1. Introduction
2. Experimental Procedure
2.1. Materials
2.2. Preparation
2.3. Sample Preparation and Testing Methods
3. Results and Discussion
3.1. NMR and FT-IR of PMB
3.2. FT-IR Spectra of WPU, PMB-WPU and PMBxCu1-WPU
3.3. XRD Analyses
3.4. Emulsion Particle Size and Zeta Potential Analysis
3.5. TG-DTG of WPU, PMB-WPU and PMBxCu1-WPU
3.6. UV-Vis Spectra of PMB, WPU, PMB-WPU and PMBxCu1-WPU
3.7. Fluorescence Spectroscopy of PMB, WPU and PMBxCu1-WPU
4. Conclusion
1. Introduction
In the interdisciplinary field of materials science, developing new materials with both functionality and environmental friendliness is key to driving industrial upgrading. Fluorescent tunable materials have become an international research hotspot due to their application potential in cutting-edge fields such as intelligent lighting, flexible displays, and biosensing.1,2,3) Schiff base Cu(II) complexes, with their unique structural characteristics and versatile functions, provide new ideas for the innovative development of fluorescent materials.4,5)
Schiff bases are a class of organic ligands containing C=N structures. The lone pair electrons of their N atoms endow them with strong coordination ability, enabling them to form complexes with diverse structures with Cu(II). Schiff base Cu(II) complexes not only exhibit excellent performance in the fields of antibacterial, anticancer, and catalysis, but also show regulable fluorescent properties due to the π-π* transition of their ligands or the metal-ligand charge transfer (MLCT) effect.6,7) Although the d-d transition of Cu(II) is forbidden, conjugated ligands can significantly enhance its luminescent efficiency, endowing it with unique advantages in fields such as photochromism and optical probes.8) However, Schiff base Cu(II) complexes have inherent drawbacks including insufficient chemical stability, poor solubility, limited processability, biological toxicity, and inadequate optical and thermal stability.9,10)
Polymerization of Schiff base Cu(II) complexes can not only effectively address the aforementioned issues but also expand their performance range. As an environmentally friendly polymer, waterborne polyurethane (WPU) is widely used in fields such as industry, agriculture, and military due to its good film-forming property, flexibility, and low volatile organic compound (VOC) emission characteristics.11) However, traditional WPU has a single function and is difficult to meet the needs of high-end applications. Currently, research on the fluorescent properties of Schiff base Cu(II) complexes mainly focuses on small molecules, while their combination with WPU is rarely reported.12)
In this study, Schiff base Cu(II) complexes were innovatively introduced into the main chain of WPU, resulting in the preparation of PMBxCu1-WPU capable of achieving precise regulation over a broad wavelength range. PMBxCu1-WPU enables precise control of fluorescence intensity and color, holding promise for opening up new application pathways in high-end fields such as intelligent anti-counterfeiting, biomedicine, and optoelectronic devices.
2. Experimental Procedure
2.1. Materials
Terephthalaldehyde, ethanolamine, p-toluenesulfonic acid, n-butanol, copper nitrate trihydrate, 1,4-butanediol (BDO, AR), 2,2-dimethylolbutyric acid (DMBA, AR), and triethylamine (TEA, AR) were purchased from Shanghai Macklin Biochemical Technology Co., Ltd. Dibutyltin dilaurate (DBTDL, AR) was purchased from Shanghai Aladdin Biochemical Technology Co., Ltd. Isophorone diisocyanate (IPDI, AR) and polytetrahydrofuran diol-1000 (PTMG-1000, Mn = 1,000 g/mol, AR) were provided by Bayer AG, Germany. Among them, IPDI and PTMG-1000 were dried under vacuum at 90 °C for 12 h before use. Other raw materials were used without treatment.
2.2. Preparation
2.2.1. Preparation of Schiff Base PMB
First, terephthalaldehyde and ethanolamine were used as reactants, p-toluenesulfonic acid as the catalyst, and n-propanol as the solvent. These were sequentially added to a three-necked flask containing toluene and thoroughly mixed. Subsequently, a water separator, the three-necked flask, and a condenser were connected to set up the reaction apparatus. The system was placed in a 120 °C environment for magnetic stirring during the reaction. When the water level in the water separator no longer changed, the reaction was judged to have reached the endpoint. Afterwards, the product was subjected to suction filtration and rinsing with absolute ethanol, and finally dried under vacuum at 50 °C for 12 h to obtain Schiff base PMB powder.
