1. Introduction
2. Experimental Procedure
2.1. Materials and instrumentation
2.2. Experimental
3. Results and Discussion
4. Conclusion
1. Introduction
Living cells play crucial roles in tissue engineering, cell therapy, transplantation, and biosensing. However, they are highly sensitive to external conditions such as temperature, ultraviolet radiation, moisture, and pH, which can damage cell membranes and cause leakage of cellular components, ultimately leading to cell death.1,2) To preserve viability, cells are typically stored at sub-zero temperatures using dry ice or liquid nitrogen, but this often results in damage due to surface ice crystal formation during thawing.3,4) Therefore, effective strategies are needed to maintain cells in a dormant state and protect them from damage during recovery.4,5)
To address this, various encapsulation approaches have been developed to coat living cells with protective materials. These include polymers, biomolecules, inorganic materials, and metal-organic frameworks (MOFs), which form an artificial exoskeleton around the cell surface.6) Polymer coatings can introduce new protective functions and serve as substrates for nanostructures.7) Biomolecule-based coatings enhance biocompatibility and can confer biological activity or targeting capabilities.8,9) Inorganic nanoparticles add specific mineral functions, while MOFs offer precise structural control and multifunctional properties suitable for cell encapsulation.2,10,11)
MOFs are hybrid materials composed of metal ions and organic linkers, offering tunable structures, high surface areas, and controllable pore sizes.12,13,14) These properties enable their use in a wide range of applications such as drug delivery, sensing, gas storage, and cytoprotection.13) For cytoprotective purposes, MOF shells are often synthesized directly around cells through a one-pot method.15,16) This artificial exoskeleton effectively shields cells from external stress, toxins, and radiation.17,18,19)
Yeast cells are particularly suitable for such applications due to their rigid cell walls, which are composed of β-glucan, chitin, and glycoproteins. These features allow them to carry both hydrophilic and hydrophobic compounds.20,21,22) However, their limited environmental resistance necessitates encapsulation to improve stability and function.23)
Among various MOFs, zeolitic imidazolate framework-8 (ZIF-8) is one of the most widely studied for cytoprotection and drug loading. It can be synthesized under mild conditions, and its zinc-imidazole coordination bonds can be selectively cleaved under mildly acidic conditions or in the presence of chelating agents.24,25,26) Due to its low cytotoxicity and good compatibility with biological systems, ZIF-8 forms a detachable crystalline shell on cell surfaces, providing protection while allowing for controlled removal.2,26,27) Furthermore, ZIF-8-based encapsulation has been successfully applied to various types of cells beyond yeast, including bacterial, probiotic, and mammalian cells. In these studies, ZIF-8 coatings were shown to enhance cell resistance to oxidative stress, protect against harsh gastrointestinal conditions, and modulate immune responses in therapeutic applications. These findings collectively demonstrate the versatility and effectiveness of MOF-based encapsulation systems in diverse biological environments.2,15,23)
In this study, yeast cells were encapsulated with ZIF-8 using a self-assembly process. The encapsulated cells were then subjected to pH stress to evaluate their stability. Morphological analysis was performed using field emission-scanning electron microscopy (FE-SEM) and field emission-transmission electron microscopy (FE-TEM). Physicochemical properties were characterized using thermogravimetric analysis (TGA), energy-dispersive X-ray spectroscopy (EDS), X-ray diffractometer (XRD), dynamic light scattering (DLS), and Zeta potential measurements. Cell viability was subsequently assessed using confocal laser scanning microscope (CLSM).
