1. Introduction
It is well-known that the physical and chemical properties of materials could be attributed to its particle size and microstructure. Novel nano- and microstructured materials were prepared, and its related fields of biological labeling and therapy,1) PEC water oxidation,2) homogeneous nanocatalysis,3) single-Molecule Biosensors,4) and enhanced Raman scattering5) futher more researched. The examination of the semiconductors in photoelectrochemical (PEC) cells is a fast and simple method tocharacterize their electrochemical behaviors.6) Although many semiconductors have been examined as photoelectrodes, 7,8) most of them have modest PEC efficiencies owing to the narrow visible light absorptions and unavoidable electron-hole recombinations.
Bismuth molybdate (Bi2MoO6) with moderate bandgap (2.6 eV) has attracted more attention, in photocatalysis research such as PEC water splitting due to its appropriate band edge positions.9,10) However, comparing with the semiconductors as TiO2 and WO3 it owns lower water oxidation kinetics, fast electron-hole recombination rates and low photocurrent density.11,12) For bare Bi2MoO6 more than 50 % of the solar spectrum cannot be sufficient used due to its limiting of photoabsorption, thus many researchs are investigated to enhance its photoabsorption efficiency of Bi2MoO6. Some materials about graphene nanosheet, carbon nanofiber and nitride13-15) were choosed to combine with Bi2MoO6 for achieving this aim.
In addition, as an emerging star with two-dimensional characteristic structure, graphic carbon nitride (g-C3N4), where tri-s-triazine units connected by amino groups in each layer, can be seen as a kind of nitrogen substituted graphene-like layered material.16) Comparing with another carbon material of graphene, g-C3N4 own unique semiconductor properties, solid alkaline, and complexing ability.17,18)
Up to now, some reports were focused on fabrication Bi2MoO6 with different morphology followed by coupling with g-C3N4 for enhancement of its photocatalytic activity or photoelectrochemical activity. Li et al.19) reported that the g-C3N4/Bi2MoO6 heterojunction with a Bi2MoO6 content of 16.1 wt% exhibited the highest photocatalytic activity, and its photocatalytic efficiency was more than three times those of pure g-C3N4 or Bi2MoO6. Zhen et al.20) reported that 100 g of g-C3N4 was integrated into 100 g of the pre-calcined Bi2MoO6 at 200 °C to produce 100 % g-CN/BMO-200, showing the highest photocatalytic performance. Ma et al.21) researched on fabrication of vertically aligned Bi2MoO6 nanosheet arrays, followed by coating the metal-free ultrathin g- C3N4 nanosheets on its surface which resulting in higher photoelectrochemical activity compared to the pure Bi2MoO6 films. Thereby, the exploration of nanosized Bi2MoO6 combining with g-C3N4 nanosheet may be a valuable research for enhancing the PEC performances of Bi2MoO6.
Herein, we demonstrate a successful and facile procedure which was focusing on the synthesis of Bi2MoO6 nanoparticle through a hydrothermal method and coupling with g-C3N4 nanosheet via heat-treatment as shown in Schematic 1, due to this route is safe, convenient and facile to synthesize g-C3N4 modified nanocomposites. It is obviously that the introduction of small sized g-C3N4 nanosheet could provide large surface area and uniform electrons transport pathways, which remarkably facilitated the charge separation, resulting in much higher photoelectrochemical activities and stabilities than pure Bi2MoO6 and g-C3N4.
2. Experimental Procedure
2.1. Materials
All of the reagents used in this experiment were analytical reagent grade and without further purification.
2.2. Synthesis of Bi2MoO6 nanoparticle
In a typical synthesis procedure, Bi2MoO6 nanoparticle was synthesized via sonochemical assisted hydrothermal method with modification.22) The detailed process is described as follows: 35 mmol of Bi(NO3)3·5H2O and a certain amount of Na2MoO4 nanopowder were added into 30 mL of ethylene glycol containing 2 mL of PEG-600 under stirring for 30 min. The above solution was sonicated at room temperature for 60 min using a controllable serialultrasonic apparatus (using 180W, Ultrasonic Processor UH600, China). The thoroughly mixed solution was transferred into a Teflon-lined stainless steel autoclave that was sealed. The contents were then warmed to 160 °C for 24 h. Finally, after the temperature of the mixture was brought down to room temperature and the mixture was filtered and washed with distill water for 3 times, then dried in a vacuum oven at 60 °C. Then the dried products were calcined at 500 °C for 2.5 h in muffle furnace and the final products becomes yellow were used as start materials in next step.
