1. Introduction
The rapid growth of the global population is leading to an increased demand for implants as biomaterials for the repair, reconstruction and replacements of damaged bone caused by diseases and accidents.1) Ceramics used for implants as a biomaterial are termed “bio-ceramics”.2) Among bio-ceramics, hydroxyapatite(HA) has been widely used as bone substitutes due to superior biocompatibility with the bone. However, when HA is used as bone substitutes solely, HA restrains the recovery of defected bone because it remains in the body during recovery of the bone due to low bio-degradable rate. But β-TCP is a one of calcium phosphates and known as high biodegradable materials in the body like magnesium. Therefore, a lot of researchers have been tried to fabricate BCP(HA + β-TCP; tri calcium phosphate) instead of HA or β-TCP cement.3-4) On the other hand, porous-structure of biomaterials is also an important factor. Because, circulation of body fluids into porous-structure is very important factor to restore the bone.5) There are a lot of methods to fabricate the porous structure.6-10) Among them, NH4HCO3 as space holder(SH) was used in this study as a volatilization method. Volatilization of SH is a simple to make porous structures by sintering.
Thus, porous structured BCP biomaterials were fabricated using SH (space holder; NH4HCO3, TiH2) by SPS (spark plasma sintering) to evaluate feasibility assessment as bone scaffolds.
2. Experimental Procedure
The HA and β-TCP powders were mixed 80 % to 20 % ratio (wt%) and the NH4HCO3 (10, 20, 30 wt%) as space holder(SH) mixed with HA/β-TCP(BCP) powders. And TiH2 as a foaming agent(FA) and MgO as a binder were added in the mixed powders at 2 and 1 wt%, respectively as listed in Table 1. And prepared powders were mixed for 24h by mechanical mixer(ABB ACS100). The mixed powders were placed in a cylindrical graphite die (outside diameter: 35 mm, inside diameter: 10.5 mm, height: 40 mm) of the SPS equipment(Spark plasma sintering, Sumitomo). The chamber was evacuated 10−2 torr and a uniaxial pressure of 20MPa was applied. The powders were sintered by two steps at 393K for 5 minutes and heated up to 1373K for 5 minutes and instantly cooled in chamber. The pores distribution and morphology of samples were observed by field emission scanning electron microscope(FE-SEM: JMS-6400, Carl Zeiss), micro-focus X-ray device(SMX-225 CT, SHIMADZU). The phase analysis was conducted by X-ray diffraction (XRD, Rigaku).
Table 1.
The designed composition of samples(wt%).
| Sample | HA | β-TCP | NH4HCO3 | TiH2 | MgO |
|---|---|---|---|---|---|
| A | 79.2 | 19.8 | X | X | 1 |
| B | 69.6 | 17.4 | 10 | 2 | 1 |
| C | 61.6 | 15.4 | 20 | 2 | 1 |
| D | 53.6 | 13.4 | 30 | 2 | 1 |
Hank’s balanced salt solution(HBSS) was prepared for immersion test. The sintered samples were soaked in HBSS at 37 °C for 1, 7, 15 and 30 days and the solutions were refreshed daily. A SEM equipped with an energydispersive X-ray spectrometer(EDS) was used to evaluate not only the surface morphologies of the samples after immersion in HBSS but also formation of apatite.
MTT assay was conducted using 5 × 104 HOB(Human osteoblast) cells. After a period of 72h in incubator under 5%CO2 atmosphere, specimens were immersed into tetrazolium-based colorimetric(MTT) solution for 4h and then the specimens were read by ELISA(Enzume-Linked Immuno Sorbent Assay) leader. Cell attachments test was conducted using human osteoblast(hFOB) cells were cultured in Dulbecco’s modified Eagle’s medium (DMEM) enriched with 10 % FBS (Gibco-Invitrogen, Carlsbad, CA. USA). 1 % penicillin and streptomycin (Sigma-Aldrich, St. Louis, MO, USA) in a 5 % CO2 humidified incubator at 34 °C Cells were maintained as a monolayer (80-90 % confluency) and used from sixth to ninth passage. The cells were seeded in 24 well plate at a density of 5 × 103 cells/well and then incubated for 24h. After incubation, cells were fixed with cold acetone for 2min and stained by DAPI(diamidino-2-phenylindole) staining method; which is one of fluorescent staining methods.
3. Results and Discussion
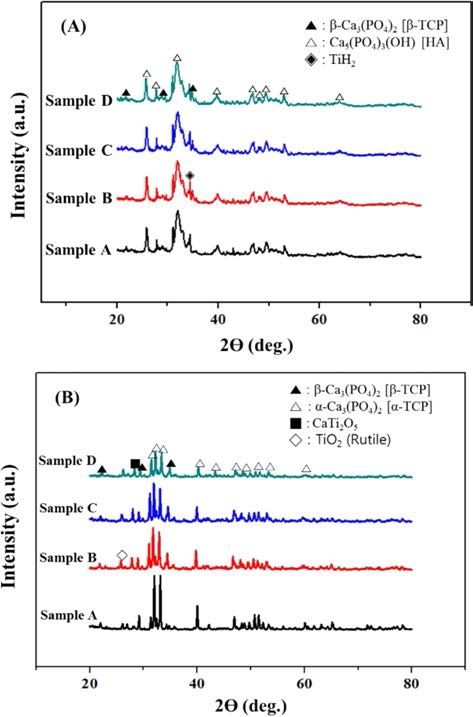
As a result of phase analysis(Fig. 1), a crystalline sintered BCP samples can be obtained through sintering and there are new phases formed by sintering such as α-Ca3(PO4)2 [TCP], CaTi2O5, TiO2 due to interaction between HA and TiH2 during sintering.11-12)
As shown in the CT images(Fig. 2), pore distribution and mean elongated pore size increased(466-525 μm; Table 2) with increasing SH contents and elongated pores was observed. Elongated pores were caused by uniaxial pressure in a SPS furnace during sintering. By uniaxial pressure, pores were easily connected with near pores.

