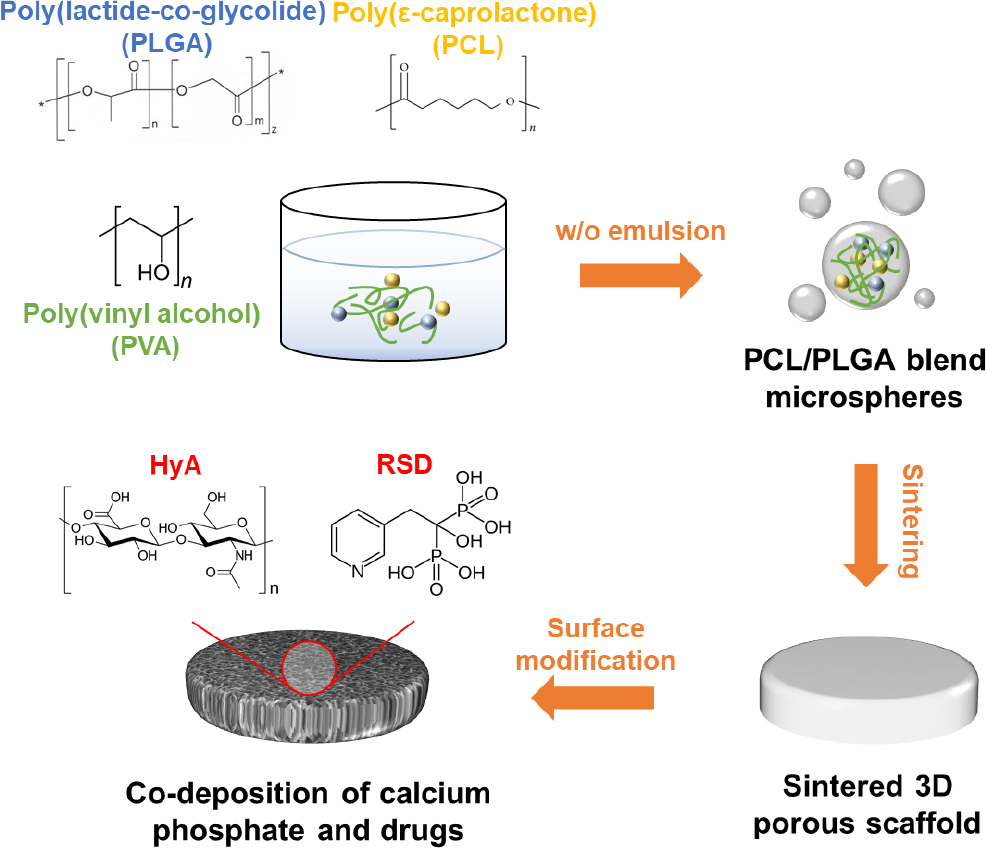
1. Introduction
2. Experimental Procedure
2.1. Materials
2.2. Preparation of PCL/PLGA scaffolds
2.3. Surface modification
2.4. Morphological analysis
2.5. Porosity measurement
2.6. Degradation test
2.7. Cell responses
3. Results and Discussion
3.1. Fabrication of microspheres
3.2. Fabrication of scaffold and characterization
3.3. Surface modification by CAP coating
3.4. Evaluation of cellular responses
3.5. FE-SEM observation of biomineralization
4. Conclusion
1. Introduction
Among synthetic materials, biodegradable polymers are widely used for creating bone substitute scaffolds because they provide structural support while gradually degrading, allowing natural tissue to grow in their place.1,2) Therefore, it is crucial for biodegradable scaffolds to have a degradation rate that matches the rate of new bone formation.3,4)
Poly(lactic-co-glycolic acid) (PLGA) and poly(ε-caprolactone) (PCL) are two polyesters known for their excellent biocompatibility, making them popular choices for fabricating biodegradable scaffolds.5,6) PLGA has been approved by the U.S. Food and Drug Administration (FDA) for use in medical devices, and its degradation rate can be tailored by adjusting the molecular weight or the ratio of lactic acid to glycolic acid.7,8) Several studies have aimed to optimize PLGA’s degradation rate to align with the bone regeneration process by increasing the lactic acid content or the molecular weight.9) PCL is also widely used for scaffold fabrication due to its low cost and ease of processing, as it has a low melting temperature (59~64 °C).10,11) Because PCL degrades more slowly than other biodegradable polymers, it has been extensively employed in long-term drug delivery systems, medical devices, and other tissue engineering applications.12)
Calcium phosphate (CAP), the inorganic phase of natural bone, is widely used in hard tissue engineering because of its excellent biocompatibility and osteoconductivity.13) However, due to its brittleness and poor mechanical properties, CAP is often applied as a coating material on metallic substrates.14) In addition to its use as a coating, CAP is frequently combined with polymers to form composite scaffolds, where the biological functionality of the polymer is enhanced by CAP.15) Recently, CAP has also been explored as a carrier for biomolecules, such as proteins, growth factors, and DNA.16)
Polymers are highly favored in biomaterial fabrication due to their versatility in forming desired shapes and sizes, as well as their adaptability for controlled drug delivery systems. PLGA, in particular, is known for its low cytotoxicity, as its degradation byproducts are naturally metabolized through the Krebs cycle.17,18) However, the degradation process can lead to an acidic environment that may harm surrounding tissues, reduce drug efficacy, or cause tissue necrosis.19) Polymers have also been extensively used in the production of 3D scaffolds for tissue engineering. The scaffold design varies depending on the application, including porous, fibrous, microsphere-based, polymer-ceramic composites, and acellular or cellular scaffolds.20) For effective bone regeneration, scaffolds with a porous 3D structure should have a pore size greater than 200 µm to allow the infiltration and proliferation of osteoblasts and facilitate angiogenesis.21) Several fabrication methods have been developed for creating 3D scaffolds, including salt leaching,22) electrospinning,23) and microsphere sintering.24)