2. Experimental Procedure
2.1. Materials and instrumentation
Zinc acetate dihydrate (98.0 %), methyl alcohol (99.5 %), acetone (99.5 %), and buffer solution (pH 2, 4, 6, 8, 10, and 12) were purchased from Samchun Pure Chemical (Korea). 2-Methylimidazole (99 %) and fluorescein diacetate (FDA, used as a stain for assessing cell viability) were purchased from Sigma-Aldrich (USA). Nitrogen gas (N2, 99.95% min, N35) was supplied by a commercial vendor. Saccharomyces cerevisiae (baker’s yeast) was used in this study. Yeast extract-peptone-dextrose (YPD) broth was obtained from KisanBio (Korea). Deionized (DI) water was prepared using a ROMAX HIQⅡ system (ROMAX, UK). A syringe pump (LEGATO 100, KD Scientific Inc., USA) and a vacuum pump (2034C-02, WELCH, Germany) were used during the experimental procedures. Also, shaking incubator (SI-600R, JeioTech, Korea), centrifugal separator (Centrifuge 5415D, Eppendorf, Germany), vacuum plasma system (CUTE, Femto Science, Korea), and freeze dryer (MCFD, ilShinBioBase, Korea). Morphological analysis was conducted using FE-SEM (Hitachi, S-4800, Japan) and FE-TEM (200 kV, JEOL LTD, Japan). Physicochemical properties were characterized using TGA (TGA N-1000, Scinco, Korea), zeta-potential (Zetasizer Nano ZS, Malvern, UK), EDS (JSM-7610F Plus, JEOL LTD, Japan), and XRD (Empyrean, Panalytical, Netherlands). Yeast cell viability was confirmed by CLSM (LSM 700, Carl Zeiss, Germany).
2.2. Experimental
2.2.1. Formation of zeolitic imidazolate framework shell on yeast cells
2 mg yeast powder was cultured in YPD broth (30 h, 180 rpm, 33 °C). The yeast cells were washed with DI water three times.2,23) The washed yeast cells were then suspended in 5 mL of an aqueous solution of 2-methylimidazole (1,600 mM). 5 mL aqueous solution of zinc acetate dihydrate (20 mM) was then added into the 2-methylimidazole solution containing the yeast cells. The mixture was placed on a shaking stage (300 rpm) for 10 min for the formation of ZIF-8 coatings at room temperature (RT). The coated cells were washed with DI water three times to remove the excess ZIF-8 precursors, and finally suspended in DI water.2,16)
2.2.2. Encapsulated yeast cells under various pH conditions
The encapsulated cells were dispersed in DI water. Cells were immersed in buffer solution (pH 2, 4, 6, 8, 10, and 12) at a concentration of 100 µL/mL. Incubation time was 1 h. After incubation, the cells were washed with DI water by centrifugation at 6,000 rpm for 1 min. The resulting pellet was resuspended in DI water and used for subsequent analyses.
2.2.3. Cell viability test
Cell viability was investigated by CLSM, after adding 5 µL of an FDA stock solution (10 mg/mL) yeast suspension (1 mL in DI water) and incubating the mixture for 15 min at 33 °C. The cells were then washed three times with DI water to remove free dyes in the solution.23)
3. Results and Discussion
In this study, individual yeast cells were encapsulated with a ZIF-8 nanoshell [Fig. 1(a, b)], and their cell protection ability was evaluated in various pH environments.
Fig. 2 presents the morphological analysis conducted using FE-SEM and FE-TEM. FE-SEM images [Fig. 2(a, b)] show the morphology of unencapsulated yeast cells and ZIF-8 encapsulated yeast cells at an accelerating voltage of 10.0 kV. In Fig. 2(a), the unencapsulated yeast cell exhibits an egg-like shape, with clearly visible cell membrane and budding structures. In contrast, Fig. 2(b) shows that the ZIF-8 encapsulated yeast cell lacks visible membrane features, as the surface is completely covered with crystal formations. FE-TEM images [Fig. 2(c, d)] further reveal that the ZIF-8 crystals predominantly exhibit straight edges and cubic shapes. These observations confirm that the surface of the yeast cells is surrounded by cubic ZIF-8 crystals, forming an exoskeleton that contrasts with the morphology of unencapsulated yeast cells.
