1. Introduction
For the past decade, a major global trend has been the search for sustainable and super-efficient alternative energy as a replacement for finite fossil fuels. In this setting, electrochemical and photocatalytic water splitting reactions have received much interest in recent years due to their importance in various energy conversion and storage technologies like hydrogen production, oxygen production, and regenerative fuel cells, among others.1,2) The OER is the kinetic bottleneck of water splitting in these systems. Because of the four-electron transfer process and slow kinetics, OER is one of the most significant and technologically hard reactions in electrochemical water splitting research.3) As a result, the effectiveness of OER is restricted by the need for a significant overpotential, and the cell voltage is determined by the anodic reaction. By reducing cathodic overpotentials and using appropriate and effective electrocatalysts, the performance and efficiency of electrochemical water splitting can be greatly increased in OER.4,5) Even though RuO2 and IrO2 are highly active OER catalysts with the lowest OER overpotentials, their high cost prevents the widespread use of precious and expensive materials.6) To date, a variety of compounds derived from non-noble metals, such as metal oxides, (oxy)hydroxides, chalcogenides, and phosphates, have been produced as efficient OER catalysts or precatalysts.7,8) However, material improvements are required to replace such precious-metal-based metal-based electrocatalysts for them to function under hard conditions and in large-scale production.9,10) The common drawbacks of these reported electrocatalysts include a small number of active sites, inefficient electrical transport, inefficient electrical contact with the electrolyte, and instability under operating conditions, necessitating chemical exfoliation or functionalization with large surface area compounds.11,12)
Currently, incorporating the Fe element into the catalyst has been proved as an effective method of increasing the active site's reactivity. Because of the synergistic effects between Cu/Co and the introduced Fe metal, the electronic structure of the host material can be optimized, resulting in a considerable increase in electrical conductivity and the exposure of more active sites.13) In addition to composition control, architectural integrity is important in improving the catalytic performance of transition metalbased catalysts. The 2D, nanosheet structure, which is popular in the structural design of many catalysts, is also appealing in the design of high-performance OER catalysts.14) The nanosheet's huge specific surface area provides materials with a lot of active sites, which helps with the multistep process in OER. Meanwhile, the nanosheet structure can supply OER with a short proton-coupled electron transfer channel, increasing diffusion efficiency.15)
In light of these considerations, we present the hydrothermal synthesis to produce Fe-doped CuCo(OH)2 nanosheets supported on nickel foam (NF). The introduction of Fe, as well as the nanosheet shape, allows Fe-doped CuCo(OH)2 to exhibit low overpotential (260 mV at 50 mA cm−2) for OER, which is significantly lower than the overpotential of a bare NF catalyst (460 mV) and certain other previously reported TMSs. For comparison, nanosheets of CuCo(OH)2 have been synthesized using the same experimental procedure as Fe-doped CuCo(OH)2 and performed in the same OER studies. In all tests, it was found that Fe-doped CuCo(OH)2 performs significantly better as an OER catalyst than CuCo(OH)2, and bare NF as an OER catalyst.
2. Experimental
2.1. Synthesis of Fe-doped CuCo(OH)2
2 mmol of Co(NO3)2-6H2O, 1 mmol of Cu(NO3)2-3H2O, and 0.5 mmol of Fe(NO3)3-9H2O were dissolved in 30 mL of pure distilled water in a conventional synthesis technique. After 10 minutes of strong stirring, 4 mmol of urea was added to the solution, which turned pink. After 15 minutes of vigorous stirring, the solution was ready. Approximately two milliliters of ethylenediamine were added to this solution, and the color of the solution changed to brown. The solution was then transferred to a 50 mL Teflon-lined autoclave. Precleaned pieces of NF with HCl, ethanol, acetone, and water were dipped into the solution. Then the autoclave was sealed and maintained at 200 °C for 12 hours. After that, the reaction mixture was allowed to cool to room temperature to complete the reaction. The final product, which had a dark black color, was washed twice with distilled water and ethanol before being dried at 50 °C for 6 hours and then stored for further investigation. CuCo(OH)2 was also synthesized in a manner similar to that described before for Fe-doped CuCo(OH)2 nanosheets, with the exception that no Fe salt was added during the synthesis of CuCo(OH)2.
2.2. Physical characterization
The X-ray diffraction (XRD) measurements were recorded on an X’Pert PRO Multipurpose X-Ray Diffractometer with monochromatized Cu kα radiation (λ = 1.54 Å). Scanning electron microscopy (FESEM, Gemini 500 EDS Oxford) was employed to observe the morphology of the samples. X-ray photoelectron spectroscopy (XPS) scans were conducted on a multifunctional imaging electron spectrometer (Thermo ESCALAB 250XI) with a monochromatic aluminum light source.