2.2.2. Synthesis of PMBxCu1-WPU
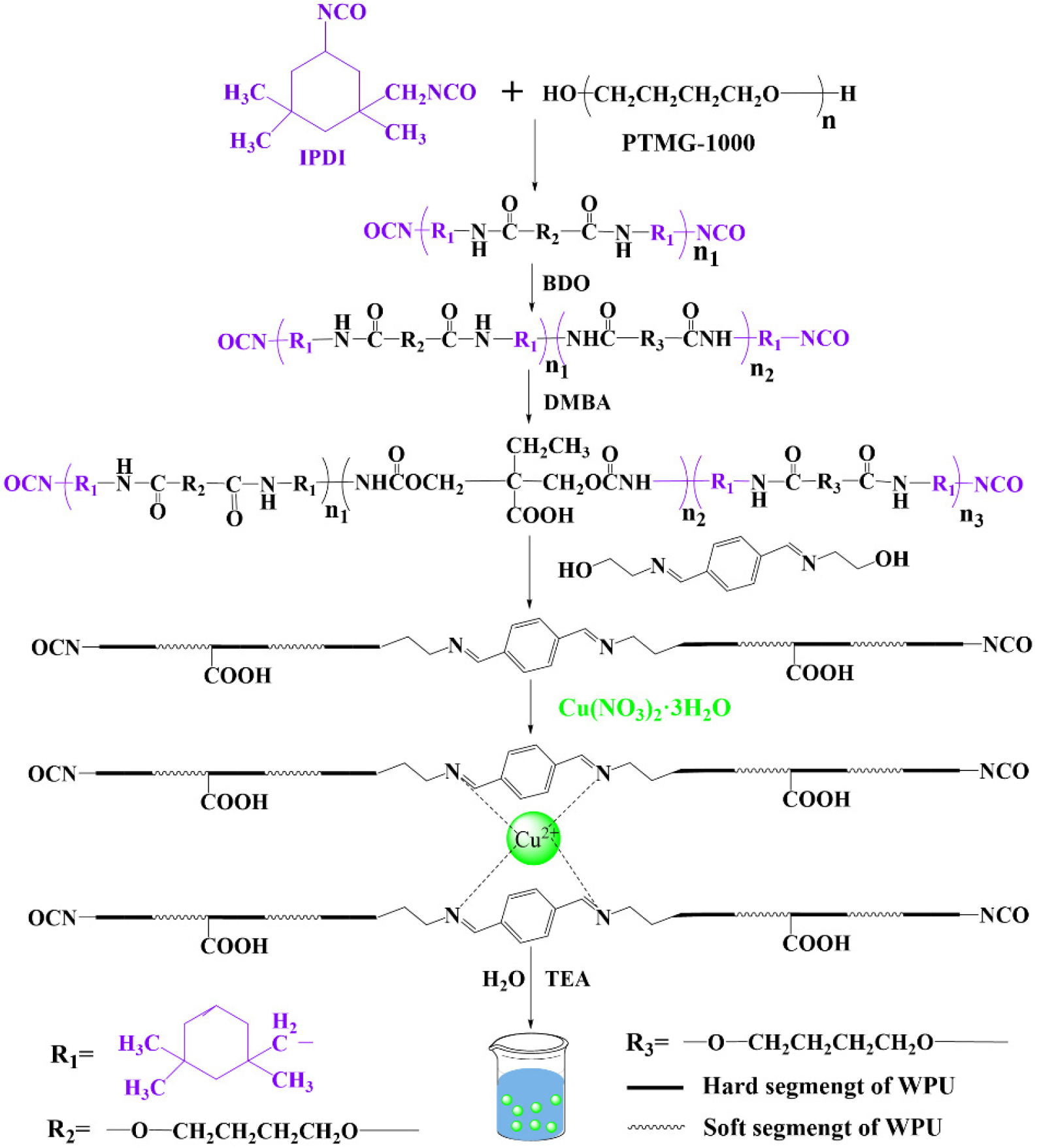
The synthesis of PMBxCu1-WPU was carried out using a modified acetone method, with the detailed procedure shown in Fig. 1. The entire reaction was performed under a nitrogen atmosphere, and the reaction apparatus employed was a four-necked flask equipped with a mechanical stirring system, a nitrogen protection device, a temperature monitor, and a reflux condenser with a desiccant. Firstly, the weighed IPDI and PTMG-1000 were added to the reaction flask, and the temperature was raised to 90 °C. The prepolymerization reaction was maintained for 2 h. After the completion of prepolymerization, the temperature of the system was lowered to 80 °C, and chain extender BDO and catalyst DBTDL were added sequentially. The reaction was continued for 2 h, during which acetone was added to adjust the viscosity of the system. The reaction progress was monitored in real-time by the standard dibutylamine titration method. When the content of isocyanate groups reached the theoretical value,13) DMBA was added for chain extension reaction. Subsequently, the ligand PMB was dissolved in acetone and then slowly dropped into the reaction system, followed by reaction at 80 °C for 2 h. Then, the acetone dispersion of Cu(NO3)2・3H2O was added, and the reaction was continued for 1 h. After the reaction, the temperature of the system was reduced to 40 °C, and TEA was added for neutralization for 0.5 h. Then, deionized water was slowly added under high-speed homogenization conditions for emulsification for 0.5 h. Finally, acetone was removed using a rotary evaporator under reduced pressure at 50 °C, yielding the target product PMBxCu1-WPU (x = 1, 2, 3).