Herein, we investigated the characteristics of a surface-modified scaffold enhanced by a CAP coating that incorporates binary drugs, hyaluronic acid (HyA) and risedronate (RSD), with the goal of improving bone repair in a 3D environment. Our findings further demonstrate that these binary drugs influence proliferation and differentiation through distinct mechanisms and in a concentration-dependent manner, highlighting the need for a careful balance between these biological activities.
2. Experimental Procedure
2.1. Materials
PCL (704605, Mn 45,000), PLGA (RG 504 H, Mw 38,000~54,000), and polyvinyl alcohol (PVA, 363170) were purchased from Sigma-Aldrich and EVONIK, respectively. Rat mesenchymal stem cells (rMSCs, ATCC) were used to analyze cellular responses.
2.2. Preparation of PCL/PLGA scaffolds
The PCL/PLGA microspheres were used as building blocks to fabricate the scaffold. Microspheres were prepared using a simple water-in-oil (W/O) emulsion method. In brief, a PCL/PLGA (30/70, w/w) solution dissolved in chloroform was added dropwise into a 2 % (w/v) PVA aqueous solution and stirred at 1,000 rpm for 2 h. The resulting microspheres were washed four times with deionized water and then dried in a vacuum desiccator. Microspheres with diameters ranging from 100 to 300 µm were separated from other sizes and placed into a polyethylene mold along with NaCl salt particles (100~200 µm in diameter). The mixture was sintered at 60 °C for 10 min. To create pores in the sintered scaffold, the structure was immersed in water for salt-leaching, with the water being changed every 24 h over a 48-h period to ensure complete removal of the salt. All scaffolds were sterilized by immersion in 70 % ethanol for 30 min, followed by washing with Dulbecco’s phosphate buffered saline (DPBS) for 30 min and with Dulbecco’s Modified Eagles Medium (DMEM) for 30 min before use.
2.3. Surface modification
To create CAP coatings with incorporated drugs on both the inner and outer surfaces of the scaffold, drug-containing ionic solutions were formulated. The prepared scaffolds were etched in 99.9 % ethanol for 10 min and then immersed in CAP solutions saturated with 4.52 mM calcium ions and 3.73 mM phosphate ions, containing either HyA at 50 µg/mL (h) or 100 µg/mL (H), and RSD at 10-7 M (r) or 10-5 M (R), to produce CAP, CAP-Rh, CAP-rH, and CAP-RH scaffolds, respectively. The scaffolds were incubated in their respective solutions at 4 °C for 1 h, followed by 5 h at 37 °C. After coating, the scaffolds were thoroughly washed with deionized water and stored in a vacuum desiccator. All prepared solutions were filtered through a 0.2 µm pore size filter before use.
2.4. Morphological analysis
The surface structures of the microspheres and scaffolds were analyzed using field emission scanning electron microscopy (FE-SEM) (S-4300, Hitachi, Japan). Before imaging, the samples were coated with platinum using an ion sputter coater (E-1045, Hitachi, Japan). The FE-SEM was operated at an acceleration voltage ranging from 5 to 15 kV.
