1. Introduction
2. Experimental Procedure
2.1. Plant material
2.2. Synthesis of Fe2O3
2.3. Fe2O3 nanoparticles characterization
2.4. Antibacterial activity evaluation
3. Results and Discussion
3.1. Fe2O3 structural characterization
3.2. Morphological characterization
3.3. EDX (energy dispersive X-ray spectroscopy) of Fe2O3 nanoparticles
3.4. FTIR Analysis of Fe2O3 nanoparticles
3.5. Antibacterial activity
4. Conclusion
1. Introduction
One of the main important threats to human and organismal health is medicine resistance, which is treated as a global health difficulty, because traditional treatment methods often use poisonous chemicals and non-biodegradable materials, which make these materials harmful to the health of living things, raises environmental concerns and limits their clinical uses. However, the use of dangerous substances in chemical compositions limits the likelihood of their medical application. To address these challenges, chemicals are replaced with plant-based materials and manufactured nanoparticles which represent a sustainable and environmentally friendly approach.1,2) Plant extracts, which contain bioactive molecules such as phenolic chemicals, terpenoids and flavonoids, it is easy to manufacture, low cost and environmentally friendly.3) That has prompted many scientists and researchers to develop alternative strategies to combat bacterial infections.4,5) Nanoparticles, especially those made from plant extracts, have gained interest as a sustainable and biocompatible alternative to conventional methods. One specific plant is the rose of Maui. Rose of Maui, a flowering plant with medical and therapeutic properties, is a potential candidate for the synthesis of nanoparticles due to its bioactive compounds, in the way that antimicrobial, antioxidant and anti-inflammatory activities.6,7,8) Iron oxide nanoparticles, particularly Fe2O3, have shown antimicrobial activity against different bacterial strains.9) This study explores the synthesis of Fe2O3 nanoparticles using chemically prepared flower plants and investigates their potential antibacterial activity.10,11)
Iron oxide nanoparticles possess antibacterial properties due to their extreme surface area, redox potential and size-dependent reactivity.12,13,14) Plant doping with iron nitrate was used as a precursor for the production of iron oxide nanoparticles because it reveals the morphological and structural properties of the nanoparticles and their antibacterial effectiveness against harmful bacteria.15,16,17,18) The detection results could lead to cheap and environmentally friendly ways to produce iron oxide nanoparticles, which may pave the way for new biomedical applications such as water treatment, implant coatings, and wound dressings.19,20)
This study aims to generate and screen isolated iron oxide nanoparticles (IONPs) extracted from chemically treated rose of Maui plants to explore their potential uses as antibacterial agents as shown in Fig. 1. Chemical synthesis will be used to generate IONPs, and different analytical methods will be used to characterize the final nanoparticles. A range of bacterial strains will be used to evaluate the antibacterial efficacy of the nanoparticles. The structural and chemical properties of the produced IONPs will be evaluated using Fourier transform infrared (FTIR) spectroscopy, transmission electron microscopy (TEM), and X-ray diffraction (XRD). The antibacterial efficacy of the IONPs against Escherichia coli (E. coli) and Staphylococcus aureus will also be evaluated.
2. Experimental Procedure
2.1. Plant material
Borago officinal is a type of plant belonging to the genus of sage, to the sage family. It is a plant that grows in dry pastures and wastelands. It also grows on the edges of roads and does not tolerate high temperatures.21) It resembles garden sage, and has wild branches, with blue flowers, approximately 46 cm long that blooms in the spring. As shown in Fig. 1(a).
2.2. Synthesis of Fe2O3
Fresh Maui rose herbs were cleaned and drained, ground into powder as in Fig. 1(b), and the plant material was extracted using a solvent for the purpose of extracting Fe2O3 nanoparticles. The extracted flowers were used as a precursor for the synthesis of Fe2O3 nanoparticles. The size and shape of the nanoparticles were achieved through precise control and regulation of reaction parameters such as temperature and reaction time. The process involved mixing 100 mL of distilled water with 2 grams of dried hibiscus plant material, heating it to a temperature of 60~80 °C, filtering it twice through a cotton filter, and refining it. High-purity analytical iron nitrate [Fe(NO3)3] was used as the chemical. The green synthesis of the NPs solution was observed through color transformation after 1 h of mixing at a temperature of 60~80 °C using a magnetic stirrer. We note from Fig. 2 when an iron nitrate solution is added to a rose water solution, the solution’s color changes to green and subsequently to brown.