In addition, for comparison with PMBxCu1-WPU (x = 1, 2, 3), WPU was synthesized under the same conditions without adding Schiff base and Cu(NO3)2・3H2O (Table 1, Fig. 2).
Table 1.
Formulations of PMBxCu1-WPU (x = 1, 2, 3).
2.3. Sample Preparation and Testing Methods
Films of WPU, PMB-WPU and PMBxCu1-WPU (x = 1, 2, 3) were prepared via the solution casting method, with the specific procedures as follows: The prepared emulsion was uniformly poured into a polytetrafluoroethylene mold, first allowed to stand and solidify at room temperature for 7 d. Subsequently, the preliminarily formed film was transferred to a vacuum drying oven and further dried at 60 °C for 24 h until the sample mass became constant.
A Nicolet 6700 Fourier transform infrared (FT-IR) spectrometer was used to record the infrared spectra of the sample films and powders in the range of 4,000-500 cm-1, with the number of scans set to 32. The structural composition of the samples was analyzed using an AVANCE III superconducting nuclear magnetic resonance (NMR) spectrometer; the fluorescence emission spectra of the sample powders and emulsions were determined using a Hitachi FL-4700 fluorescence spectrometer. Using a Malvern Nano-ZS nanoparticle size and zeta potential analyzer, the samples were diluted 1,000 times with ultrapure water, and three parallel tests were performed under constant temperature conditions (25 ± 0.1 °C). The average value was taken as the data for the particle size distribution and surface potential of the sample emulsions. The optical properties of the sample films were characterized using a Shimadzu SolidSpec-3700 ultraviolet-visible spectrophotometer in the wavelength range of 200-800 nm. Phase analysis was conducted using a Rigaku SmartLab 9 kW X-ray diffraction (XRD) (Japan) within the scanning angle range of 5-80° (2θ). A PerkinElmer STA8000 synchronous thermal analyzer was employed to test the thermal stability of the films in the temperature range of 30-700 °C under a nitrogen-protected atmosphere (flow rate: 50 mL/min) with a heating rate of 10 °C/min.
3. Results and Discussion
3.1. NMR and FT-IR of PMB
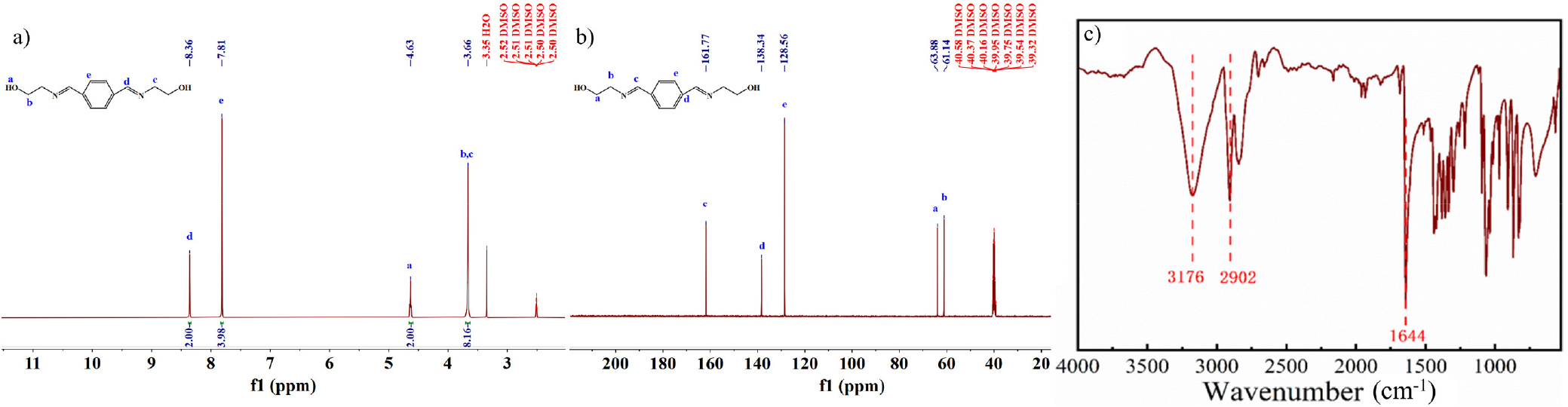
NMR spectra and FT-IR spectrum of PMB are shown in Fig. 3. Among them, Fig. 3(a) and Fig. 3(b) correspond to the 1H NMR and 13C NMR spectra of PMB, respectively, which clearly reflect the chemical environments of the relevant hydrogen and carbon atoms.