2.5. Porosity measurement
The porosity of the fabricated scaffold was measured using the liquid displacement method. The dry weight (Ws) and volume (Vs) of the scaffold were recorded. The dried scaffold was then slowly immersed in deionized water and left submerged for a specified period. Afterward, the scaffold was carefully removed, and the excess surface water was gently blotted away. Then, the wet weight (Ww) of the scaffold was measured. The volume of water (Vw) was determined by calculating the weight difference between the wet and dry scaffold samples, utilizing the density of water (ρ) at room temperature, as described in Eq. (1). The scaffold porosity (ε) was then calculated using Eq. (2).
2.6. Degradation test
The amount of calcium ions released during the degradation of CAP-coated composite scaffolds over 14 days in DPBS was measured using an inductively coupled plasma mass spectrometer (ICP-MS, ELAN DRC-e, PerkinElmer, USA). The scaffolds were placed in glass vials at room temperature and gently rotated. Samples were taken at predetermined time intervals, and an equal volume of DPBS was replenished. To analyze the degradation pattern based on surface modification, non-modified and CAP scaffolds were incubated in DPBS at 37 °C for 10 weeks. At designated time points, the scaffolds were removed, fully dried in a vacuum desiccator, and weighed.
2.7. Cell responses
For the proliferation, rMSCs were seeded at a density of 2.5 × 104 cells per scaffold. To prevent cell loss, 150 µL of media was added after 1 h, followed by 850 µL of media after 12 h. On days 3, 5, and 7, 10 % Cell Counting Kit-8 solution (CCK-8, CK04, Dojindo, Japan) was added, and the scaffolds were incubated at 37 °C with 5 % CO2 for 2 h. After incubation, the solution was transferred to a 96-well plate, and absorbance was measured at 450 nm using a microplate reader. For the ALP activity, cells were seeded onto the prepared specimens at a density of 1 × 105 cells/scaffold, and cultured for one week. The media was supplemented with 50 µg/mL ascorbic acid, 10 mM β-glycerophosphate, and 10-7 M dexamethasone, and the cells were grown for 5, 10, and 15 days. For measurements, the media was removed, and the cells were washed with 0.9 % NaCl. Then, lysis buffer was added, and the cells were incubated for 10 min to induce lysis. The cell lysate was centrifuged at 14,000 rpm for 15 min, and the supernatant was mixed with 100 µL of pNPP. The mixture was incubated at 37 °C for 1 h, and absorbance was measured at 405 nm.
For the biomineralization, cells were seeded onto the scaffolds at a density of 5 × 105 cells/scaffold and cultured in media for 2 h, 5 h, 5 days, 10 days, and 15 days. During the experiments, the scaffolds were washed twice with DPBS and fixed with 10 % neutral formalin for 15 min. The fixed samples were washed twice with deionized water and thoroughly dried before analyzing their morphology using FE-SEM.
3. Results and Discussion
Fig. 1 illustrates the overall process for surface modification of a 3D scaffold incorporating drugs, achieved through heat sintering of microspheres. The PCL/PLGA blend scaffold is fabricated using a bottom-up approach, where microspheres act as the building blocks. These microspheres are formed via a W/O emulsion process using PVA as an emulsifier and the previously mentioned polyesters. The blended microspheres are then heat-sintered to create a disc-shaped, porous 3D scaffold. This porous structure is crucial for promoting effective cell proliferation and differentiation, essential for bone regeneration. Additionally, surface modification through CAP deposition, along with the incorporation of HyA and RSD, further enhances the scaffold’s bone regeneration capabilities.
3.1. Fabrication of microspheres
Fig. 2 displays FE-SEM images of microspheres that were fabricated using a 30/70 PCL/PLGA blend to create scaffolds. The microspheres were prepared under different conditions. The left panels of Fig. 2 represent microspheres produced in 1 % PVA, while right panels show those produced in 2 % PVA. All images were observed at 300× magnification. Microspheres fabricated under the 1 % PVA condition largely failed to maintain a stable shape, resembling acorns in appearance. On the other hand, the microspheres prepared using 2 % PVA with stirring at 1,000 rpm for 2 h, maintained a spherical shape and had a size range of 100~300 µm. Additionally, although the PVA concentration remained the same, when stirring time was extended or stirring speed was increased, the microspheres were not properly formed. It means that the appropriate emulsifying conditions and shear stress are essential factors in achieving a homogeneous composite.
