1. Introduction
Zinc Oxide(ZnO) is an attractive wide bandgap semiconductor with potential applications in electronics, optoelectronics and sensing such as ultra-violet(UV) light emitting diodes and laser diodes, field-emitting devices, high power electronic devices, solar cells and gas sensors. Its wide direct band gap energy of 3.37 eV is suitable for UV light emitting. Its large exciton binding energy of 60 meV allows for efficient excitonic laser emission under low threshold power even at room temperature. Moreover, ZnO is transparent to visible light due to its wide bandgap, which makes it possible to use in transparent electronic devices and solar cells. Thus much effort has been employed to develop ZnO-based devices.
Recently, because of its interesting properties, ZnO has been studied for applications in nano-electronic and nanophotonic devices. So, many studies have intensively been made on the synthesis and electro-optical characterization of ZnO micro/nanostructures. Many methods have been used to synthesize ZnO micro/nanostructures including thermal evaporation,1) chemical vapor deposition,2) carbothermal method,3) sol-gel method,4) hydrothermal method5) and so on. Among these methods, thermal evaporation method has been commonly used due to the simplicity and low cost. In the thermal evaporation method, the morphology and phase structure of product is dependent on source material as well as the processing parameters. Morphological variation is ascribed to the difference between the volatility of source materials. ZnO micro/nanostructures have mainly been synthesized by using inorganic compounds such as Zn, ZnO and ZnS as source materials in the thermal evaporation method.6-9) However there are hardly reports on the thermal evaporation synthesis of ZnO micro/nanocrystals using zinc salts such as zinc acetate, zinc carbonate and zinc iodide as zinc source materials, which have higher vapor pressures than those of Zn, ZnO and ZnS. Microcrystals define as crystals with micrometer-scale sizes and nanocrystals mean crystals with a size in the nanometer range.
In this paper, we report the synthesis of ZnO micro/ nanocrystals through thermal evaporation of various zinc salts under air atmosphere and their luminescence properties.
2. Experimental Procedure
0.5 g of zinc acetate(99.99 %, Sigma-Aldrich), ZnCO3 (Purum p.a., Sigma-Aldrich) and ZnI2(99.99 %, Sigma- Aldrich) powder were used as the source materials for synthesizing ZnO micro/nanocrystals. For the comparison, Zn powder(99.99 %, Sigma-Aldrich) was also used as a source material, and the morphologies and the luminescence properties of the as-prepared products were characterized. The source material was put in a alumina crucible. Then the crucible was inserted into the center of quartz tube in a horizontal tube furnace in air under atmospheric pressure. The furnace was heated to the processing temperature. The processing temperatures were 1000 °C. The source powders were evaporated and reacted with oxygen in air at the temperature. The processing time was 1 hr. After one hour of evaporation and oxidation process, the furnace was turned off and cooled down to room temperature
The oxidized products were collected for the characterization. The morphology of the products was observed using scanning electron microscope(SEM, Quanta 200 FEG, FEI Co., Netherlands, 15 kV). The components of the products were studied using energy dispersive X-ray (EDX, Quanta 200, EDAX INC., USA, 15 kV) spectroscope. The crystal structure was analyzed by X-ray diffractometry(XRD, X’Pert Pro Multi Purpose Diffractometer, PANalytical Co., Netherlands, 40 kV, 30 mA) with Cu-Kα radiation. The cathodoluminescence(CL) measurement was performed at room temperature using CL spectroscope(Mono CL4, GATAN Co., United Kingdom, 15 kV).
