1. Introduction
The soaring oil price hikes and increased emissions of greenhouse gas forced us to look upon suitable alternatives for fossil fuels. In this view, biomass-based biofuel production through biological conversion of CO2 has received wide attention. Among the various feedstocks, microalgae are looked upon as one of the most promising sources for biodiesel production.1,2) They gain several advantages over the conventional oil crops such as nonedible biomass, high productivity per area and high lipid content. Furthermore, microalgae are good feedstock for the value-added pigments. In spite of such remarkable advantages, the production of microalgae-based biodiesel and pigments still remains expensive due to factors associated with cultivation and harvesting of microalgae.3)
Harvesting microalgae is an energy demanding process which covers 20 ~ 30 % of total cost of microalgal biodiesel.2) Microalgae are uniformly dispersed in the culture medium often under 2 g/L concentration. The dilute dispersion along with their small size of a few micrometers and electronegative surface imposes difficulties during harvesting.4) The widely used techniques to harvest microalgae include centrifugation, filtration, ultrasound electrolysis-based technologies, which demand high energy.5-8) Usage of chemical flocculants such as polyvalent cations, cationic polymers and inorganics was thought to be an efficient harvesting method.9,10) These flocculants break the stable dispersion of negatively charged microalgae by introducing positively charged components without expense of energy. However, using metal salts such as alum and ferric chloride for flocculation of microalgae results in contaminations both in the harvested biomass and in the culture which could not be reused for further cultivation. To overcome such contamination issue, recently, it has been suggested functionalizing the positively charged flocculants on separable particles such as magnetic particles, which still remains a challenge of up-scale.11,12)
In addition to biological conversion of CO2, mineral carbonation has thought to be an efficient conversion process of anthropogenic CO2.13,14) It has been reported that microalgae can induce mineralization of CaCO3 under certain environments such as calcium saturated freshwater. 15-17) It is not biologically controlled mineralization such as CaCO3 formation in mollusk shell and Fe3O4 formation in magetotactic bacteria but rather extracellular precipitation of CaCO3 induced by the properties of cell surface.18-20) Negatively charged surface of microalgae can provide binding sites of Ca2+ which is precipitated into CaCO3 in the presence of CO32−.
In this study, we investigate the formation of CaCO3 in the presence of microalgae, which leads to flocculation of microalgae by breaking the stable dispersion, as an alternative method for microalgal harvesting. For this, various concentration of calcium and carbonate source was introduced into the culture after 7 days’ cultivation of Chlorella sp. KR-1. The harvesting efficiency and physicochemical properties of formed CaCO3 according to the concentration of CaCO3 precursor have been investigated. Chlorella sp. KR-1 was chosen as a model species with its high lipid content [approximately 37 ~ 41 % (w/w) of triacylglycerol accumulation per cell mass] and difficulties in harvesting due to small size of around 3 μm in diameter and high stability of dispersion in wide range of pH.21)
2. Methods
2.1 Strain and cell culture
Chlorella sp. KR-1, an indigenous freshwater microalga, was isolated from a stream in Yeongwol-gun, Korea. The microalgae were cultivated in a 1 L Pyrex bubble-column photobioreactor containing 500 mL medium supplied with 10 % (v/v) CO2 in air at a rate of 0.75 L/min under illumination of 12 fluorescent lamps (light intensity: 80 mmol photons/ m2·s) in a temperature-controlled room at 30 °C. A modified N8 medium was used in this study, which contains 3 mM KNO3, 5.44 mM KH2PO4, 1.83 mM Na2HPO4, 0.20 mM MgSO4·7H2O, 0.12 mM CaCl2, 0.03 mM FeNaEDTA, 0.01 mM ZnSO4·7H2O, 0.07 mM MnCl2·4H2O, 0.07 mM CuSO4 and 0.01 mM Al2(SO4)3· 18H2O. The pH of culture was kept around 6.5 during 7 days’ cultivation. All the conditions of cultivation were previously optimized for the high lipid containing microalgae.11,21)
2.2. Mineralization of CaCO3 and microalgal harvesting
After 7 days’ cultivation, the culture was flushed by air bubbling at a rate of 0.75 L/min for 1 h to exclude the effect of CaCO3 mineralization by dissolved CO2. Then the calculated amount of 10, 30, 50 and 100 mM CaCl2· 2H2O (≥ 99.0 %, Sigma-Aldrich, USA) powder was added into the bubbling culture followed by pumping 100 mL of equivalent mole of Na2CO3 (≥ 99.5 %, Sigma- Aldrich, USA) using peristaltic pump for 6 h. The samples of the flocs. and the resulted harvesting efficiencies were noted by the concentration of the mineral precursor to be ‘0 mM’, ’10 mM’, ‘30 mM’, ’50 mM’ and ‘100 mM’. After the air bubbling was ceased, the flocs. started to sediment and the supernatant was taken from 1/4 spot of the culture from the top at 0, 10, 30 and 60 mins of time points. The harvesting efficiency was calculated by the following equation based on the optical density (OD) of the supernatant.11,21)
where ODi and ODt were the initial OD and the OD of the supernatant taken at t = 0, 10, 30 and 60 mins, respectively.