In Fig. 3(a), the absorption peak at 8.35 ppm is attributed to the hydrogen atom attached to the carbon in the C=N group; the signal at 7.81 ppm corresponds to the hydrogen atoms on the benzene ring; the absorption peak at 4.63 ppm originates from the active hydrogen of the hydroxyl group; and the signal at 3.65 ppm is caused by the hydrogen atoms attached to the carbon in the C-N bond. Fig. 3(b) shows that the absorption peak at 161.77 ppm is derived from the carbon atom in the C=N group; the signal at 38.34 ppm corresponds to the carbon atom in the benzene ring directly connected to C=N; the signal at 128.56 ppm comes from the carbon atoms in the benzene ring not connected to C=N; the absorption peak at 63.88 ppm is attributed to the carbon atom connected to the hydroxyl group; and the signal at 61.14 ppm is caused by the carbon atom in the C-N single bond. The above analysis of PMB’s NMR spectra clearly reveals the corresponding relationship between each absorption signal and the molecular structure.
In addition, in Fig. 3(c), the peak at 3,176 cm-1 is the absorption peak of the hydroxyl group, the peak at 2,902 cm-1 corresponds to the absorption peak of the methylene group, and the peak at 1,644 cm-1 is attributed to the absorption peak of the C=N group.14,15,16) Based on the comprehensive analysis of the above spectral characterization results, it can be fully confirmed that PMB has been successfully synthesized.
3.2. FT-IR Spectra of WPU, PMB-WPU and PMBxCu1-WPU
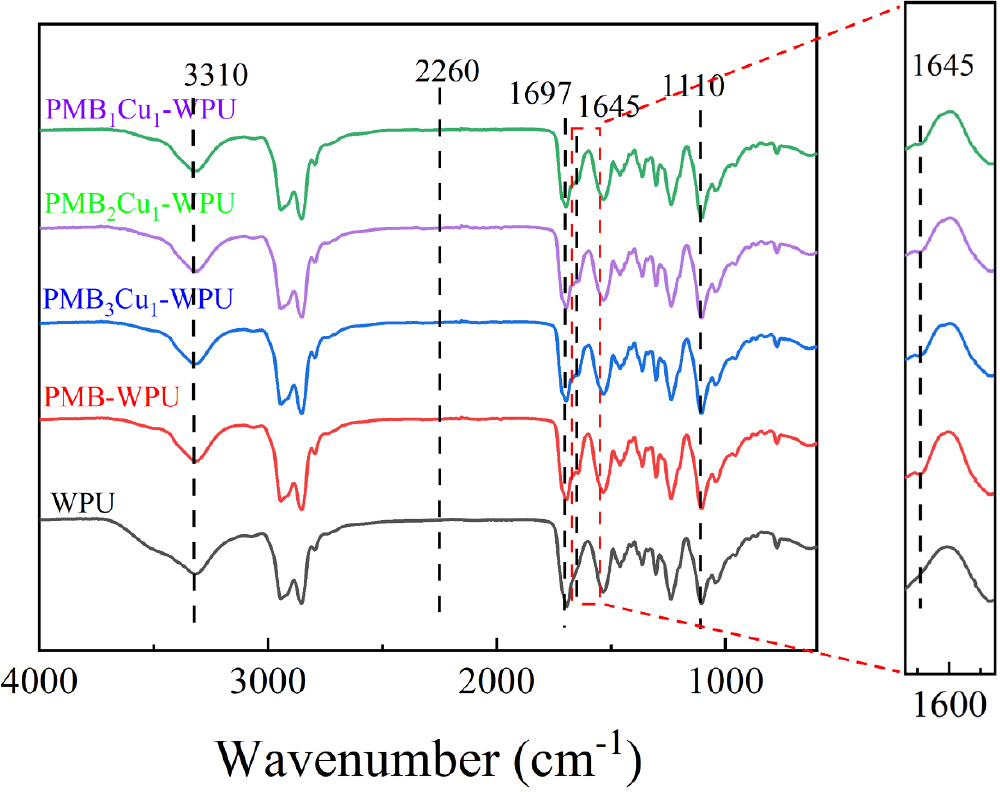
The FT-IR spectral results of WPU, PMB-WPU and PMBxCu1-WPU (x = 1, 2, 3) are shown in Fig. 4. The results indicate that WPU, PMB-WPU, and PMBxCu1-WPU, all exhibit typical characteristic absorption peaks of polyurethane: the broad absorption band at 3,310 cm-1 can be attributed to the stretching vibration of N-H bonds in carbamate groups; the multiple absorption peaks in the wavenumber range of 2,860-2,940 cm-1 originate from the stretching vibrations of C-H bonds in methylene and methyl groups within the molecular chain; the strong characteristic peak at 1,697 cm-1 corresponds to the stretching vibration of carbonyl groups (C=O); and the absorption peak around 1,110 cm-1 confirms the presence of ether bonds (C-O-C) in the molecular structure.17,18,19) The appearance of these characteristic peaks fully confirms the successful construction of the polyurethane structure. Meanwhile, no characteristic absorption peak of -NCO groups was detected around 2,260 cm-1, indicating that -NCO groups were fully involved in the reaction.