3.2. Fabrication of scaffold and characterization
Fig. 3 presents the properties of the fabricated 3D scaffold. Fig. 3(a) shows an image of the 3D scaffold formed through heat sintering of microspheres. The sintering process was carried out at 60 °C, near the glass transition temperature of PCL, resulting in a self-standing structure. Fig. 3(b) is an FE-SEM image of microspheres with diameters ranging from 100 to 300 µm, sintered together while retaining their spherical shape. Fig. 3(c) illustrates the porosity of the fabricated porous scaffold, measured based on the weight ratio of NaCl porogen. Using the water displacement method, the porosity was found to be 50.7 % at a 2 : 1 ratio and 46.3 % at a 3 : 1 ratio. Higher porosity creates a more favorable environment for cell ingrowth and differentiation, as it forms an interconnected network, which is critical for 3D cell proliferation. Also, Borden et al.4) prepared a microsphere-based scaffold using thermal sintering of PLGA microspheres that contain hydroxyapatite. Their scaffold displayed porosity about ~40 %, and the mechanical properties were similar to human cancellous bone. Hence, the scaffold was fabricated at a 2 : 1 ratio to achieve optimal porosity. The formed microstructure plays a crucial role in mechanical properties as well as in cell proliferation and differentiation. A microstructure with appropriate pore size and porosity is essential for achieving mechanical strength comparable to that of sponge bone. Additionally, since human cells function in a three-dimensional manner, a proper three-dimensional microstructure is vital for effective bone repair. Furthermore, the microstructure is essential for manufacturing the 3D scaffold, as they create pathways that allow waste products and necessary nutrients to move as cells grow into the voids of the microstructure. Fig. 3(d) shows the degradation profile of the scaffold over a 10-week period, comparing pre- and post-surface modification with CAP. In the degradation study, the unmodified scaffold lost about 23 % of its weight within the first 2 weeks, indicating rapid degradation. In contrast, the CAP-modified scaffold exhibited much slower degradation, with less than 10 % weight loss over the same period. This slower degradation rate suggests successful formation of the CAP thin film. The similar weight loss observed around the 4-week mark is attributed to