2.3. Electrochemical measurements
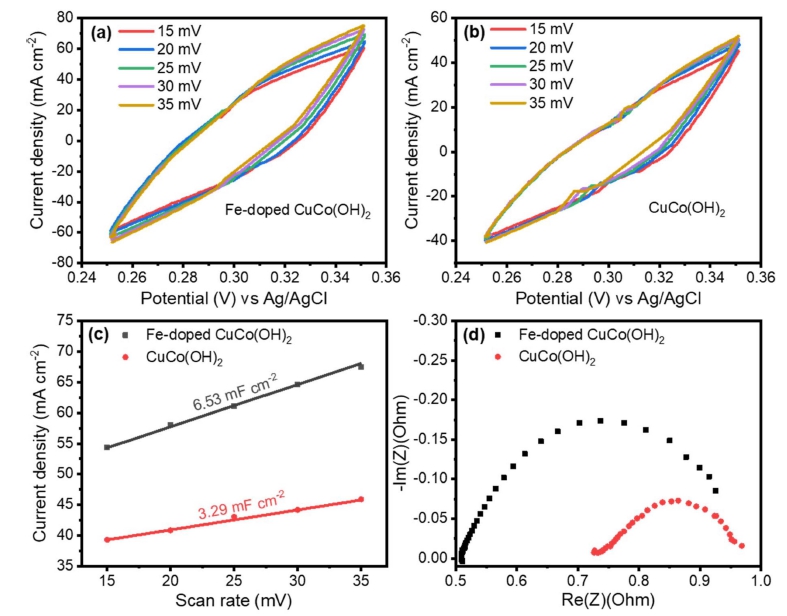
The OER measurements were performed in 1 M KOH using a three-electrode system on a CHI660E workstation (Shanghai, Chenhua). The reference electrode was Ag/ AgCl, and the counter electrode was a platinum wire. Freshly prepared Fe-doped CuCo(OH)2 was used as a working electrode. Linear sweep voltammetry (LSV) was carried out at a scan rate of 1 mV s–1 in a potential window of 0 ~ 0.7 V. All reported potential values were converted to reversible hydrogen electrode (RHE) potentials using the following equation: ERHE = EAg/AgCl + 0.197 + 0.059 pH. Cyclic voltammetry (CV) was used to measure the electrochemical double-layer capacitance (Cdl) of catalysts at various scan rates (15, 20, 25, 30, and 35 mV s−1) ranging from 0.25 to 0.35 V vs Ag/ AgCl. The Cdl is the slope of the line of current density plotted against scan rate at 0.34 V vs Ag/AgCl. The electrochemically active surface (ECSA) was determined using Cdl according to eq.
ECSA = Cdl / CS
CS denotes the catalyst's double-layer capacitance on a unit surface area, which is typically assumed to be 0.04 mF cm−2. Electrochemical impedance spectroscopy (EIS) was carried out in a frequency range of 105 Hz to 0.1 Hz with an amplitude of 10 mV. The catalyst's OER stability was tested for 20 hours using chronopotentiometry (CP) at a constant current density of 10 mA cm–2.