2.3. Synthesis of g-C3N4 modified Bi2MoO6
The g-C3N4 modified Bi2MoO6 was synthesized by heating melamine with pre-prepared Bi2MoO6 nanopartiles in a muffle furnace according to published literature but with some modifications.23,24) Briefly, 0.2 g Bi2MoO6 and calculated amount of melamine were put into an agate mortar. After fully grinding for 30mins it was transferred into an alumina crucible with a cover. The crucible was then placed in a muffle furnace and heated to 550 °C (heating rate 5 °C min−1), and was heat treated at 550 °C for 3 h. After finishing the first step of heattreatment, the products were collected and re-ground for 30 mins before heated to 500 °C for another 2 h. According to this method, g-C3N4/Bi2MoO6 composites with calculated amount of melamine with mass ratios from 5 to 9 wt% were synthesized and named as BMC 5, BMC 7, and BMC 9, respectively.
2.4. Characterization and photoelectrochemical measurements
The crystal structures of prepared compounds were performed by X-Ray Diffraction (XRD) with Cu Kα irradiation (X'Pert3, PANalytical), while the morphology and microstructure of the compounds were characterized by Scanning Electron Microscopy (SEM, Nova NanoSEM 450) and Raman spectra of the samples were observed using a spectrometer (Jasco model name NRS-3100) having an excitation laser wavelength of 532.06 nm. The photoelectric behaviors of the samples were analysed in a typical three-electrode system via computercontrolled electrochemical workstation (CHI660E). And a counter electrode (Pt foil), reference electrode (saturated Ag/AgCl electrode) and working electrodes (glassy carbon electrode with a diameter of 3 mm) were used with 0.1 mol/L Na2SO4 aqueous solution as electrolyte.25) To prepare the working electrode, 4 mg of prepared samples and 30 mL of Nafion solution were put into 1 mL of water-ethanol solution followed by sonicating for 2 h. Then 5 mL of this mixture was transferred onto the GCE electrode, and was thorough dried under an infrared light. A solar simulator (500 W Xe Lamp, NBET Company) with a UV-cutoff filter (> 420 nm) was employed with illumination intensity of 100 mWcm-2. In a three-electrode cell (CHI660E, China), the frequency was choosed as 10 K to 0.01 Hz and the amplitude was selected as 5 mV to obtain the EIS characteristic.
3. Results and Discussion
Fig. 1(a) to (c) shows the morphologies with small and high resolution of the as-prepared sample, indicating that some irregular size particles were formed with a little agglomeration due to surfactant of PEG-600 was used as morphology modifier which restrain severely agglomeration of Bi2MoO6. The higher-magnification image in Fig. 1(a,b) details the nanoparticles with small size below 100 nm. More specifically, in Fig. 1(c) shows us some irregular size which may conjecture that some Bi2MoO6 nanoparticles were grow together to form bulk or irregular sphere. Similar type of result was reported by Ali et al.26) while surfactant was removed via annealing at high temperature at 500 °C. As shown in Fig. 1(d), the main peaks nearby 28.3°, 32.5°, 46.7°, and 55.6° may attributed to orthorhombic Bi2MoO6 (JCPDS 21–0102), which was agreed with Zhang’s report.27)
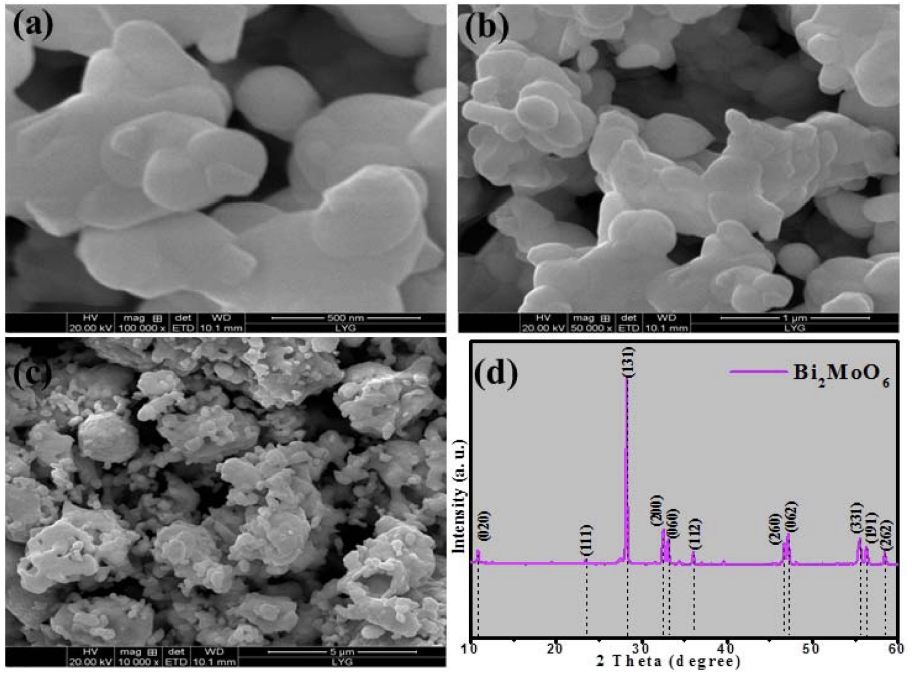
Fig. 1
FE-SEM micrographs with different resolution (a to c) and XRD pattern (d) of as-prepared pure Bi2MoO6 nanoparticles.