3. Results and Discussion
Fig. 1 shows the SEM images of the as-prepared products synthesized by using different source materials: Zn, zinc acetate, ZnCO3 and ZnI2. When the product is prepared by using Zn powder as a source material, tetrapod-shaped crystals with straight and uniform four legs are formed in large quantity. The diameter of the four legs gradually decreases along the growth direction, resulting in a tapered shape. Granular-shaped ZnO nanostructures are observed in the SEM image of the product prepared by using zinc acetate as a source material. The granular-shaped nanostructures are 40 nm in an average diameter and 100 nm in length. In the case of ZnCO3 source material, the as-prepared product consists of spherical particles with an average diameter of 600 nm. Crystals with crayon shape are observed in the product prepared by using ZnI2 as a source material. The crayon shaped crystals have the diameter of 1.2 μm and the length of 9 μm. As shown in Fig 1(d), the crayon-shaped crystals have hexagonal cross sections, indicating that the crystals grow along the [001] direction. According to the growth habit of ZnO crystal with wurtzite crystalline structure, the growth rate along the [0001] direction is faster than any other direction.10) Thus ZnO crystal grows fastest along the [0001] direction at the initial stage of the growth, leading to the formation of anisotropic shape such as nanorods and nanowires. As the growth proceeds, the crystal plane with rapid growth rate tends to disappear, whereas the crystal planes with slow growth rates remain. Accordingly, (0001) plane in ZnO crystal will disappear gradually, resulting in the formation of the crayon-shaped morphology. The SEM results showed that source materials played an important role in the variation of morphology of the products. The morphology is likely to be associated with the Zn vapor concentration (or Zn vapor pressure). Zinc acetate decomposes at 237 °C(melting and boiling point) and ZnCO3 has a boiling point of 333.6 °C, while Zn and ZnI2 have relatively high boiling points of 907 °C and 1,150 °C, respectively. The vapor pressure increases generally as the boiling point decreases. Therefore, in case of zinc acetate and ZnCO3 source materials, the concentration(or vapor pressure) of growth species on the surface of nuclei is very high, which leads to the 3 dimensional(3D) growth on all facets of the nuclei. Thus the growth rate of the facets of the nuclei tends to be equal, resulting in the formation of granular and spherical shaped nanoparticles. The concentration of growth species is low when Zn and ZnI2 with relatively high boiling points are used as the source materials. The low concentration allows the long surface diffusion distance of growth species, leading to the increase in the probability of two dimensional growth on the surface of nuclei. The growth rate of the surface plane with high density of growth sites such as ledge, ledge-kink and kink is fast. ZnO with wurtzite crystal structure is a polar crystal, in which c-axis is the polar axis. Thus, ZnO crystal has the positively charged Zn-terminated (0001) plane and negatively charged O-terminated (000-1) plane. The Zn-terminated (0001) plane is chemically active, while the O-terminated (000-1) plane is inert. The active Zn-terminated plane would provide the preferential growth sites for the growth of ZnO crystal. Therefore, the growth rate along [0001] direction is fast, resulting in the formation of one dimensional morphologies like tetrapod and crayon shape with high aspect ratio.

Fig. 1
SEM images of the as-prepared products synthesized via thermal evaporation of different source materials: (a) Zn, (b) zinc acetate, (c) ZnCO3 and (d) ZnI2.
Fig. 2(a-d) shows the XRD patterns of the products prepared by using the source materials of Zn, zinc acetate, ZnCO3 and ZnI2, respectively. Similar XRD results are observed irrespective of the source material. All the peaks in the XRD patterns can be well indexed to ZnO with the hexagonal wurtzite structure. The XRD results shows that all the products are ZnO materials with the hexagonal wurtzite crystalline structure.
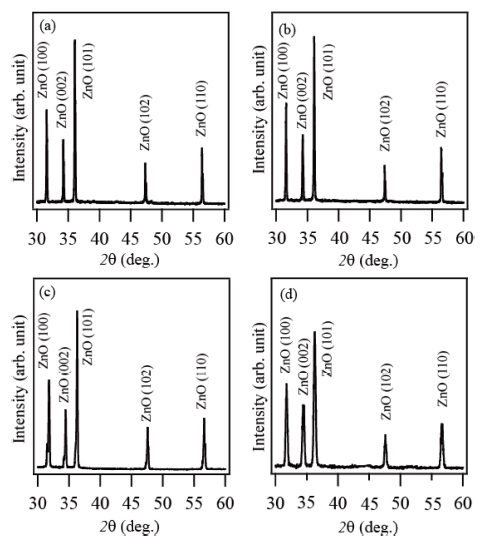
Fig. 2
X-ray diffraction patterns (XRD) of the products prepared via thermal evaporation of different source materials: (a) Zn, (b) zinc acetate, (c) ZnCO3 and (d) ZnI2.
Fig. 3(a-d) shows the EDX spectra of the products prepared by using the source materials of Zn, zinc acetate, ZnCO3 and ZnI2, respectively. In the EDX spectra taken for all the products, the elements Zn and O are detected, indicating the products are highly pure ZnO.