The chemical reagent used in this synthesis was Fe(NO3)3, chosen for its efficiency in nanoparticle production. The green synthesis process was visually monitored through distinct color transformations, serving as indicators of the reaction's progress. Specifically, the mixture exhibited a noticeable color change after 1 h of mixing at 60~80 °C, facilitated by continuous stirring with a magnetic stirrer. As illustrated in Fig. 2, the addition of the iron nitrate solution to the rose water extract triggered a sequence of color changes. Initially, the solution turned green, signifying the onset of nanoparticle formation. Over time, the solution further transformed to brown, confirming the successful synthesis of Fe2O3 nanoparticles. These visual cues reflect the reduction and stabilization processes inherent in green nanoparticle synthesis.
2.3. Fe2O3 nanoparticles characterization
The synthesized Fe2O3 nanoparticles were characterized using various analytical methods, including XRD analysis, to determine the crystalline structure and phase purity of the nanoparticles. Scanning electron microscopy (SEM) was used to observe the morphology and size distribution of the nanoparticles. FTIR was working to identify the functional groups present in the nanoparticles. The antibacterial activity of the nanoparticles was evaluated using the agar diffusion system against pathogenic bacteria.22,23,24)
2.4. Antibacterial activity evaluation
The antibacterial activity of the Fe2O3 nanoparticles will be assessed against a panel of bacterial strains, including both gram-positive and gram-negative bacteria. Standard microbiological methods, such as the agar-well diffusion method or broth dilution method, will be employed to determine the minimum inhibitory concentration (MIC) and zone of inhibition values.25,26,27)
3. Results and Discussion
3.1. Fe2O3 structural characterization
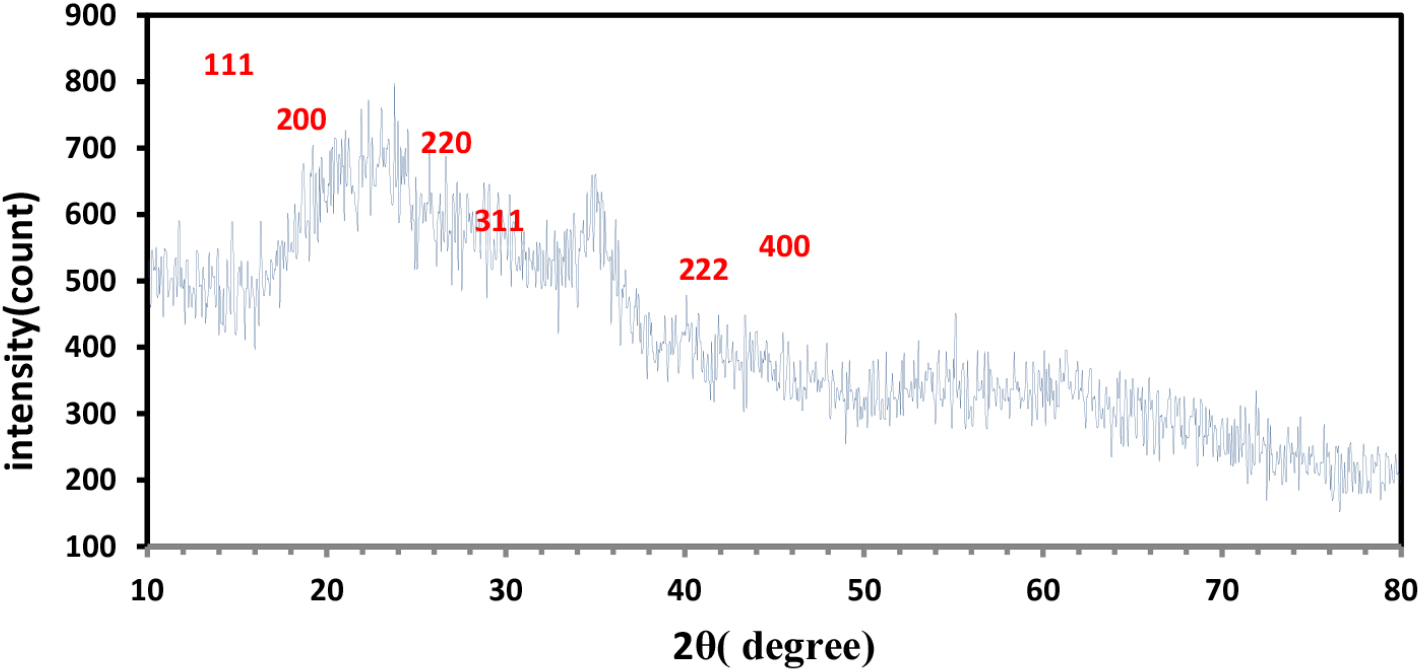
The XRD technique was used to characterize and detect the XRD pattern of the synthesized Fe2O3 nanoparticles where XRD was studied using a Philips PW 1730 XRD device made in the Netherlands, with parameters: scan type pre-set time, start position (2θ) (10.0400), end position (2θ) (79.99), step size 2θ (0.0500) with operating condition under measurement temperature (25οC) using anode material CU, K-Alpha 1.54060 Å, K-Beta 1.39225 Å.