Fig. 2.
3D-CT images of HA/β-TCP biomaterials: (a) Sample A; (b) Sample B; (c) Sample C; (d) Sample D.
Table 2.
The mean elongated pore size of HA/β-TCP biomaterials.
| Sample | Mean Pore Size(μm) |
|---|---|
| A | 2 |
| B | 466 |
| C | 492 |
| D | 525 |
The pores on surface of the sintered BCP samples increased with increasing SH contents and large pores were successfully fabricated(Figs. 3, 4) by floating H2 gas on the surface from TiH2.12) Rough surfaces promote greater amounts of protein adsorption and efficiently stimulate more new bone formation than conventional surface due to their high specific surface area.13)

Fig. 3.
The surface images of HA/β-TCP biomaterials: (a) Sample A; (b) Sample B; (c) Sample C; (d) Sample D.
After immersion test in HBSS, the new particles were formed after 7 days and covered on the surface as a layer after 30 days(Fig. 5). By EDS analysis of this layer(Table 3), oxygen, phosphorous and calcium are detected as main elements of layer. Those elements are main elements of apatite layer [Ca5(PO4)3(OH, F, Cl)]. In immersion test of sample D(SH added samples), apatite layer was also formed(Fig. 6). As a result of ICP analysis(Table 4), calcium and phosphorous ions decreased after immersion test. It indicated that this apatite layer was formed by ion exchange between BCP samples and HBSS. The formed apatite layer has been reported to increase the osteo-conductivity by promoting the adsorption of the osteoblasts. 14-15) Thus, BCP samples fabricated in this study indicated good biological properties in HBSS.
Table 3.
The EDS result of sample A after immersion test for 30 days.
| Element | Wt% | At% |
|---|---|---|
| O | 68.95 | 82.53 |
| Na | 0.18 | 0.15 |
| Mg | 2.19 | 1.72 |
| P | 13.48 | 8.34 |
| K | 0.25 | 0.12 |
| Ca | 14.95 | 7.14 |
| Total | 100 | 100 |
Table 4.
The ICP analysis of HBSS before immersion(B.I.) and after immersion(A.I.).
| Sample | Ca(ppm) | P(ppm) | ||
|---|---|---|---|---|
| B.I. | A.I. | B.I. | A.I. | |
| HBSS(after 30 days) | 25.132 | 9.109 | 11.786 | 3.610 |
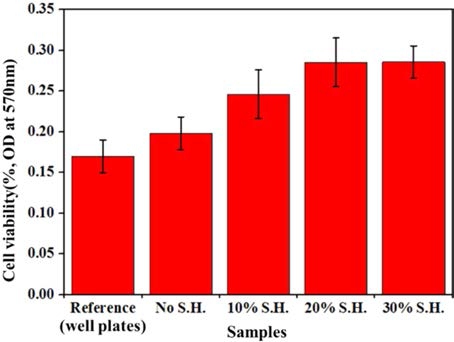
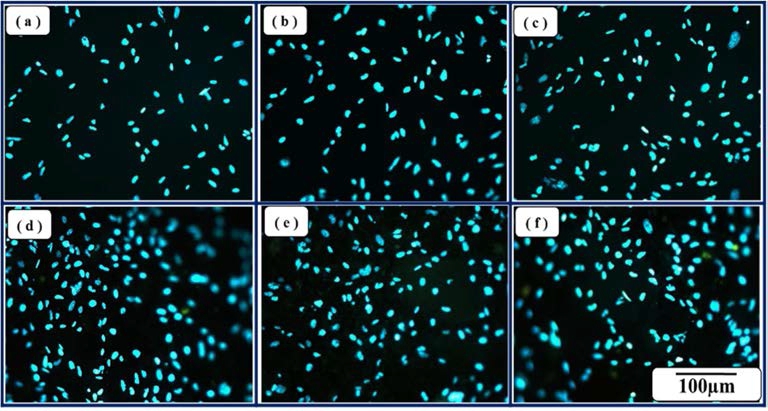
The cell viabilities of the porous BCP samples and reference sample are shown(Fig. 7). Cell viabilities of BCP samples were improved with increasing SH contents. It causes high cell attachments of materials because of specific surface area increased by adding SH(Fig. 8). Therefore, cell viabilities of the porous BCP biomaterials increased by increasing SH contents.
4. Conclusions
From the experimental results, we summarized as follows;
The porous HA/β-TCP biomaterials with the macropore size of 466-525 μm were successfully fabricated by adding NH4HCO3 as space holder, MgO as binder and TiH2 as foaming agent by using rapid sintering of SPS.
The HA/β-TCP(BCP) biomaterials fabricated by adding TiH2 have rough surface of pores.
Bio-activity of HA/β-TCP biomaterials had excellent properties in HBSS.
Cell viabilities of BCP biomaterials were improved with increasing SH content.