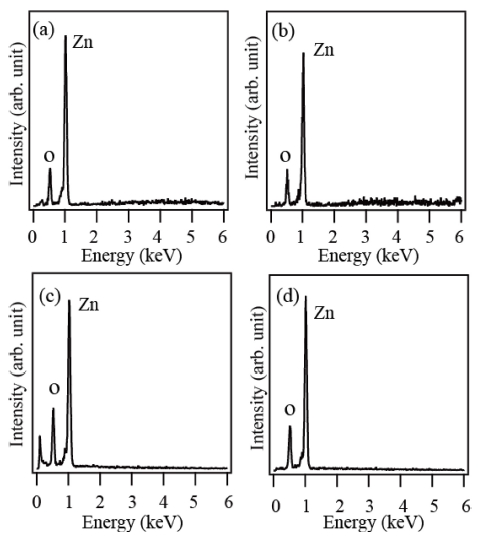
Fig. 3
EDX spectra of the products prepared via thermal evaporation of different source materials: (a) Zn, (b) zinc acetate, (c) ZnCO3 and (d) ZnI2.
Fig. 4(a-d) shows the CL spectra of the products prepared by using the source materials of Zn, zinc acetate, ZnCO3 and ZnI2, respectively. A strong ultraviolet(UV) emission centered at 380 nm is observed in the CL spectrum of the product prepared via thermal evaporation of Zn. A strong and broad green emission centered at around 500 nm is observed in the CL spectra of the products prepared by using zinc acetate and ZnCO3 as source materials. In the CL spectrum of the crayonshaped ZnO crystals synthesized via thermal evaporation of ZnI2, both the UV and green emissions are observed. However, the intensity ratio of the UV to the green emission is low. It is known that the UV emission also called near band edge emission is attributed to the free exciton recombination, while the green emission originates from the recombination of a photo-generated hole with an electron trapped in singly ionized oxygen vacancy in ZnO.11) Thus the intensity ratio of UV to green emission may be used for relative evaluation of crystalline quality of ZnO crystals. The ZnO crystals would have better crystalline quality according to the higher intensity ratio. The highest UV-to-green emission ratio was observed in the CL spectrum of the ZnO crystals produced using Zn metal powder, whereas UV emission was hardly detected in the CL spectra of the ZnO crystals prepared using zinc acetate and ZnCO3. The UV-to-green emission ratio of the ZnO crystals synthesized using ZnI2 is relatively very low. From the result, it is found that the ZnO crystals with the best crystalline quality were achieved when Zn metal powder was used as source material.
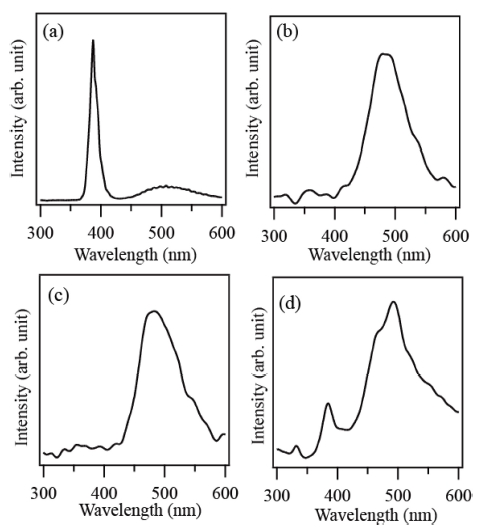
Fig. 4
Room temperature CL spectra of the products prepared via thermal evaporation of different source materials: (a) Zn, (b) zinc acetate, (c) ZnCO3 and (d) ZnI2.
In the present work, any catalysts were not used in the synthesis of ZnO crystals, suggesting that the growth of the ZnO crystals proceeded in vapor-solid mechanism. ZnO micro/nanocrystals with diverse morphologies were synthesized by tuning the source materials, which indicates that the source material is a key factor in the morphological control of ZnO crystals. High Zn vapor pressure enabled the isotropic growth of ZnO crystals in spherical shape, while low vapor pressure of Zn might favor the anisotropic growth of ZnO crystals in wire shape.
4. Conclusion
Under the same experimental conditions, ZnO crystals with different morphologies were synthesized by employing the different source materials. When zinc acetate was used as zinc source material, nanogranular shaped crystals with an average diameter of 40 nm were formed. Unique ZnO crystals with crayon shape were obtained through thermal evaporation of ZnI2 source material. For the product prepared by using ZnCO3 source, ZnO crystals with spherical shape were observed. In the case of pure Zn source material, tetrapod-shaped ZnO crystals were synthesized. It is suggested that the morphological variation of ZnO crystals may originate from the difference between the volatility of source materials. All the ZnO crystals had wurtzite crystalline structure. Cathodoluminesence spectra showed that a strong UV emission was observed from the tetrapods, while a strong green emission was observed from the other products. The strong green emission is indicative of high density of structural defects in the ZnO crystals.