The results shown in Fig. 3 showed distinct diffraction peaks consistent with the hematite phase. Using the width and diffraction angle of the peaks, the crystal size was calculated using the Scherer equation according to the formula of Eq. (1) mentioned below.28,29,30)
where, Ds (nm) is the crystallite size by Scherer, θ is Bragg’s angle in radian unit, K is the factor of Scherrer ≈ 0.9, λ X-ray wavelength = 1.54060 Å, β is a line broadening at full-width half maximum peaks in radian unit.31,32)
The crystal size of the crystal was calculated. The crystalline size was found to be approximately 0.085~0.166 nm, which indicates the formation of particles. The small crystalline size which has a role in improving the properties of antibiotics, as the results showed that reducing the crystal sizes has a significant effect on the ability of antibiotics to kill harmful bacteria in the bodies of living organisms. Results of crystal size calculation are listed in Table 1. When analyzing the XRD pattern, we can observe that the dominant peak appears at 27.85° (2θ) with an intensity of 650 units, corresponding to the (200) crystal plane. This indicates that F2O3 nanoparticles possess a large amount of crystal planes oriented in the (200) direction. In addition, there are other peaks observed at 23.7° (2θ), 35.13° (2θ), 42.07° (2θ), 50.92° (2θ), and 56.42° (2θ), corresponding to (111), (220), (311), (222), and (400) crystal planes, respectively. These peaks indicate the presence of multiple crystallographic orientations within the Fe2O3 nanoparticles.
Table 1.
Results of crystallite size Ds, intensity, Miller indices, FWHM and d-spacing by using Scherrer method.
|
2θ (degrees) |
Intensity (counts) |