3. Results and discussion
Fig. 1(a-c) shows FE-SEM images of Fe-doped CuCo(OH)2 at various magnifications, revealing that the Fe-doped CuCo(OH)2 has a 2D-nanosheet-like morphology. Fig. 2a shows the XRD spectrum of Fe-doped CuCo(OH)2. The (111), (200), and (220) planes of the NF substrate are responsible for the strong diffraction peaks at 44.8o, 52.2o, and 76.8o, respectively. The diffraction peaks indexed 002, 111, 022, and 130 planes are of Cu(OH)2 (JCPDS 80-0656).16,17) The first doublet, measured at 932.7 and 933.8 eV, and the second at 952.3 and 953.8 eV, are attributed to Cu 2p3/2 and Cu 2p1/2 core levels, respectively [Fig. 2(b)]. The presence of the Cu2+ oxidation state is confirmed by the presence of very strong shakeup satellite peaks at 942.5 and 962.3 eV, as well as corresponding mainline peaks at 933.5 and 953.5 eV, respectively; the satellite feature attests to the presence of the Cu2+ oxidation state.7) The core level of the Co 2p spectrum was deconvoluted into two spin-orbit doublets and two shakeup satellites in high-resolution XPS spectra of Fedoped CuCo(OH)2 [Fig. 2(c)]. The two prominent peaks at binding energies of 780.7 and 795.8 eV, correspond to the Co 2p3/2 and Co 2p1/2 spin-orbit peaks, respectively, confirming the existence of two types of cobalt oxidation states: CO2+ and Co3+.12) As shown in Fig. 2(d), the peak at ~ 712.6 with a satellite peak at 716.5 eV is attributed to Fe 2p3/2. Meanwhile, the peak at ~ 725.3 eV is attributed to Fe 2p1/2.15) The O 1s spectra in Fig. 2(e) demonstrate two peaks, one at 530.2 which corresponds to metal oxide bond as well and another peak at 532.4 eV can be attributed to metal hydroxide bonding in Fe-doped CuCo(OH)2.3)
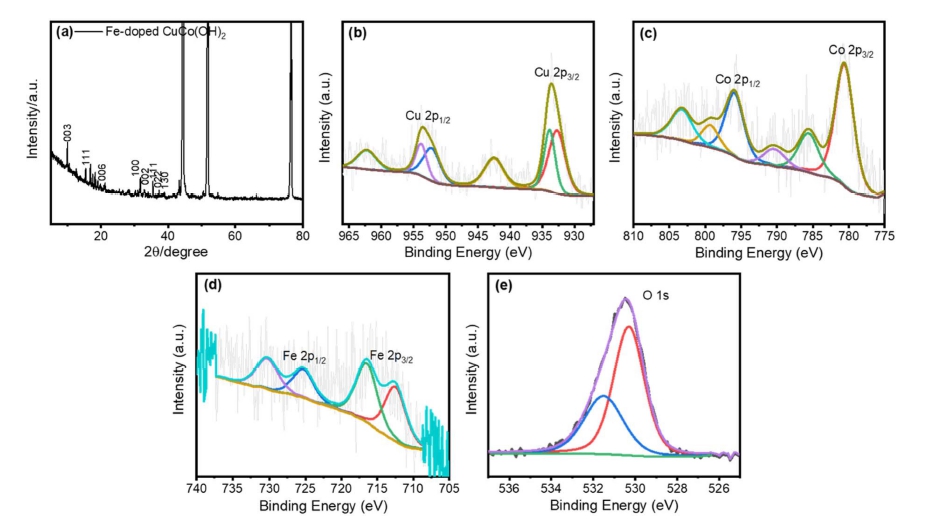
Fig. 2
(a) XRD spectrum of Fe-doped CuCo(OH)2, (b-e) High resolution XPS spectra of Cu 2p, Co 2p, Fe 2p, and O 1s of Fe-doped CuCo(OH)2 respectively.
The electrocatalytic performance of Fe-doped CuCo(OH)2, CuCo(OH)2, and bare BF toward the OER was tested in a 1 M KOH aqueous solution. The blank NF exhibits poor OER activity, as shown by the LSV curves [Fig. 3(a)], requiring an overpotential of 460 mV to deliver a current density of 50 mA cm−2. It is worth noting that the OER performance of CuCo(OH)2 is better than that of bare NF, with a 320 mV overpotential to deliver a current density of 50 mA cm−2. Surprisingly, when Fe-doped CuCo(OH)2 is grown on NF, the catalytic performance toward OER improves significantly requiring an overpotential of 260 mV to reach a current density of 50 mA cm−2. The Tafel slope is then calculated in order to investigate the reaction kinetics of the catalysts. Among the catalysts, Fe-doped CuCo(OH)2 has the lowest Tafel slope of 72.9 mV dec−1, suggesting superior OER reaction kinetics to pristine CuCo(OH)2 (80.6 mV dec−1) and bare NF (90.3 mV dec−1) [Fig. 3(b)]. The overpotential and Tafel values of all electrocatalysts are displayed in Fig. 3c. Additionally, the long-term stability of Fe-doped CuCo(OH)2 was evaluated. The potential to deliver the current density of 10 mA cm−2 slightly changed during the continuous evolution of gas for 20 h [Fig. 3(d)], confirming the excellent electrocatalytic performance toward water oxidation.
