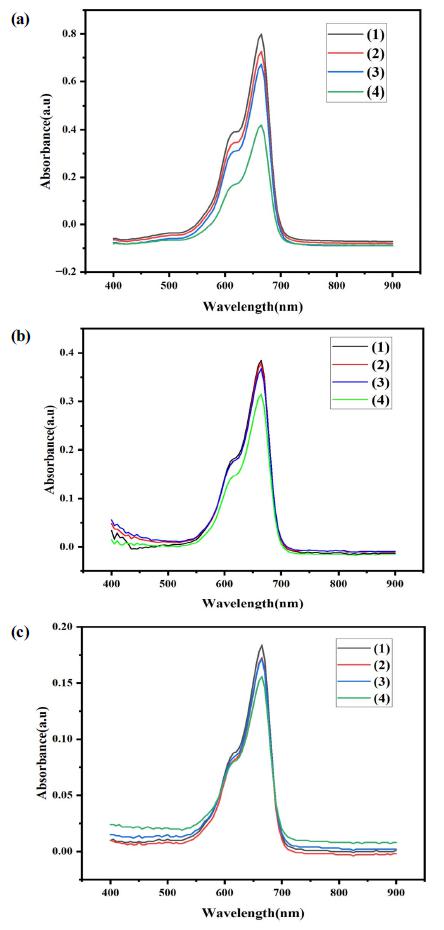
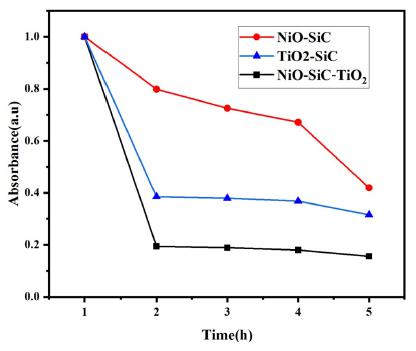
The photocatalytic activity of the nanocomposites examined by the degradation of Methylene blue under the UV-light irradiation. The absorbance spectrum of the photodegradation of MB-dissolved solution were expressed in Fig. 6. The experiment conducted under 4 h, and in every 1 h, the liquid sample was withdrawn from the glass reactor and analyzed by using the UV-spectrometer. It is obvious that the absorbance peak of Methylene blue was located at 664 nm wavelength region. After 4 h of light irradiation, the absorbance intensity was significantly reduced which is indicating that all nanocomposites were effectively degraded the methylene blue under the light irradiation. Especially, the ternary structured NiO-SiC-TiO2, and it is indicating that our proposed ternary nanocomposite upgraded the type of nanocomposite by one for the Methylene blue degradation. Fig. 6(a) expressed the UV-vis spectrum of degraded MB over the NiO-SiC photocatalyst. It has been found that the MB effectively degraded on NiO-SiC. About the case of TiO2-SiC, the degradation of MB was quite good as NiO-SiC. It is confirmed that the effective charger separation and carrier process of electron and holes are potential factors that can define the catalytic activity of the nanocomposites. However, the quite effective degradation process obtained on NiO-SiC due to the bandgap energy value. The better response to the light can be related to the bandgap energy of the photocatalyst, thus the lower bandgap energy obtained on NiO-SiC.
Other research studies proposed the ZnO/SiC composites,21) TiO2/GO22) and TiO2 impregnated ceramic material23) for the MB degradations under the light irradiation. The maximum degradation rates of MB over ZnO/SiC was 95.7 %, pure NiO degraded the MB over 90 % and TiO2 impregnated ceramic material was degraded to 60~70 % under the different intensity light irradiation. Our proposed ternary NiO-SiC-TiO2 nanocomposite degraded the MB over 93.0 % after the 4 h of UV-light irradiation. Other two binary NiO-SiC and TiO2-SiC nanocomposites even highly degraded the MB under same experiment condition, the degradation rates were between 75.0~90.0 %. In conclusion from the published papers and the Methylene blue degradation over ternary NiO-SiC-TiO2, the introduction of 2D material offered abundant reactive sites where the reductant substance can be adsorbed, moreover, the loading of metal oxides semiconductor offered the approach to adjust the bandgap energy, thus enhanced the charge carrier and separation mechanism, and created the higher catalytic performance. The final ternary nanocomposite exhibited the high catalytic activity with a high quantum efficiency. The strong MB degradation over NiO-SiC-TiO2 started to be obtained after 1 h of UV-light irradiation. It means that the irradiated UV-light activated the ternary photocatalyst thus leading to offer the large number of photoexcited electron and holes. In addition, the large number of holes were participated in the oxidation of Methylene blue and enhanced the catalytic activity. Besides, the holes produced from the photoexcitation favourable reacted with the water, thus form the hydroxyl radicals, and those radicals promoted the decomposition of MB.24-27) Fig. 7 expressed the comparison results of the three different photocatalysts. As mentioned earlier, the ternary structured photocatalyst had a better response for the Methylene blue degradation under the UV-light.
4. Conclusion
In summary, the junction of Ti2O, NiO and the 2D SiC fiber allowed the adjust the bandgap energy with the enhanced charge carrier and separation process. Thus, the final ternary nanocomposite has an excellent degradation performance for the Methylene blue. In addition, it has been found that the change of surface of the photocatalyst, the morphology profile with the structure greatly affected to the MB degradation. The close contact the 2D material and semiconducting metal oxides expanded the scope of absorption wavelength and longitude the stability of the photocatalyst. Thus, the unique structure changes offered the high potential for degradation of organic pollutants.