Ds (nm) | Miller indices |
FWHM (2θ) |
d-spacing (Ao) |
| 23.719 | 790 | 0.085 | (111) | 1.5744 | 3.75123 |
| 35.1325 | 650 | 0.166 | (220) | 0.7872 | 2.55439 |
Two peaks were observed in the table at angle 2θ = 23.7194 and angle 2θ = 35.1325 whose intensity values were 183.42 and 206.34 respectively.
3.2. Morphological characterization
3.2.1. SEM (scanning electron microscopy)
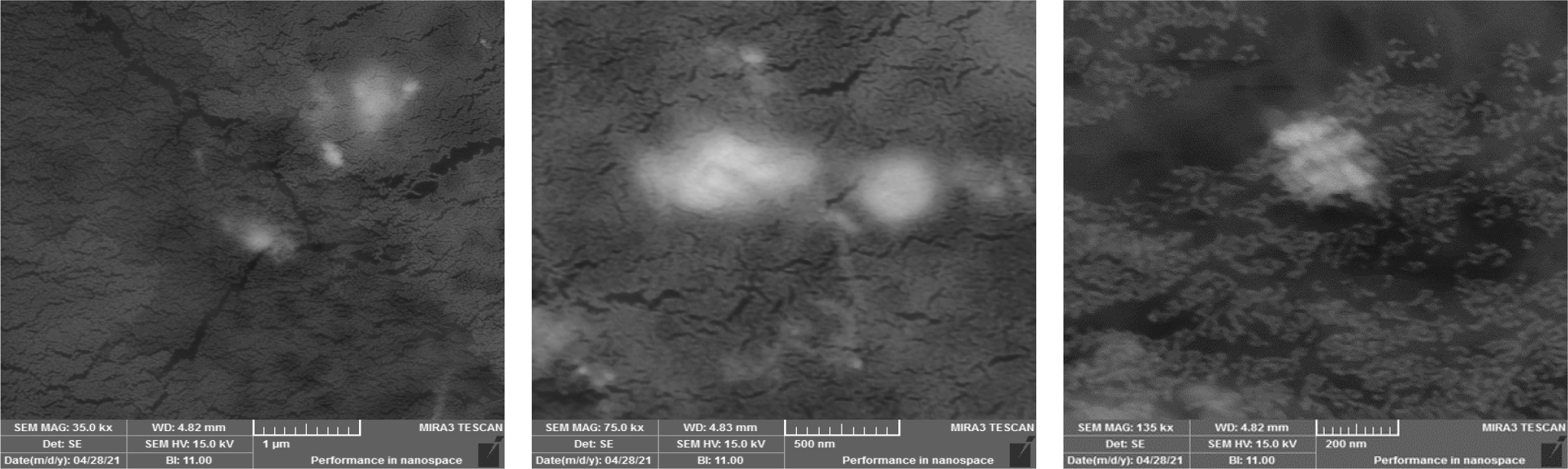
Nanoparticles iron oxide (Fe2O3) samples were examined using a scanning electron microscope (Tescan Mira 3 field emission scanning electron microscope) device made in the Czech as shown in Fig. 4. It is noted from this figure that IONPs, with a spherical shape and an average particle size of 200 nanometers, showed good stability due to their homogeneous dispersion and non-agglomeration, which is beneficial for their potential uses.
The results also showed through the images the presence of clear homogeneity of the model, as we notice the presence of dark areas (representing large areas of the surface of the models) which are heavy elements in (Fe2O3) such as (iron (while the bright areas represent light elements such as oxygen, which has a role in improving the properties of antibiotics, as the results showed a change in sizes and the formation of new phases, as the decrease in the grain size leads to an increase in the area of the grain boundaries, which helps in increasing the effectiveness of the antibiotic, and this is due to the nano effect of (Fe2O3).
3.2.2. AFM (atomic fluorescence microscopy)
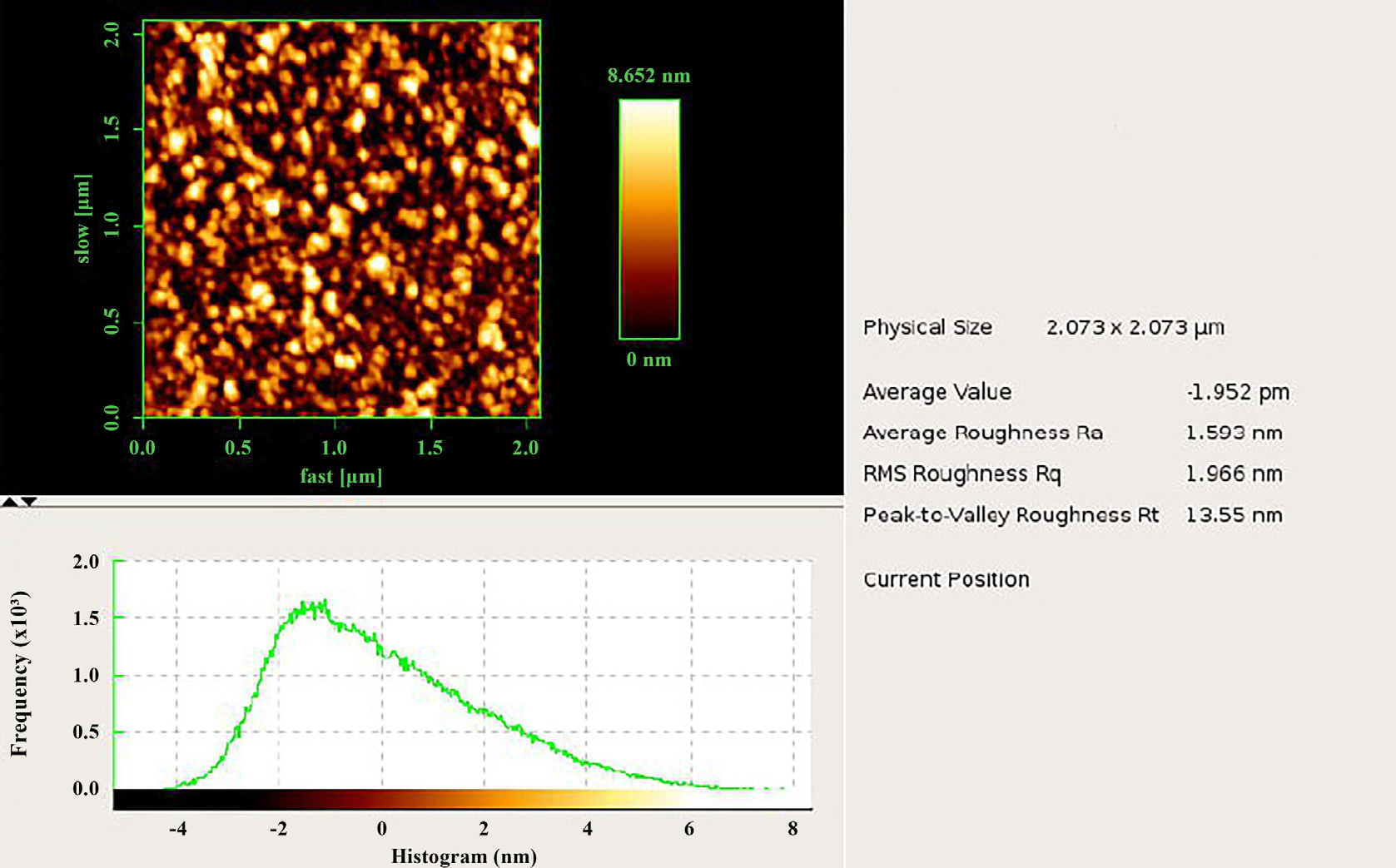