Fig. 3 shows the elemental distribution analyzed by EDS. The presence of carbon (51.00 %), nitrogen (19.37 %), oxygen (18.42 %), and zinc (11.20 %) was confirmed. These elements originate from the imidazole ligands (providing C, N, and O) and the metal nodes of ZIF-8 (providing Zn). In particular, the detection of zinc strongly supports the successful encapsulation of yeast cells within a ZIF-8 shell.
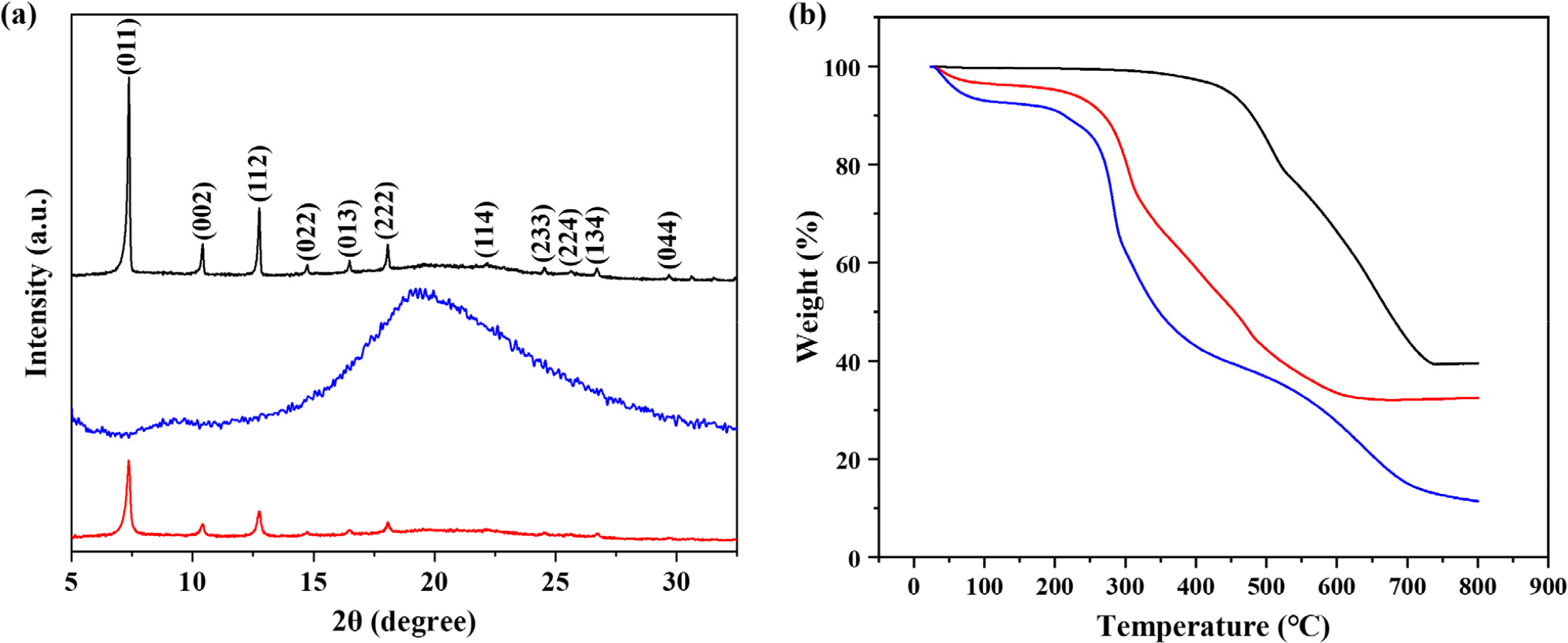
Fig. 4 presents the physicochemical characterization of the samples. (a) XRD patterns and (b) TGA curves are shown for ZIF-8 crystals (black), unencapsulated yeast cells (blue), and ZIF-8 encapsulated cells (cell@ZIF-8, red). (a) The XRD pattern of ZIF-8 crystals displays distinct diffraction peaks between 5° and 35° (2θ), indicating a porous crystalline structure. Prominent peaks corresponding to the (011), (002), (112), (022), (013), and (222) planes were identified. In contrast, unencapsulated yeast cells exhibit a broad, diffuse pattern due to their amorphous nature. The XRD pattern of cell@ZIF-8 closely resembles that of pure ZIF-8 crystals, confirming successful encapsulation of yeast cells within the