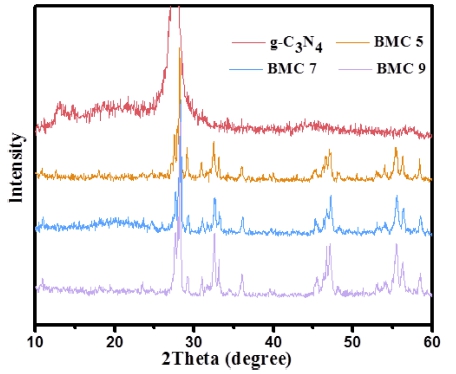
Fig. 2 shows us the XRD patterns of pure g-C3N4 and g-C3N4 modified Bi2MoO6 nanocomposites. Two peaks at values of 13.2° and 27.3° were detected and it may belong to (100) and (002) planes of g-C3N4, this result is agreed with Zhao’s report 28) The XRD patterns of g- C3N4/Bi2MoO6 (BMC 5-9) demonstrate no evident changes on the crystals structure, compared with pure Bi2MoO6. However, there are no obvious characteristic diffraction peaks of g-C3N4 can be observed, which may be ascribed to low amount of melamine used in this work and the overlapping characteristic peaks between (002) of g-C3N4 and (131) of Bi2MoO6.29)
The microstructures of as-prepared g-C3N4/Bi2MoO6 nanocomposites are revealed by FE-SEM images with two kinds of magnification (Fig. 3). With increasing the amount of melamine and annealing at different high temperature for two times, the morphology and particle size of g-C3N4 and Bi2MoO6 are changed. The morphology of Fig. 3(b) is similar with Fig. 1(c), but the distribution state of Bi2MoO6 particles in Fig. 3(a) is become better than pure Bi2MoO6 as shown in Fig. 1(b). From Fig. 3(a) and (b), it is difficult to distinguish which part related to g-C3N4, it could be ascribed to some amount of melamine was evaporated among annealing process, thus leads to low content of g-C3N4 generated.30) When the amount of melamine was increased to 7 wt% and 9 wt%, some g- C3N4 nanosheets can be clearly observed as shown in Fig. 3(c) to (f) which related to sample BMC 7 and BMC 9. From Fig. 3(c) and (d), the g-C3N4 displays a platelet-like morphology with curve surface. While most Bi2MoO6 nanoparticles were covered or exhibit agglomerate dispersion state, which leads to low amount of Bi2MoO6 can be exposed. However for sample BMC 9, after introduction of situable amount of melamine about 9 wt% in this system, the sheet structure of g-C3N4 and regular Bi2MoO6 nanoparticles keeps well and abundantly scattered state as shown in Fig. 3(e) and (f). The finely combined microstructure of BMC 9 will be benefit for the efficient charge transmission between g-C3N4 and Bi2MoO6, and may improve its photoelectrochemical properties.28)

Fig. 3
FE-SEM micrographs with different resolution of as-prepared g-C3N4/Bi2MoO6 nanocomposite: (a, b) BMC 5, (c, d) BMC 7, (e, f) BMC 9.