Iron oxide Fe2O3 nanoparticles, made up of iron and oxygen atoms in a crystal lattice structure, can vary in size and shape. AFM can provide valuable insights into their morphology and surface properties. AFM images show a two-dimensional representation of the nano-iron oxide material’s surface structure,33,34) combining topography, graphs, and numerical values to provide a comprehensive understanding of its topographic features, quality evaluation, and the impact of factors on its roughness properties.30,35) Nanoscale iron oxide (Fe2O3) samples were examined using AFM (NaioAFM 2022, Nanosurf, Switzerland) as shown in Fig. 5. It is noted from this figure that there are bright spots and dark spots with radii dimensions on the nano scale, which show the IONPs. These results are consistent with the results of XRD in terms of a decrease in crystal sizes, which leads to the aggregation of nanoparticles, making the sizes of the particles smaller, which plays a role in improving the properties of antibiotics.
3.3. EDX (energy dispersive X-ray spectroscopy) of Fe2O3 nanoparticles
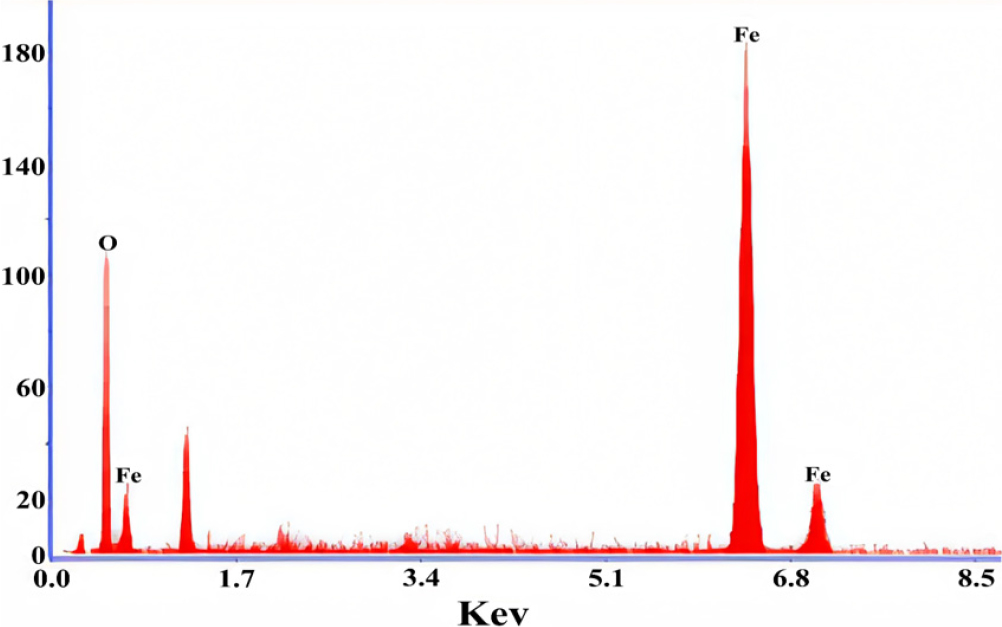
The EDX spectrometer is used to determine the quantity and quality of the elements involved in the formation of chemical compounds using the X-rays produced, which allows the desired element to be identified. A qualitative analysis approach, EDX analysis, helps determine the concentration of sample elements, allowing quantitative investigation of unknown substances. The element concentration increases with the height of the peak on the graph,36) as shown in Fig. 6, and information can be extracted from deeper samples with higher electron beam amplitude and lower atomic weight of the element. The presence of Fe, O and oxygen was verified, and the atomic weight percentage of the molecules was confirmed.