ZIF-8 matrix. (b) TGA analysis was conducted under a nitrogen atmosphere, with a heating rate of 20 °C/min from 25 °C to 800 °C. ZIF-8 crystals exhibited a 58 % weight loss between 504 °C and 720 °C, corresponding to the decomposition of imidazole ligands. Subsequently, an additional 39 % weight change indicates oxidation and the formation of zinc oxide. For unencapsulated yeast cells, an initial 10 % weight loss between 30 °C and 122 °C corresponds to moisture evaporation. A major 51 % weight loss occurred between 197 °C and 436 °C, attributed to the thermal degradation of exopolysaccharides (EPS), with approximately 90 % total weight loss at the end. For cell@ZIF-8, initial weight loss occurred between 30 °C and 114 °C due to water evaporation. The second stage showed a 54 % weight loss from 209 °C to 484 °C, corresponding to decomposition of yeast cell components, particularly EPS. At the end of the analysis, approximately 32 % of the material remained, likely corresponding to zinc oxide residues.
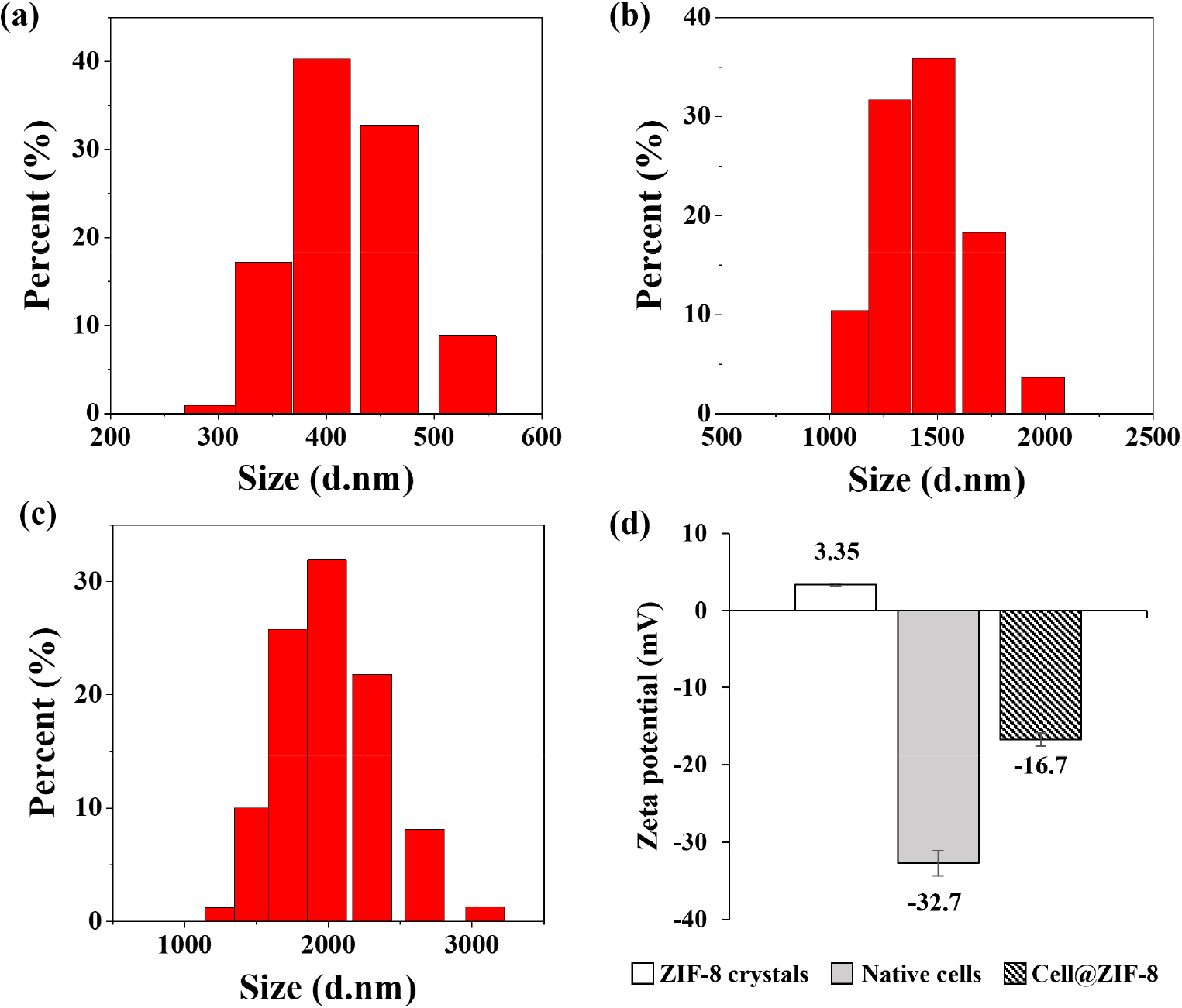