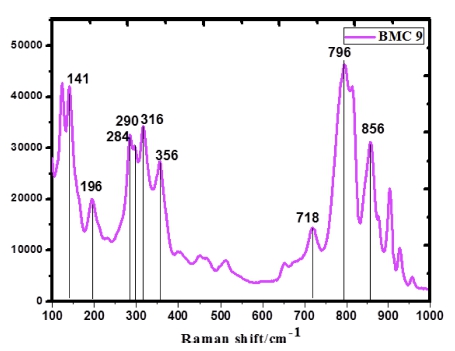
The Raman spectra of BMC 9 sample synthesized via physical sintering method is shown in Fig. 4. They were classified into three regions: < 180 cm−1, 180 ~ 500 cm−1, and 700 ~ 850 cm−1. Generally speaking, the peak above 600 cm−1 is related to be Mo-O stretching vibration, while the peak below 400 cm−1 is corresponded to the bending, oscillation and other vibration. Vibration peak at 144 cm−1, specified as the lattice mode of Bi3+ atoms, was mainly in the direction normal to the layers. The main absorption peak at 796 cm−1, 856 cm−1 and 718 cm−1 is related to deformed MoO6 octahedrons with stretching mode of the bismuth-oxygen tensile vibration. The intense Raman modes near 290 and 280 cm−1 seemed to be from the Eg bending vibrations. The mode below 400 cm−1, such as peaks at 356, 284, 196 and 136 cm−1 also represent the vibration of the Mo-O bond.31-33)
The PEC activities of as-prepared samples were investigated by transient photocurrent responses (Fig. 5) at an applied potential of -0.35 V vs Ag/AgCl in 0.1 M Na2SO4 aqueous solutions without sacrificial agent under visible light illumination (λ > 420 nm). As shown in Fig. 5(a, b), pure Bi2MoO6 exhibits a higher photoelectric current than pure g-C3N4 which exhibits tapering photoelectric behavior and obvious photocorrosion characteristic. According to the photoelectric properties of pure Bi2MoO6 and g-C3N4, both of them have the same problem of high recombination rate of photogenerated electron-hole pairs. To confirm this transportation of photoelectron, electrochemical impedance spectrum was carried out as shown in Fig. 5(f). The charge-transfer resistance about of pure Bi2MoO6 and g-C3N4 is nearby 85 W. After combining Bi2MoO6 with g-C3N4, the photocorrosion problem was reduced to a certain extent. The visible light response properties of the as-obtained g-C3N4/Bi2MoO6 composites (BMC 5-9) were fairly improved shown in Fig. 5(c-e). Special for sample BMC 9, it exhibits higher and stable photocurrent response characteristics. In general, the smaller slope of arc represents and BMC 9 nanocomposite exhibits the lowest charge-transfer resistance about 73 W. Therefore, the results of EIS and photocurrent response demonstrated that the introduction of g-C3N4 nanosheet was effective to improve the charge separation and the transfer efficiency of g-C3N4/Bi2MoO6, resulting in the enhancement of its photoelectric behavior.21)