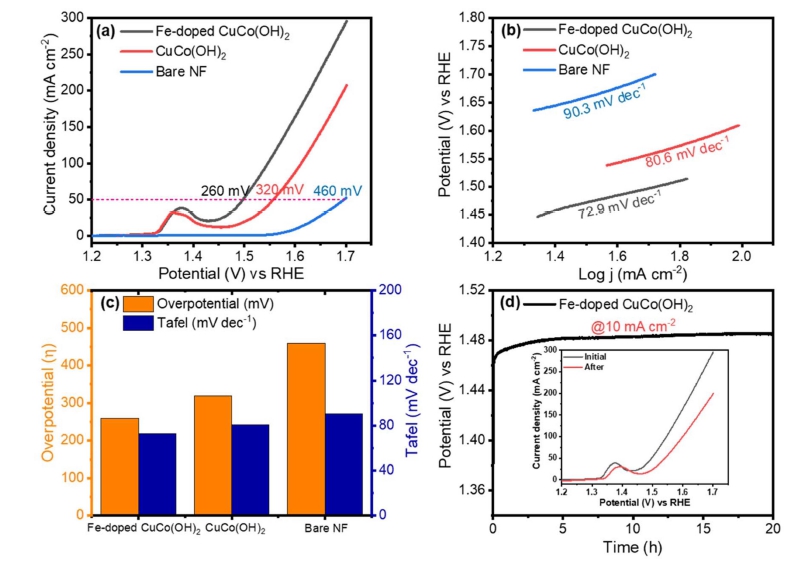
Fig. 3
OER performance of the catalysts (a) LSV curves, (b) Corresponding Tafel slopes, (c) Comparative bar chart of overpotential and Tafel values of all catalysts, (d) Stability of Fe-doped CuCo(OH)2 and inset is the OER curves before and after long term stability.
To understand the origin of the high OER activity of Fe-doped CuCo(OH)2, the corresponding Cdl was measured via CV tests [Fig. 4(a-c)] which was an important parameter to measure the electrochemically active surface area (ECSA). The Cdl values for Fe-doped CuCo(OH)2, and CuCo(OH)2, are 6.53, and 3.29 mF cm−2, respectively. Additionally, according to the above equation for calculation of ECSA, Fe-doped CuCo(OH)2 owns the largest ECSA of 163.25 cm−2 than CuCo(OH)2 (82.25 cm−2). In addition, electrochemical impedance spectroscopy (EIS) was used to determine the charge transfer efficiency [Fig. 4(d)]. Fe-doped CuCo(OH)2, and CuCo(OH)2 have charge transfer resistances (Rct) of 0.516, and 0.734 Ω, respectively. The lowest Rct of Fe-doped CuCo(OH)2 reveals the fastest electron transfer rate on the catalyst-electrolyte surface and the most efficient OER reaction kinetics, which may be linked to the incorporation of Fe into the pristine material.
4. Effect of Fe-doping
Owing to the advantage of the nanosheet structure and high average valence, the Fe-doped CuCo(OH)2 is bound to have superior electrochemical performance. As shown in Fig. 4(c), the Cdl value of Fe-doped CuCo(OH)2 is 6.53 mF cm−2 which is almost double that of CuCo(OH)2 (6.53 mF cm−2), suggesting that the Fe incorporation is beneficial to increasing the ECSA of the catalyst. The conductivity also plays a decisive role in OER activity, so the electrochemical impedance spectra of the catalysts are tested at an overpotential of 300 mV. The charge transfer resistance (Rct) of Fe-doped CuCo(OH)2 is only 0.516 Ω, which is much smaller than those of CuCo(OH)2 (0.734 Ω). Moreover, the LSV curves as well as corresponding Tafel plots of Fe-doped CuCo(OH)2, and CuCo(OH)2 are displayed in Fig. 3(a,b). The Fe-doped CuCo(OH)2 possesses excellent OER performance. It only needs a 260 mV overpotential to afford 50 mA cm−2 w ith a Tafel slope of 72.9 mV dec−1, which is much better than CuCo(OH)2 (320 mV, 80.6 mV dec−1).
5. Conclusion
In summary, we synthesized Fe-doped CuCo(OH)2 nanosheets for the first time using a simple hydrothermal method, demonstrating a remarkable electrolytic OER performance in 1.0 M KOH with a high current density (50 mA cm−2) at the lowest overpotential (260 mV) and a decreased Tafel slope (72.9 mV dec−1) indicating accelerated reaction kinetics. The resulting catalyst demonstrated long-term stability with continuous durability for a maximum of 20 hours with a very small deviation. Under difficult industrial conditions, this catalyst can maintain unaffected performance. The excellent OER activity is attributed to the incorporation of Fe into the CuCo(OH)2, which provides an abundance of electrochemically active sites and high conductivity. This work paves the way for further research into iron-cobalt-copper-based electrodes as a stable and advanced OER electrocatalyst.