Fig. 5(a-c) presents the particle size distribution analyzed using DLS. The average diameters were approximately (a) 400 nm for ZIF-8 crystals, (b) 1,500 nm for unencapsulated yeast cells, and (c) 2,200 nm for ZIF-8 encapsulated yeast cells. Among the three, ZIF-8 encapsulated yeast cells exhibited the largest size, which is attributed to the formation of the ZIF-8 shell around the yeast cells. Fig. 5(d) shows the comparison of zeta potentials. ZIF-8 crystals exhibit a positive surface charge due to unsaturated Zn2+ ions. Native yeast cells possess a negative surface charge of -32.7 mV, attributed to negatively charged components of the cell membrane and wall. In contrast, the zeta potential of cell@ ZIF-8 was -16.7 mV, indicating reduced surface negativity due to the relatively positive ZIF-8 coating. This change further supports the successful encapsulation of yeast cells by ZIF-8 crystals.
Fig. 6 shows CLSM images used to assess cell viability under neutral conditions (pH 7). FDA is hydrolyzed by intracellular esterases present only in live cells, resulting in green fluorescence. Live cells appear green, while dead cells remain unstained. The viability of (a) native yeast cells and (b) ZIF-8 encapsulated yeast cells (cell@ZIF-8) was confirmed to