Compared with WPU, PMB-WPU and PMBxCu1-WPU (x = 1, 2, 3) show a new absorption band at 1,645 cm-1, which corresponds to the characteristic vibration of C=N bonds in Schiff base ligands. Notably, with the increase in the amount of Cu(NO3)2・3H2O added, the intensity of this absorption peak gradually decreases. This phenomenon indicates that PMB ligands have been successfully bonded to the polyurethane molecular chain, and Cu(II) have formed coordination bonds with the imine nitrogen atoms in the ligands.20) Based on the comprehensive analysis of the above infrared spectral characterization results, it can be fully confirmed that PMBxCu1-WPU has been successfully synthesized.
3.3. XRD Analyses
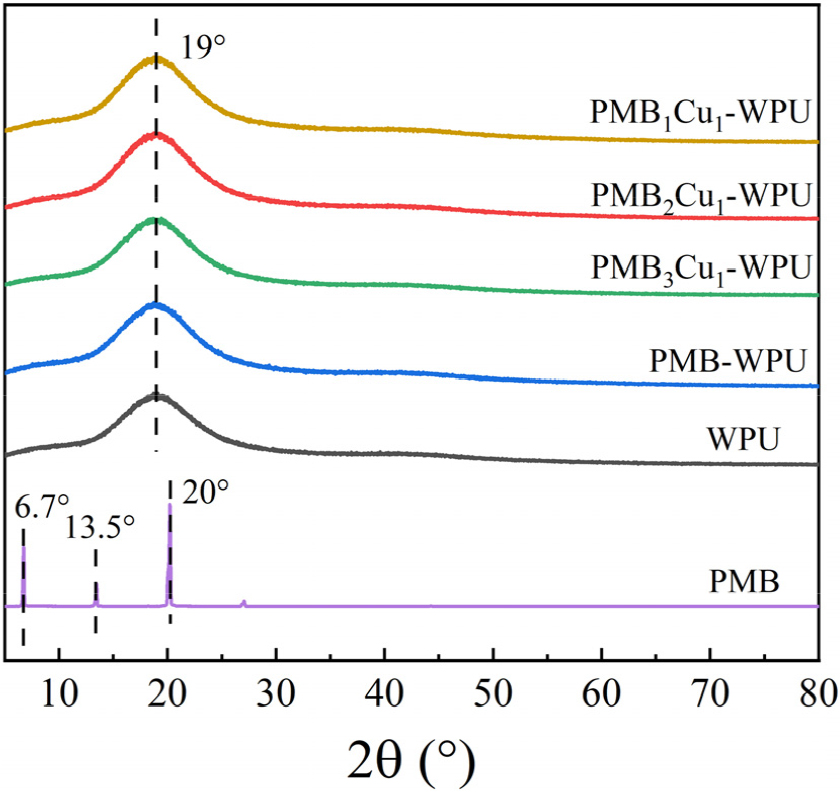
Fig. 5 shows the XRD patterns of PMB, WPU, and PMBxCu1-WPU (x = 1, 2, 3). The results indicate that PMB exhibits multiple sharp diffraction peaks in the low-angle region, a characteristic that fully confirms its good crystallinity.21) In sharp contrast, the PMB-WPU, PMBxCu1-WPU (x = 1, 2, 3), and WPU only exhibit a broadened diffuse diffraction peak near 2θ≈19°. In the XRD patterns of PMBxCu1-WPU (x = 1, 2, 3), the characteristic diffraction peaks of PMB and Cu(NO3)2・3H2O are absent. Moreover, a broadened peak shape persists across all samples, indicating that PMB, Cu(NO3)2・3H2O, and polyurethane components are uniformly dispersed at the molecular level within the polyurethane matrix, thereby forming a homogeneous phase and demonstrating good compatibility. This can be attributed to the successful coordination of Cu(II) with the Schiff base (PMB) incorporated into the polyurethane molecular chain via chemical bonding. Such coordination effectively prevents phase separation or particle aggregation, providing structural evidence for the material’s homogeneity and indirectly verifying the effectiveness of the synthesis process in regulating the material’s microstructure.22)
3.4. Emulsion Particle Size and Zeta Potential Analysis
Dynamic light scattering (DLS) was employed to investigate the colloidal properties of WPU, PMB-WPU and PMBxCu1-WPU (x = 1, 2, 3) emulsions, as presented in Table 2. The test results show that the particle size of the emulsions increases significantly with the increase in the dosage of Cu(NO3)2・3H2O. This phenomenon can be attributed to the coordination of Cu(II) with PMB ligands in the polyurethane molecular chains, which promotes the formation of crosslinking structures between polymer chains, thereby leading to a gradual increase in the molecular weight of the system.23)
Table 2.
Emulsion particle size, zeta potential and appearance.