2.3. Analytical Methods
OD was measured by UV-Vis spectrophotometry (Optizen 2120UV, Mecasys Co., Korea) at 660 nm. The optical microscopic images were obtained with an oil immersion objective lens (Microscope Axio Imager.A2, Carl Zeiss Microscopy GmbH, Germany). For field emission scanning electron microscope (FE-SEM) and energy-dispersive spectroscopy (EDS) analysis with mapping (S-4800, HITACHI, Japan), the mineralized microalgae were freeze-dried at - 55 °C after harvesting. The cross-sectional view of the mineralized microalgae was imaged after focused ion beam (FIB) cutting by using Dual Beam FIB System (Nova 200, FEI, Netherlands). The crystal structure of the mineralized microalgae was analyzed by using a high power powder X-ray diffractometer (XRD, Rigaku, D/MAX-2500(18 kW)) with a Cu Ka radiation source (k = 1.5406 Å) at 40 kV and 300 mA.
3. Results and Discussion
The addition of Na2CO3 solution into the cultures in the presence of calcium generated CaCO3 precipitates forming flocs. of CaCO3 and microalgae. The stability of microalgal suspension was broken by the formation of flocs., which leads to the fast sedimentation of flocs. after the bubbling was stopped. Fig. 1(a) shows the sunk flocs. at the bottom of bubble-column photobioreactors after 60 mins of gravitational sedimentations for 10 ~ 100 mM, whereas no significant change was observed for the 0 mM. Over 95 % of harvesting efficiency was obtained within 10 minutes in case of 50 mM and 100 mM, which reached to 98 ~ 99 % in 60 mins [Fig. 1(b)]. The harvesting efficiency showed the increasing trend according to the increased concentration of mineral precursors, Ca2+ and CO32−. For 10 mM, the harvesting efficiency reached to 90 % after 60 min. The pH of cultivation media was increased as Na2CO3 solution was added and reached up to pH = 10 where the concentration of Na2CO3 did not significantly affected on the pH after flocs. formation [Fig. 1(c)].
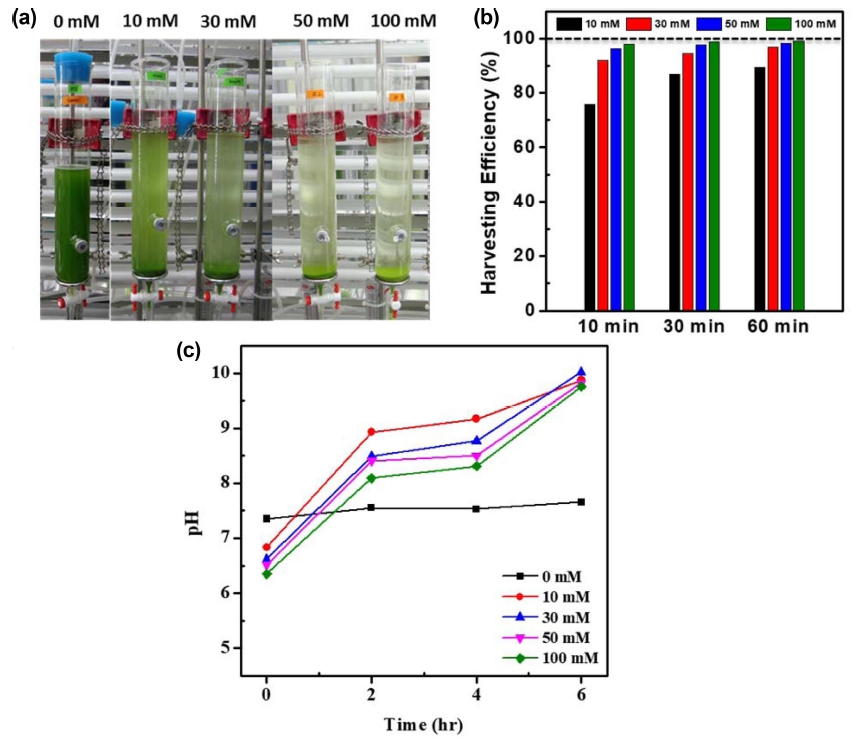
Fig. 1
(a) Photos of microalgal cultures in bubble-column photobioreactors with gravitational sedimentations of flocs. for 60 min, (b) harvesting efficiency, and (c) pH changes of cultivation media.