be 97.73 % and 95.32 %, respectively. These results indicate that the ZIF-8 shell does not significantly affect cell viability, as the survival rate difference before and after encapsulation is only about 2 %.
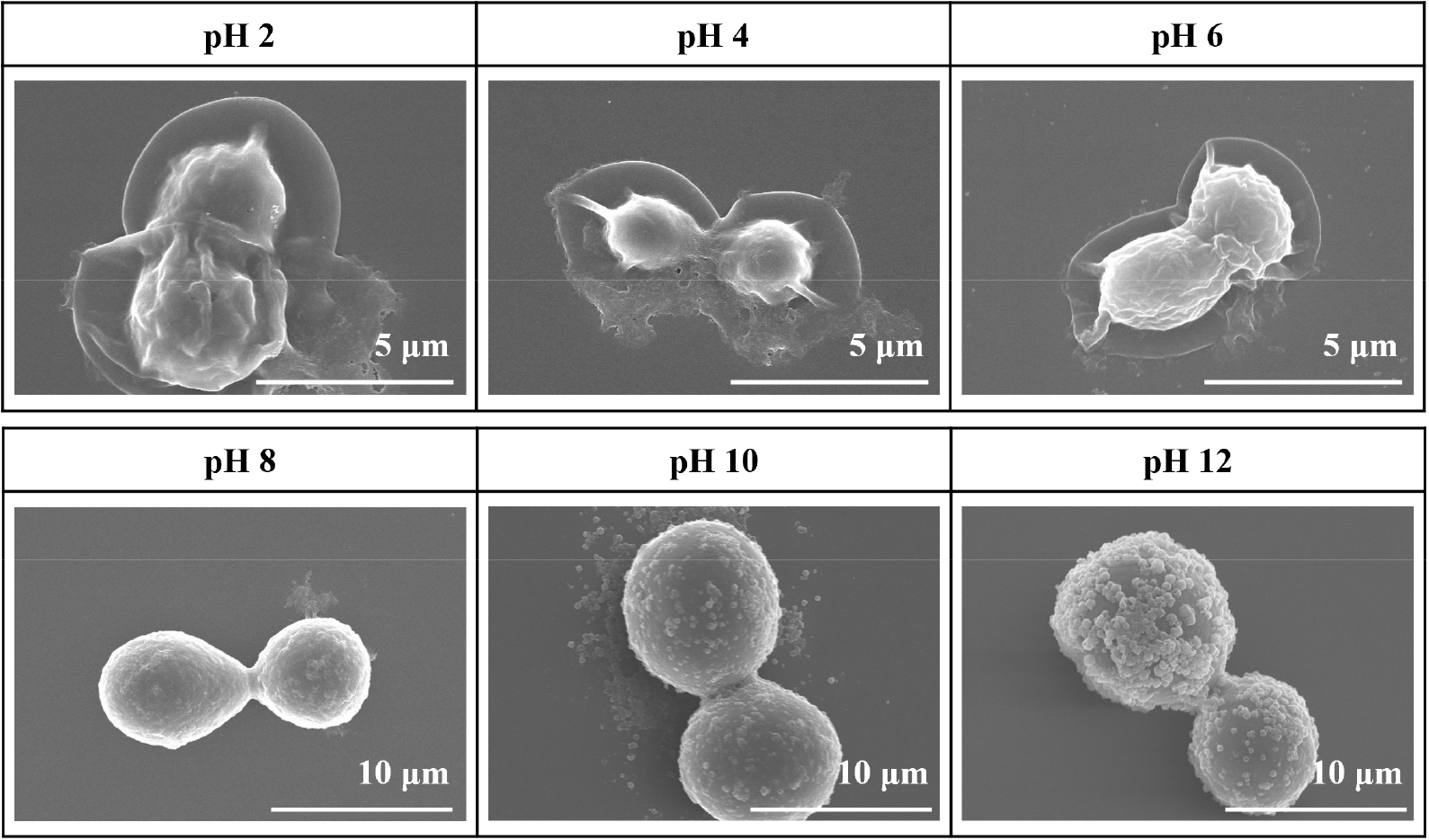
As shown in Fig. 7, FE-SEM images display the morphology of cell@ZIF-8 after storage under various pH conditions. In acidic environments (pH 2 and 4), hydrogen ions cleave the coordination bonds between the imidazole ligands and zinc nodes, leading to the degradation of the ZIF-8 shell. At pH 6, phosphate ions in the buffer solution coordinate with the zinc nodes, further disrupting the ligand-metal bonds and resulting in shell removal. At pH 2, 4, and 6, the ZIF-8 shells were completely removed from the cell surface, and ruptured cell membranes were observed. At pH 8, no distinct ZIF-8 crystal structure is visible, but the overall shell morphology is still partially retained. At pH 10 and 12, the ZIF-8 crystals remain intact on the cell surface due to the chemical stability of ZIF-8 under alkaline conditions.28) Thus, the ZIF-8 shell morphology is at least partially maintained at pH 8, 10, and 12.