the accumulation of hydrolysis products within the scaffold, preventing their diffusion into the aqueous solution.25) The degradation rate of the PCL/PLGA system can vary from several months to years, depending on the blend ratio, allowing for tunable degradation speeds based on the intended application. To further slow degradation, the scaffold can be combined with HA nanoparticles.26) The release of calcium ions from the scaffolds with CAP surface modification was measured over time during incubation in DPBS solution. As shown in Fig. 3(e), the cumulative amount of calcium ions released was 937 ppb on day 3, 1,575 ppb on day 5, 2,302 ppb on day 7, and 2,809 ppb on day 14. The release of calcium ions was most active up to day 7, after which the amount released gradually decreased. These results suggest that the scaffold can rapidly increase the local concentration of calcium ions in the implantation site during the initial phase, which can positively influence bone regeneration by promoting osteogenesis through calcium ion regulation.27)
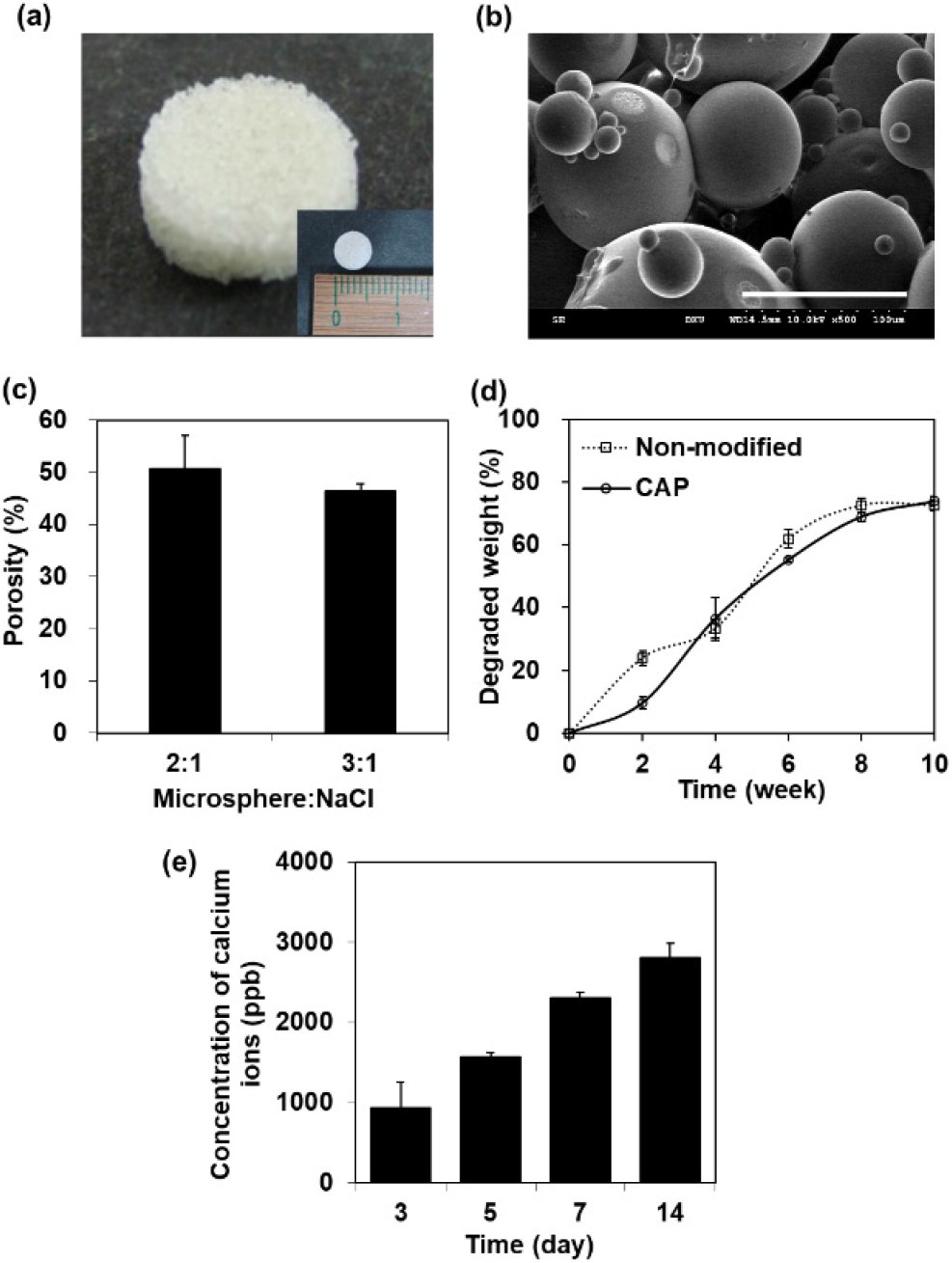
Fig. 3.
(a) Photographic and (b) FE-SEM images of sintered PCL/PLGA 3D scaffold (scale bars: 100 µm). (c) Porosity of fabricated scaffold depending on the amount of NaCl and its (d) degradation pattern for 10 weeks before and after surface modification. (e) Released calcium ions from coating layer of CAP for 14 days.
3.3. Surface modification by CAP coating
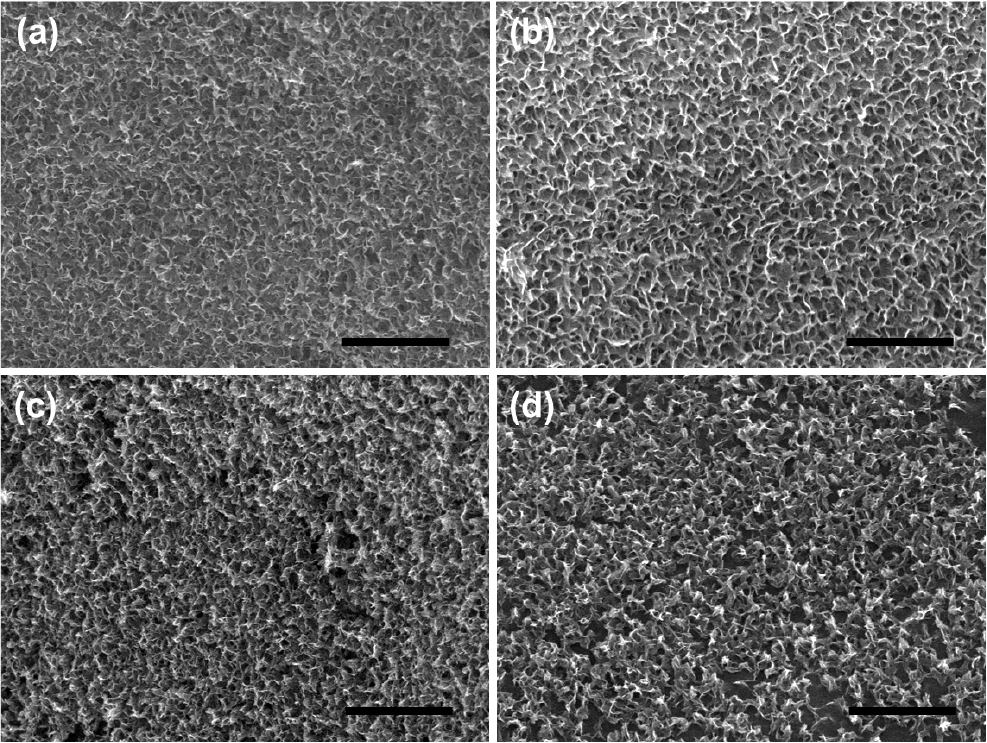
Fig. 4 shows FE-SEM images of scaffolds that were surface-modified into four types: CAP, CAP-Rh, CAP-rH, and CAP-RH. Fig. 4(a) depicts the surface of the CAP scaffold, which was modified solely with a CAP without any drug incorporation. In contrast, Fig. 4(b) shows the surface of the CAP-Rh scaffold, modified with 10-5 M RSD and 50 µg/mL HyA. Fig. 4(c) displays the surface of a scaffold modified with 10-7 M RSD and 100 µg/mL HyA, while Fig. 4(d) illustrates the surface of the CAP-RH scaffold, which contains 10-5 M RSD and 100 µg/mL HyA. Although the surface morphology varies slightly depending on the concentration of the incorporated drugs, all images confirm that the scaffolds were successfully modified with a CAP thin film across the three-dimensional surface. This indicates that the surface modification was effective in each case, demonstrating successful incorporation of the drugs into the CAP layer.