3.4. FTIR Analysis of Fe2O3 nanoparticles
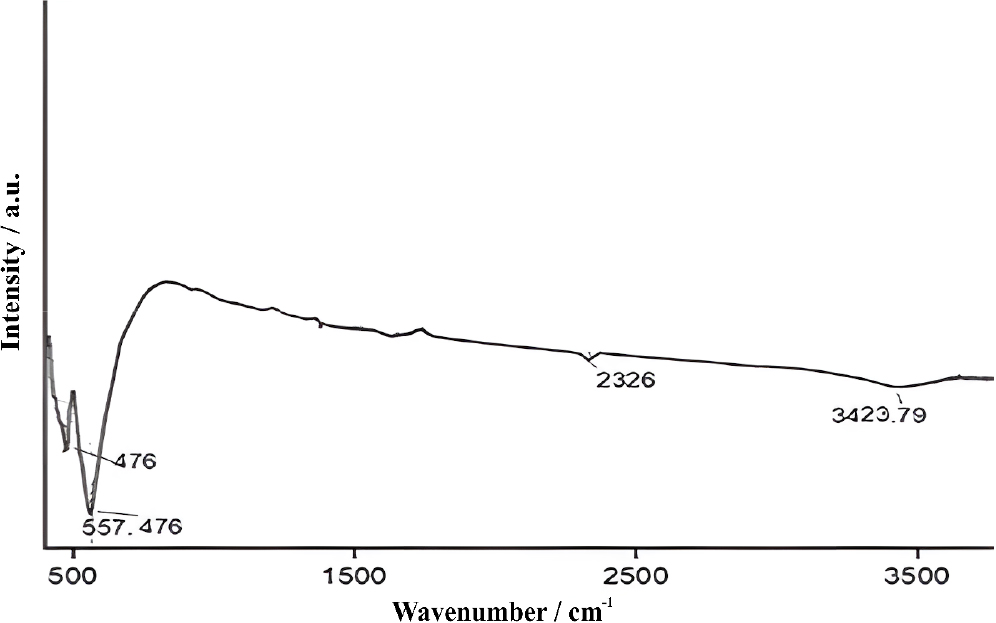
Fe2O3 nanoparticle samples were examined using FTIR (Shimadzu 1800) made in Japan. The spectra of Fe2O3 nanoparticles as in Fig. 7 that they had distinct absorption bands at 3,423, 2,326, 557, and 476 cm-1, indicating efficient synthesis. The absorption peak in the range of 3,423 cm-1 indicates the reduction of hydroxyl group and OH stretching vibrations between water molecules.37,38,39) The Fe-O bending vibrations correspond to the illumination values, confirming the effectiveness of the fabrication process.39,40)
3.5. Antibacterial activity
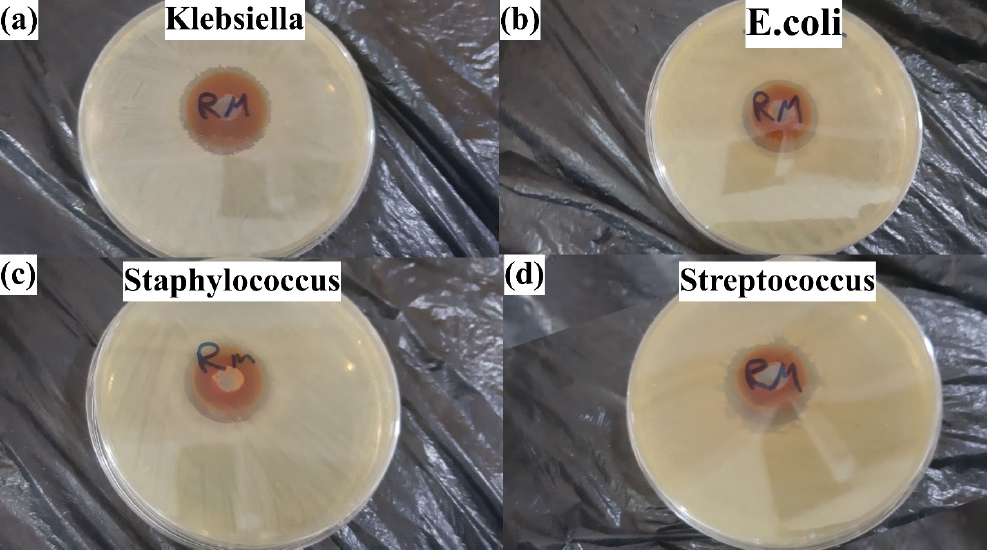
The antibacterial activity of Fe2O3 nanoparticles was evaluated against a range of pathogenic bacteria, including E. coli, Staphylococcus aureus, and Pseudomonas aeruginosa. The agar diffusion system showed significant zones of inhibition around the nanoparticle-loaded discs, indicating the antibacterial activity of Mothra nanoparticles against all bacteria tested. The antibacterial activity is attributed to the physical interaction between nanoparticles and bacterial cell walls, leading to cell membrane damage and bacterial death. These nanoparticles have been shown to have a powerful antibacterial effect against E. coli, causing cell leakage and bacterial death.40) They too inhibit the growth of Staphylococcus aureus by damaging the bacterial cell membrane and inducing oxidative stress. They have also shown antibacterial effect against Streptococcus species, such as S. pneumoniae and S. pyogenes, by causing oxidative stress, damaging bacterial cell membranes, and affecting cellular functions. They have likewise demonstrated antibacterial features against Klebsiella species, including Klebsiella pneumoniae, by making oxidative stress, interfering with vital cellular processes, damaging the integrity of the bacterial cell membrane, and preventing Klebsiella development. The antibacterial activity of Fe2O3 nanoparticles has been directed toward Pseudomonas aeruginosa, a multidrug-resistant bacteria associated with diverse diseases.41)Fig. 8 shows the antibacterial effect profile on Fe2O3 NPs using Borago. The disk diffusion method is used to find them. Tested against gram-positive (Staphylococcus, Streptococcus) and gram-negative (Klebsiella, E. coli) bacteria. The biological activity of each sample from the two eyes in Fig. 8 was tested and the results were obtained and are listed in Table 2.