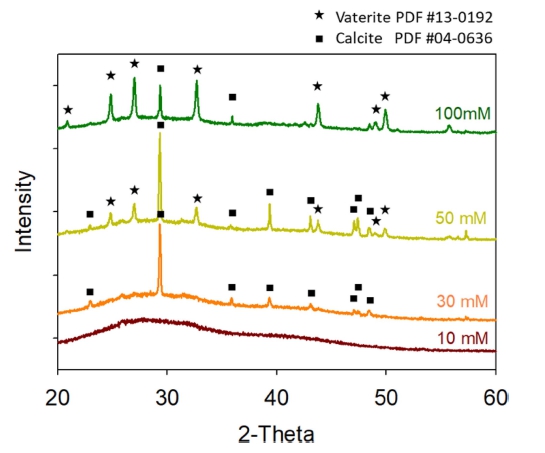
XRD patterns in Fig. 2 reveals that the polymorphs of precipitates varied depending on the concentration of mineral precursors (Fig. 2). XRD patterns confirm the formation of amorphous and calcite (PDF #04-0636) phases in case of 10 mM and 30 mM, respectively. When the concentration of mineral precursors was increased up to 50 and 100 mM, vaterite (PDF #13-0192) phase was newly formed to have mixed phases of vaterite and calcite. The vaterite phase was increased as the concentration of mineral precursor was increased.
In 10 mM sample, the presence of precipitates is not clear by microscope observation in bright field mode [Fig. 3(a)]. A microscopic observation in differential interference contrast (DIC) mode confirms the presence of precipitates in the flocs. between microalgae [inset of Fig. 3(a)]. As revealed by XRD analysis, these precipitates are amorphous [Fig. 2 and 3(a)]. Precipitates with rhombohedral shape, the typical morphology of calcite, were formed in 30 mM [Fig. 3(b)]. The crystalline phase was confirmed to be calcite by the XRD result (Fig. 2). The CaCO3 precipitates with spherical shape were shown in 50 mM and 100 mM together with rhombohedral-shaped calcite crystals with well-developed facets [Fig. 3(c) and 3(d)]. The vaterite phase of CaCO3 is known to have spherical shape. The presence of vaterite phase in the precipitates of 50 mM and 100 mM was double-checked by XRD (Fig. 2).
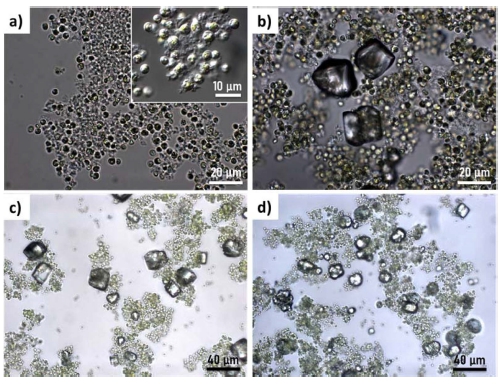
Fig. 3
Optical microscope images of microalgae-CaCO3 flocs. for (a) 10 mM, (b) 30 mM, (c) 50 mM, and (d) 100 mM.