3.4. Evaluation of cellular responses
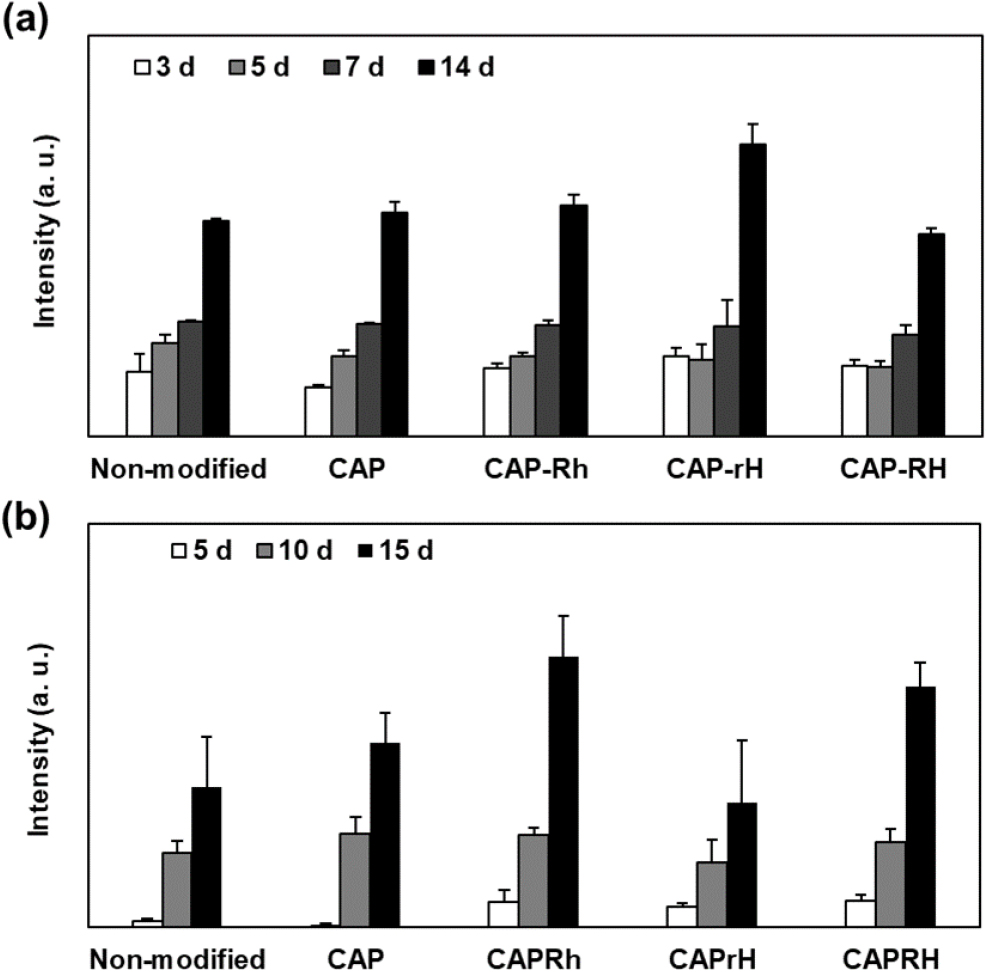
Fig. 5(a) illustrates the proliferation of rMSCs cultured on a non-modified scaffold and CAP, CAP-Rh, CAP-rH, and CAP-RH-coated PCL/PLGA scaffolds. The CAP-rH coated scaffold produced the highest proliferation of all the scaffolds. The drug-incorporated CAP-rH enhanced osteoblastic proliferation compared with the CAP scaffold indicating that both RSD and HyA affect osteoblastic proliferation. We also observed that the concentrations of RSD and HyA induced considerable changes in proliferative behavior. HyA produced a stimulatory effect on osteoblast proliferation. For example, proliferation on the CAP-rH scaffold showed the highest osteoblastic proliferation among all CAP-related samples. After 14 days in culture, the proliferation in CAP-rH increased by 1.5-fold compared with CAP. On the other hand, proliferation on the CAP-RH scaffold notably decreased, indicating that the RSD concentration can induce inhibitory effects on osteoblastic proliferation. Additionally, we observed that the proliferation ability showed no significant differences depending on the CAP coating or the concentration of the incorporated drugs. This may be attributed to the excellent cellular activity of the PCL/PLGA scaffold itself. In particular, PLGA is known for its remarkable cell proliferation and differentiation capabilities, which may limit the ability to demonstrate effects that surpass the intrinsic cellular activity of the polymer. Nevertheless, it is interesting to note that at specific concentrations, we can observe an effect on cell proliferation. To evaluate the differentiation of the rMSCs, alkaline phosphatase (ALP) activity which is a critical factor for the early differentiation, was investigated for 15 days after 7 days proliferation. Fig. 5(b) shows ALP activity depending on the modified surface. ALP activity was the highest in CAP-Rh and the lowest in CAP-rH. This result suggests that RSD incorporation served to promote ALP activity of osteoblast and that HyA appeared not to be directly related to the ALP activity. High RSD-incorporated scaffold, i.e., CAP-Rh and CAP-RH showed relatively higher ALP activity. Interestingly, CAP-rH showed lowest differentiation capability cost of highest osteoblastic proliferation. It indicates that proper proliferation and sequential differentiation must be achieved for effective bone regeneration. Furthermore, despite the high cellular activity of the scaffold itself, the CAP scaffold demonstrated higher ALP activity compared to the non-modified scaffold. This suggests that the calcium and phosphate ions released from the CAP effectively induce the differentiation of rMSCs. At the same time, it can be argued that a purely polymeric bone substitute may not sufficiently support effective bone