Notably, the absolute values of zeta potentials for all samples exceed 40 mV. According to the colloid stability theory, this value indicates that the emulsion systems exhibit excellent dispersion stability, ensuring uniform dispersion of nanoparticles in the aqueous phase and effectively inhibiting particle aggregation. Such good stability is of great significance for the performance of PMBxCu1-WPU (x = 1, 2, 3) in practical applications, providing a reliable theoretical basis for its potential applications in fields such as coatings and biomedicine.24)
3.5. TG-DTG of WPU, PMB-WPU and PMBxCu1-WPU
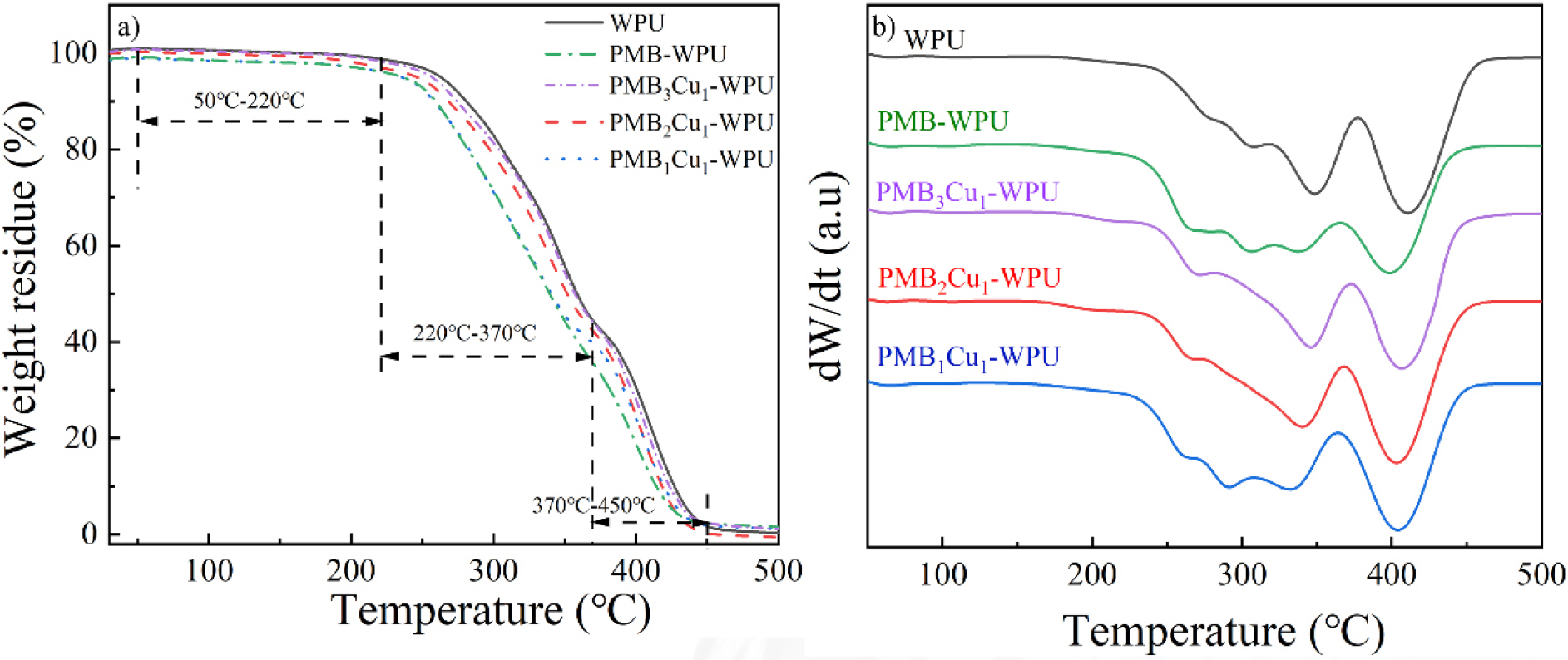
The thermogravimetric (TG) and derivative thermogravimetric (DTG) curves of WPU, PMB-WPU and PMBxCu1-WPU (x = 1, 2, 3) films are shown in Fig. 6(a) and Fig. 6(b), respectively. The results indicate that all exhibit three distinct decomposition stages.25,26,27)
The mass loss in the first stage (50-220 °C) is mainly attributed to the volatilization of residual solvents and adsorbed water in the samples. The second stage (220-370 °C) primarily corresponds to the degradation of the hard segment structure in polyurethane materials. During this process, urethane groups and allophanate bonds undergo cleavage, and the thermal decomposition behavior is closely related to the thermal stability of polar groups in the polymer. The third stage (370-450 °C) involves the cracking of the soft segment carbon chains.
Further analysis reveals that the reaction between -OH groups in PMB molecules and isocyanate groups significantly increases the proportion of allophanate bonds formed in PMB-WPU series materials. The reaction between -OH groups and isocyanate groups promotes the formation of more urea bond structures in the polymer network, which in turn leads to a slight decrease in the onset temperature of the second thermal decomposition stage. This phenomenon confirms that the complex has been successfully bonded to the polyurethane molecular chain.28) Meanwhile, compared with WPU, the introduction of PMB leads to a higher residual carbon yield of PMB-WPU after high-temperature carbonization. It is noteworthy that the initial decomposition temperature of PMBxCu1-WPU remains around 220 °C, indicating that the introduction of Schiff base Cu(II) complexes does not significantly affect the thermal stability of the material, and the PMB-WPU series materials still possess excellent thermal stability.