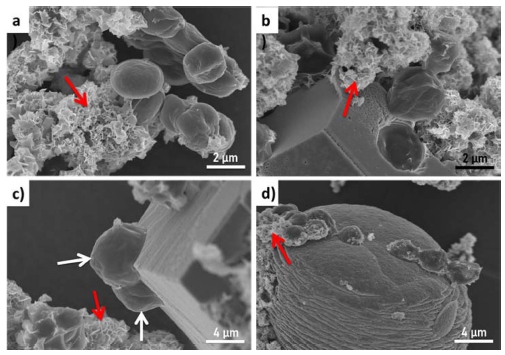
SEM images reveal the amorphous precipitates are in the shape of nano-sized flakes, which are observed in all samples as indicated by red arrows (Fig. 4). We speculate that the stability of microalgal suspension was broken by the formation of these amorphous nano-flakes, which leads to the fast sedimentation of flocs. EDS point elemental analyses and mapping were performed for one calcite crystal including microalgae and nano-flakes as shown in Fig. 5. EDS point elemental analyses on different 3 points (A for nano-flakes, B for calcite, and C for microalgal cell) confirm that the amorphous nanoflakes includes Na, P, Cl and K in addition to Ca, C and O whereas the calcite crystal is composed of Ca, C, O and little amount of Mg. Na, P, Cl and K are the main components of the modified N8 medium. The inclusion of Na, P, Cl and K is thought to inhibit the crystallization of CaCO3. Interestingly spherical microalgal cells are partly embedded in the rhombohedral calcitic crystals as indicated by white arrows [Fig. 4(c)].

Fig. 5
SEM image of microalgae-CaCO3 flocs. (30 mM) with EDS point elemental analysis and mapping. (a) SEM image, (b) EDS point elemental analysis of A, B, C in (a), EDS elemental mappings of (c) calcium, (d) carbon, (e) oxygen and (f) phosphorus.
Cross-sectional SEM images of the rhombohedral calcitic crystal after FIB-cutting clearly shows a micronsized hole generated by the embedment of a microalgal cell (Fig. 6). It is interesting from the viewpoints of mineralization as well as harvesting of microalgae by flocculation. It is a well-known strategy in biomineralization compositing inorganic single-crystal with organic matrix to improve mechanical properties of pure crystals.22-23) Organic matrix composed of protein and other biomacromolecules are often incorporated in single crystalline biominerals such as the calcite prism in mollusk shells.24) Kim et al. have synthetically showed that single calcite with well-developed crystal facets could be prepared with incorporation of 20 nm-sized micelles in the crystal matrix.25) The sharp facets of crystals with defined angles, in principle, are developed following the underlying symmetry of the unit cell, which is the most energy favorable growth of single crystal. When single calcite with the sharp facets are grown with incorporation of foreign matrix such as copolymer micelles and microalgae of this study, it means that the foreign matrix has very low surface energy enough to be occluded in calcite matrix. Kim et al. reported that micelles with anionic carboxylated chains have a negative zeta potential of -20 mV at the precipitation pH of 8-9, which leads the flocculation of micelles in the presence of Ca2+ ion by strong binding.25) In general, microalgae have negatively charged surface which enables their stable suspension and maximal utilization of light for photosynethesis. The zeta potential measurements of chlorella sp. KR-1 have shown negative value around -10 mV.26,27) This surface properties of microalgae could provide binding sites for Ca2+ ion, which further induce nucleation of CaCO3 on the surface of microalgae followed by microalgal embedment in the crystals. We speculate that not only the compatibility of microalgal surface with CaCO3 but also the amorphous to crystalline transformation mechanism facilitates embedding micron-sized cell in single crystalline calcites.28) Amorphous nano-flakes which are not phase transformed into crystals are prone to be formed in the presence of other cations such as Na+ and K+. Furthermore, the strength of charge and the presence of functional groups such as carboxyl, hydroxyl and amide groups on the surface of microalgae are dependent on the species of microalgae, which would result in different behaviors of flocculation and crystallization. Therefore, further researches should be performed for the upscale and versatile applications of mineralization-based microalgal harvesting.
4. Conclusion
Microalgae were efficiently harvested through mineralization- mediated flocculation. The harvesting efficiency was varied according to the concentration of mineral precursor, Ca2+ and CO32−: from 90 % for 10 mM to 99 % for 100 mM after 60 mins’ of gravitational sedimentation. The addition of mineral precursor into the microalgal culture resulted in the formation of 3 types of mineral precipitates: amorphous nano-flakes, rhombohedral calcites, and spherical vaterites. The nano-flakes playing the crucial role of floccuation were formed in amorphous phase due to the incorporation of other atoms than Ca2+, such as Na+ and K+, which are the main components of the culture medium. The negatively charged surface of microalgal cell is compatible not only for the nano-flakes attachment but also for the growth of calcitic crytals in which microalgal cells were embedded.