regeneration. The optimal drug incorporation should aim to maximize both proliferation and differentiation, but these two processes often conflict with each other. Therefore, ensuring an appropriate period for proliferation and the timely entry of proliferated cells into the proper differentiation stages is crucial. Consequently, appropriately regulated drug incorporation within CAP multilayers could be employed as a strategy in future studies. Bisphosphonates can be classified into nitrogen-containing and non-nitrogen-containing types. Although their binding affinity to bone minerals varies, both types inhibit bone resorption by inducing apoptosis in osteoclasts.28) Additionally, bisphosphonates have been reported to directly promote the proliferation and differentiation of osteoblasts.29) In studies where rat and human bone marrow cells were cultured in media containing dissolved bisphosphonates, etidronate showed effectiveness in early bone regeneration at concentrations between 10-5 M and 10-9 M, while alendronate was effective at concentrations below 10-7 M, but exhibited inhibitory effects at higher concentrations.30) Similarly, when pamidronate was applied to human osteoblasts, significant enhancement in proliferation and differentiation was observed at low concentrations (1 to 3 µg/mL), whereas higher concentrations resulted in diminished effects.31) Murayama et al.32) also reported that at high concentrations of RSD (10 µM or more), apoptosis and growth inhibition were induced in various osteogenic sarcoma cell lines. These findings highlight that bisphosphonates affect osteoblasts in a concentration-dependent manner, with differing effects depending on the specific type used.
HyA is a major component of the extracellular matrix in animal cells and plays a role in various cellular responses, such as proliferation, differentiation, and adhesion.33) HyA also regulates the activity of bone morphogenetic protein-2 (BMP-2), a key factor in bone regeneration, by promoting osteoblast differentiation. It has been reported that 100 µg/mL of HyA in culture media increased BMP-2-induced differentiation by suppressing the expression of BMP-2 receptor antagonists.34) Recently, CAP has been widely used as a drug delivery carrier. For example, a BMP-2 release system was developed by adsorbing a nanocomposite of chondroitin sulfate and BMP-2 onto a hydroxyapatite-coated titanium disc.35) Additionally, using a co-precipitation method with calcium and phosphate ions, an amorphous CAP thin film containing collagen was successfully fabricated, which effectively delivered collagen and regulated the cellular response of chondrocytes.36) Thus, incorporating both RSD and HyA into CAP thin films provides a strategy to simultaneously enhance osteoblast proliferation and improve differentiation capacity within the scaffold.