3.6. UV-Vis Spectra of PMB, WPU, PMB-WPU and PMBxCu1-WPU
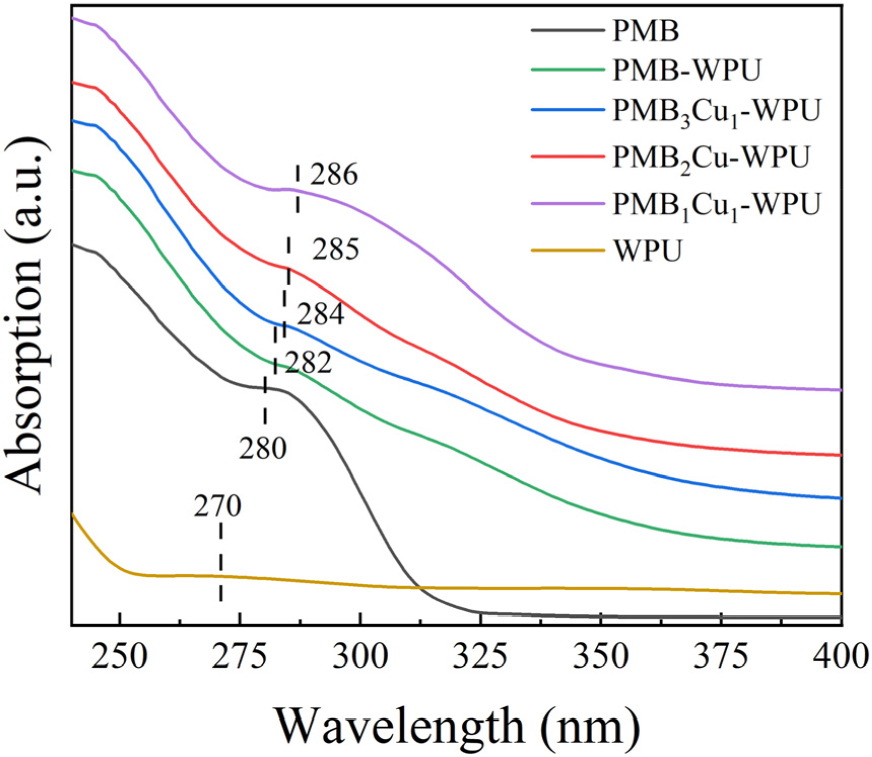
Fig. 7 shows the UV-Vis spectra of WPU, PMB, and PMBxCu1-WPU (x = 1, 2, 3). It can be seen from the figure that WPU has a weak absorption peak at 270 nm, which corresponds to the n-σ* transition absorption of the C-N bond in the urethane bond.29,30) PMB exhibits a strong absorption peak at 280 nm, which is related to the π-π* transition of the conjugated structure of PMB.31,32)
When PMB is incorporated into the polyurethane chain, the absorption peak redshifts to 282 nm. With the increase in the degree of coordination between Cu(II) and PMB-WPU, the absorption peak redshifts from 282 nm to 286 nm. This is because the coordination of Cu(II) alters the electron distribution of the conjugated structure of PMB,33,34) indicating that Cu(II) has successfully coordinated with PMB.
3.7. Fluorescence Spectroscopy of PMB, WPU and PMBxCu1-WPU
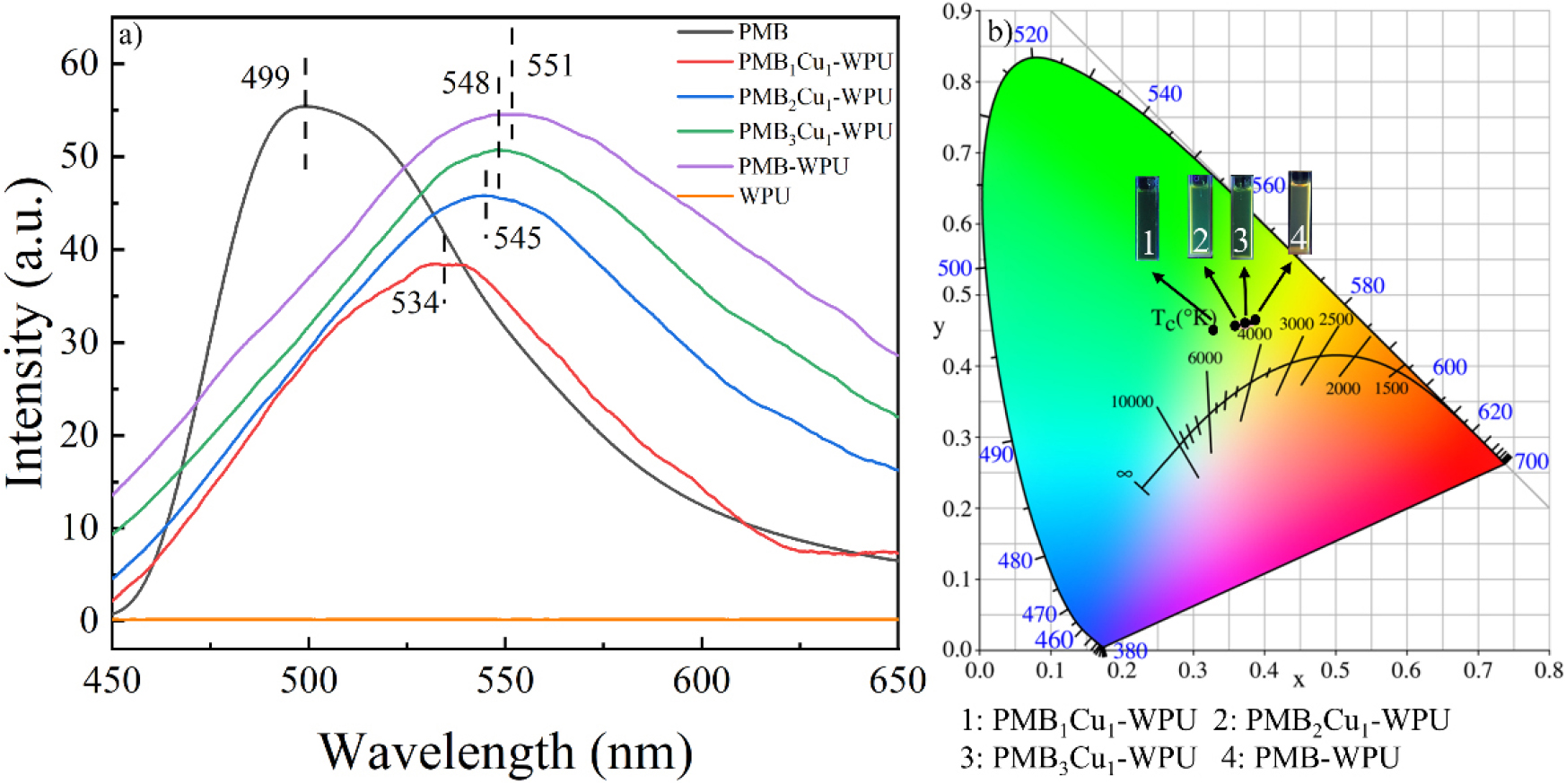
Fig. 8(a) and Fig. 8(b) show the fluorescence spectra and CIE coordinate diagrams of WPU, PMB, PMB-WPU and PMBxCu1-WPU (x = 1, 2, 3), respectively. Among them, WPU basically has no fluorescence properties. Under the excitation of 360 nm ultraviolet light, PMB shows a fluorescence emission peak at 499 nm, which originates from the π-π* transition of the C=N bond in the PMB molecule.35,36,37) It is worth noting that compared with PMB, the emission peak of PMB-WPU has a significant red shift, shifting to 551 nm, with a red shift amplitude of 52 nm. The cause of this phenomenon may be that after PMB is incorporated into the WPU chain, it not only expands its own conjugated system but also changes the polarity of the microenvironment where it is located. Through the above dual effects, the electronic structure and energy level distribution of the molecule are changed, and finally the energy difference of the luminescent transition is reduced.