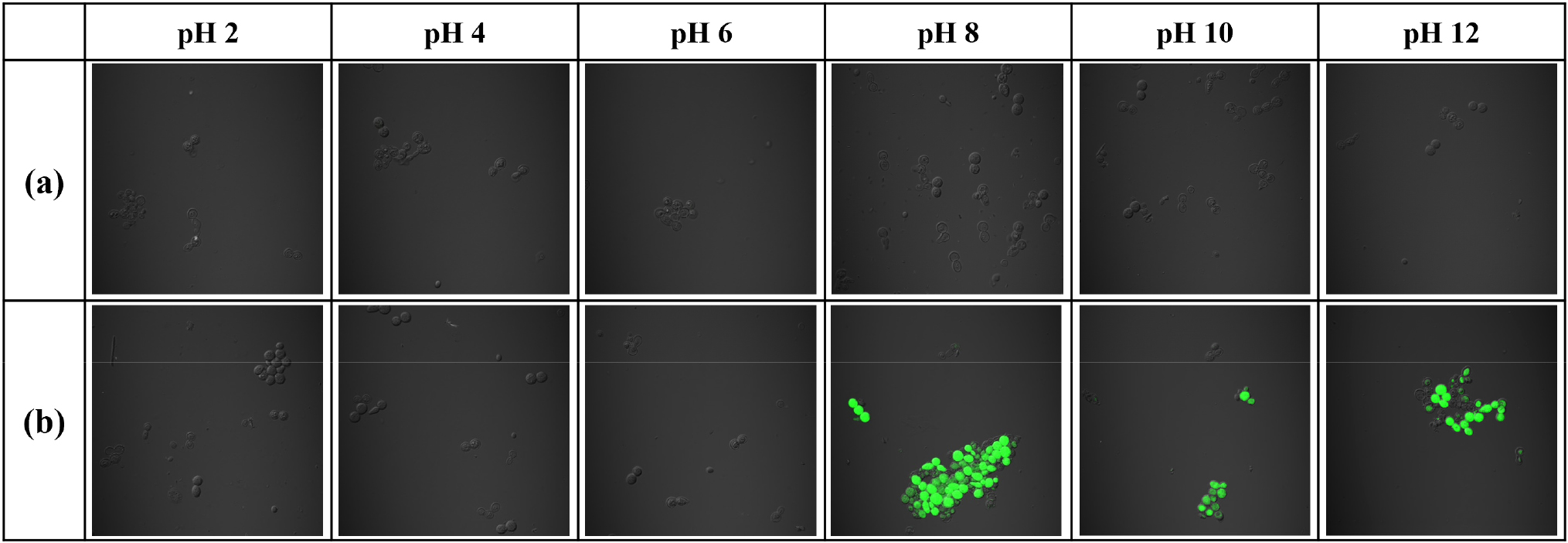
Fig. 8 shows CLSM images of yeast cells exposed to various pH conditions. (a) Native yeast cells showed no FDA staining under all pH conditions, confirming 0 % cell viability and indicating complete cell death. (b) ZIF-8 encapsulated yeast cells also exhibited 0 % viability at pH 2, 4, and 6, as the ZIF-8 shell was fully degraded under acidic conditions, leaving the cells unprotected and susceptible to damage. In contrast, at pH 8, 10, and 12, the ZIF-8 shell remained partially intact, providing a protective barrier for the yeast cells. As a result, the encapsulated cells exhibited significantly higher viabilities: 62.32 % at pH 8, 63.15 % at pH 10, and 52.94 % at pH 12. These findings clearly demonstrate that the ZIF-8 shell effectively protects yeast cells in moderately to strongly alkaline environments.
4. Conclusion
In conclusion, yeast cells were successfully encapsulated with a ZIF-8 shell, forming a stable crystalline layer on the cell surface. The encapsulated cells were tested under a wide range of pH conditions (2~12) to evaluate their environmental stability. While unencapsulated yeast cells did not survive at any tested pH level, ZIF-8 encapsulated cells showed significant protection in alkaline environments. At pH 8, 10, and 12, the shell structure remained intact, and more than 50 % of the cells retained viability. In contrast, under acidic to neutral conditions (pH 2, 4, and 6), the ZIF-8 shell was degraded, resulting in membrane rupture and cell death. These findings suggest that alkaline conditions provide the most favorable environment for preserving the viability and structural integrity of ZIF-8 encapsulated cells. This encapsulation strategy may also hold potential for pH- responsive drug delivery and enhancing biocatalyst stability under extreme conditions.