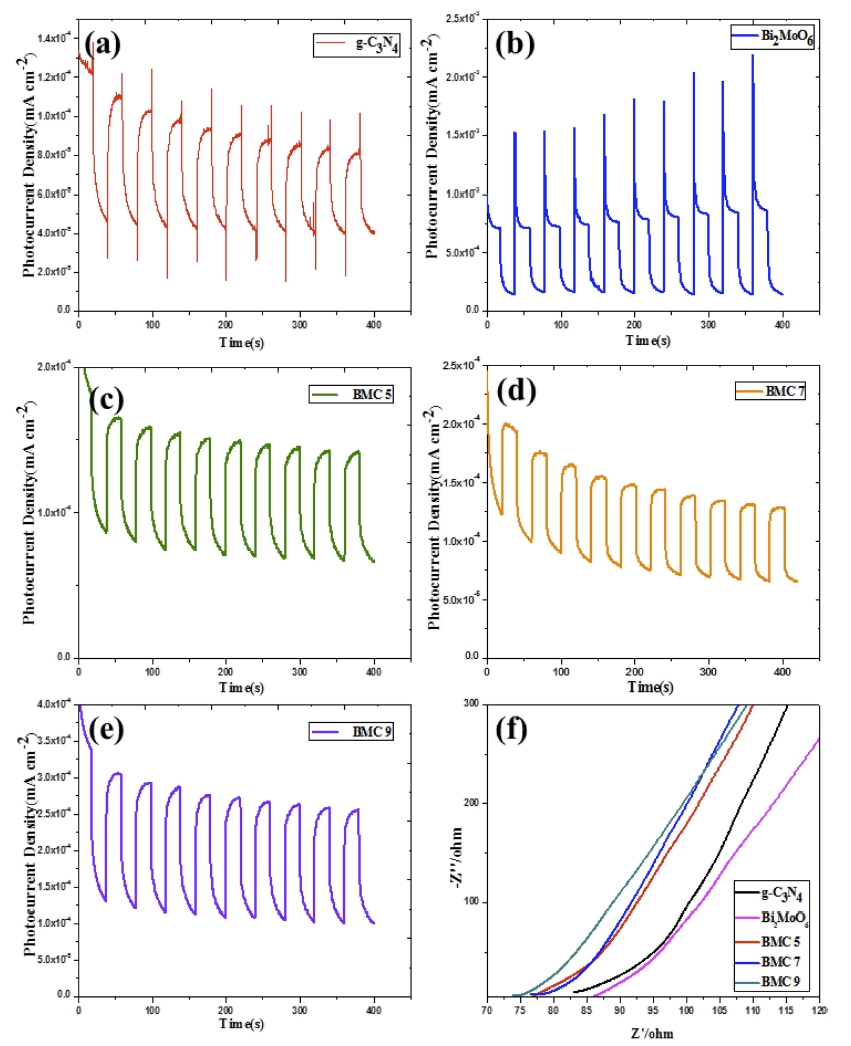
Fig. 5
Transient photocurrent response (a-e) of as-prepared samples: (a) g-C3N4, (b) Bi2MoO6, (c-e) BMC 5-9; and Electrochemical impedance spectroscopy (f).
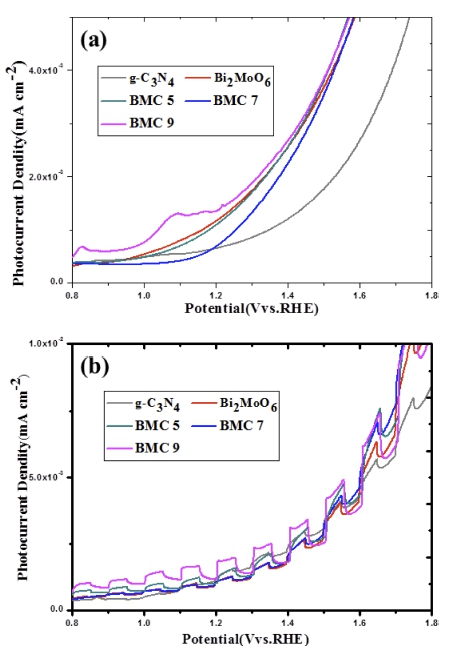
The PEC activities of the as-prepared samples were further investigated by linear sweep voltammetry (LSV) as shown in Fig. 6. As shown in Fig. 6(a), the linear sweep voltammetry (LSV) was carried out in a wide potential range of 0 ~ 1.9 V vs. RHE without visible light irradiation. Compared with pure Bi2MoO6 and g-C3N4, BMC 9 exhibits higher current density under the same potential which could be attributed to it has smaller impedance. In order to investigate its visible light photoresponse activity of as-prepared samples, an average chopped LSV was carried out under intermittent visible light irradiation with an illumination intensity of 100 mWcm−2. As vividly, for g-C3N4 nanosheet modified Bi2MoO6 nanocomposites, the photocurrent density all get enhancements compare with pure Bi2MoO6 and pure g-C3N4. Under visible light chopped condition, BMC 9 exhibits highest and stable photoelectric behavior at same potential. Based on the above results, we believe that the best PEC performance of BMC 9 composite is mainly due to better electron transport and efficient electron hole separation. In summary, it is clear that g-C3N4 nanosheets can be used as excellent co-catalyst of Bi2MoO6 to improve its PEC properties.
4. Conclusion
In this work, g-C3N4 nanosheet modified Bi2MoO6 nanocomposites were prepared by two-step method. The morphology of Bi2MoO6 was well changed and its distribution state also became better through annealing process of melamine. Ascribed to combining Bi2MoO6 with g-C3N4, its photocorrosion problem was reduced to a certain extent. The g-C3N4/Bi2MoO6 can be directly used as electro and exhibit an excellent response to visible-light, which realized two orders of magnitudes enhancement in PEC activity than pure Bi2MoO6 and g- C3N4. In addition, BMC 9 composite exhibits a much higher photocurrent density and stable PEC properties may attribute to interconnected network of Bi2MoO6 nanoparticles and g-C3N4 nanosheets.