3.5. FE-SEM observation of biomineralization
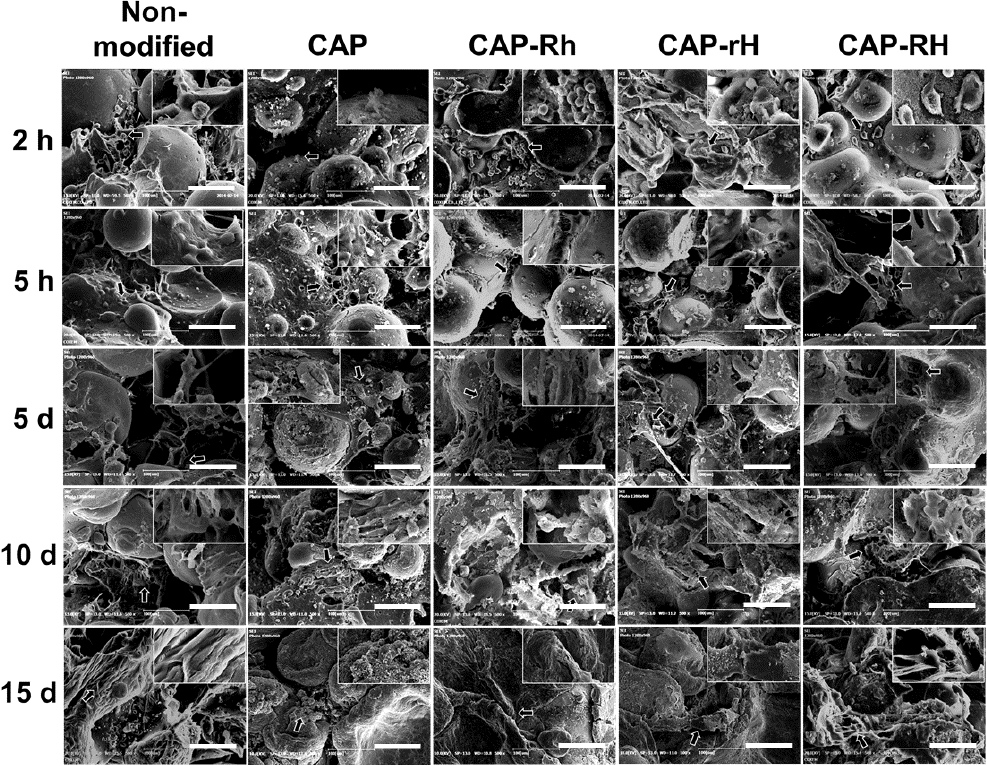
To observe biomineralization, FE-SEM imaging was conducted from 2 h after cell seeding, when initial cell attachment occurs, up to 15 days, when the mineralized matrix fully covered and interconnected the scaffold as shown in Fig. 6. At 2 h post-seeding, cells were seen individually attached to the scaffold with minimal intercellular connections. By 5 h, the cells began to spread and form connections with one another. At 5 days, matrix formation was evident, and individual cells were no longer distinguishable. By 10 days, a substantial portion of the scaffold was covered with matrix, indicating significant mineralization. Finally, by 15 days, the microsphere structure was no longer visible as the scaffold was completely enveloped by the mineralized matrix. Especially, CAP-Rh showed effective bone nodule and more biomineralized morphology with cell-to cell and cell-to-scaffold interactions. It supports that RSD served to promote osteoblastic differentiation of rMSCs.
4. Conclusion
This study successfully developed a novel PCL/PLGA-based scaffold using a microsphere approach, enhanced with CAP coatings containing drugs to improve bone regeneration. The scaffold’s porous structure, created through heat sintering and salt-leaching, demonstrated favorable characteristics for supporting cell proliferation and differentiation. The CAP coatings reduced the scaffold’s early degradation rate, providing a stable environment for tissue regeneration. Additionally, the slow release of calcium ions from the modified scaffold promoted osteogenic activity, essential for bone healing. Among the modifications, the CAP-Rh scaffold, containing high concentrations of RSD (10-5 M) and low concentration of HyA (50 µg/mL), showed the highest osteoblast proliferation and increased ALP activity, highlighting its potential for cell growth and early differentiation. It indicates that those drugs affect osteoblast behavior in a concentration-dependent manner, requiring a balance between proliferation and differentiation for optimal bone regeneration. Therefore, PCL/PLGA scaffolds with CAP coatings and surface modifications hold great promise for bone tissue engineering. Future studies should focus on optimizing drug concentrations and conducting long-term in vivo evaluations to further validate the scaffold’s potential in promoting bone regeneration. Moreover, CAP shows great potential as a carrier for natural product drug mixtures in traditional oriental medicine, particularly for the elderly.