It is worth noting that with the increase in the degree of coordination between Cu(II) and PMB-WPU, the fluorescence emission peaks of PMB-WPU and PMBxCu1-WPU (x = 1, 2, 3) blue-shifted from 551 nm to 534 nm. This is because Cu(II) coordinates with the N atoms on the PMB molecule, which changes the conjugation effect of the molecule, causes a change in the charge distribution of the ligand, and reduces the chromogenic ability of the chromophore,38,39,40) which further confirms that Cu(II) has successfully coordinated with PMB.
4. Conclusion
In this study, Schiff base Cu(II) complexes were successfully introduced into the main chain of WPU, resulting in the preparation of fluorescence-controllable PMBxCu1-WPU (x = 1, 2, 3). Structural characterization confirmed the successful synthesis of the Schiff base ligand PMB, as well as its stable coordination with Cu(II) and incorporation into the polyurethane molecular chain. Within PMBxCu1-WPU, PMB and Cu(II) achieved molecular-level uniform dispersion, and the emulsion exhibited excellent dispersion stability along with good thermal stability (with an initial decomposition temperature of approximately 220 °C). Moreover, its optical properties could be regulated by the coordination degree of Cu(II): the ultraviolet-visible absorption peak redshifted with increasing coordination, and the fluorescence emission peak underwent a red shift from 499 nm (for PMB) to 551 nm (for PMB-WPU) and further to 534 nm (for PMB3Cu1-WPU), realizing precise fluorescence regulation over a broad wavelength range. This research provides a new approach and foundation for the application of high-performance fluorescence-tunable waterborne polyurethane materials in fields such as intelligent anti-counterfeiting and biomedicine.