Table 2.
Antifungal and antibacterial effect of Fe2O3 NPs.
|
Streptococcus (cm) |
Staphylococcus (cm) |
Klebsiella (cm) |
E. coli (cm) |
| 2.6 | 2.4 | 2.8 | 2.5 |
As for the antibiotics that are effective, the effectiveness of the treatment depends on the following: the degree of absorption of the drug into the bloodstream, the amount of the drug that reaches the sites of infection in the body, and the speed of the body’s elimination of the drug. These three factors are characterized by nano-iron oxide, which is very effective when forming small granule sizes. This is what was obtained from extracting it from the Maui rose. Therefore, it is expected that using Maui rose seeds is effective as an antibiotic against bacteria.
4. Conclusion
A group of bacterial strains was used to evaluate the antibacterial effectiveness of nanoparticles extracted from flower plants (Borago). Study the structural and chemical properties of the produced ions using FTIR, TEM, and XRD. The antibacterial efficacy of IONPs against E. coli and Staphylococcus aureus will also be evaluated. The results showed the: XRD showed distinct diffraction peaks consistent with the hematite phase. This confirms the crystalline nature of the synthesized nanoparticles, which is critical for applications requiring stable structural integrity, such as antibacterial and catalytic uses. It is noted from SEM technique that IONPs, with a spherical shape and an average particle size of 200 nanometers, showed good stability due to their homogeneous dispersion and non-agglomeration, which is beneficial for their potential uses. It is noted from AFM technique that there are bright spots and dark spots with radii dimensions on the nano scale, which show the IONPs. The Fe2O3 nanoparticles were effectively synthesized, as evidenced by the FTIR spectroscopy results. The distinct absorption bands at 3,423, 2,326, 557, and 476 cm-1 indicate successful fabrication with characteristic functional groups and molecular interactions. The effect of Fe2O3 nanoparticles was tested and it was observed that they effectively affect all types of bacteria and germs under experiment.