1. Introduction
The hydrogen reduction of nickel oxides has been studied extensively over past decades and many results of kinetic, mechanistic and thermodynamic analyses have been reported.1-15) Although some of the numerous investigations have reported on the use of a fluidized bed in the hydrogen reduction,13-15) the data from the literature are thought to be not sufficient for the commercial design and operation of a fluidized-bed reactor of the hydrogen reduction process. In fluidized bed reactors, the heat and mass transfer rates between gas and solid particles are high and the rapid mixing of solid leads to isothermal conditions throughout the reactor.16) This advantage, however, also leads to nonuniform residence times of solids and thus to nonuniform product at a high conversion level in a continuous operation reactor.16) Therefore, the average conversion of the product is somewhat different from what can be obtained from the reduction in a fixed or moving bed. Another problem of the fluidized bed is the agglomeration and sintering(sticking) of Ni particles at a high temperature when the size of particles is very fine. This sticking results in the defluidization of particles, and when this happens the operation should be finally stopped for the cleaning of the reactor. The main objective of this research is to obtain experimental data which could be effectively used in the commercial operation of a fluidized-bed reactor for the hydrogen reduction of nickel oxides. For this purpose, it was tried to find a best kinetic model describing this fluidized-bed reduction by the use of Avrami equation,2) as well as the optimal operational conditions. The Avrami equation is expressed as follows,
where “X” is the fractional conversion at time “t”, “k” is an overall rate constant and “m” is an exponent whose value depends on grain geometry and reaction rate control mechanism. The Avrami equation, which is applied usually to describe how solids transform from one phase(state of matter) to another at a constant temperature, can be also applied generally to the phase changes in materials, like chemical reaction rates.17)
2. Experimental
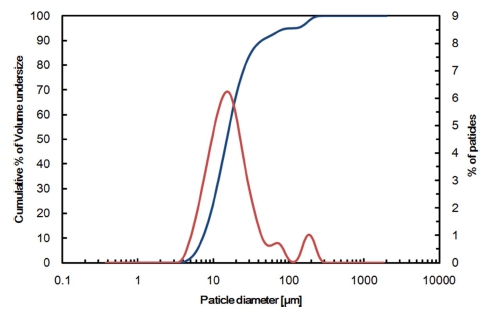
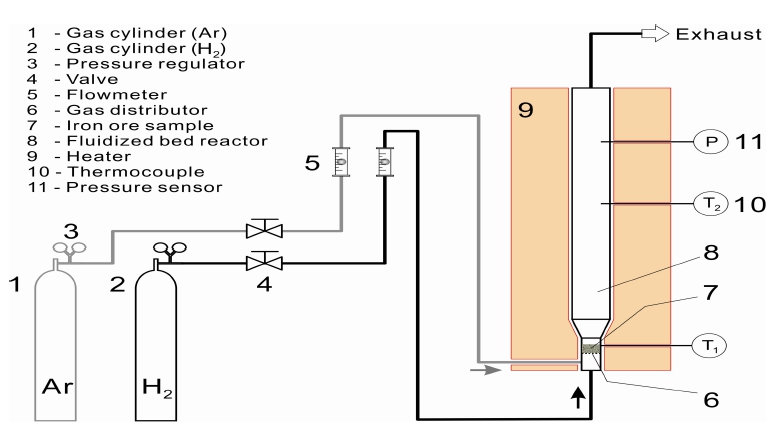
A commercial NiO(green nickel oxide, 86 wt% Ni) powder was used in this study, and the chemical composition and the particle size distribution are given Table 1 and Fig. 1, respectively. The nickel oxide particles are mostly sized in the range between 10 and 30 μm, as can be seen in Fig. 1, and has a bulk density of 2,950 kg/m3. The nickel oxide powder is made up of NiO, and the total content of other oxides such as SiO2, MgO, MnO, and Fe2O3 only counts for less than 0.7 % of the total mass. That is, the oxide sample used in this study is found to be almost pure NiO powder. The NiO powder was reduced by hydrogen gas using a batch-type fluidizedbed reactor in the temperature range from 500 to 600 °C and in the residence time range from 5 to 90 min. The sample amount was 75 g. If the total gas amount used is meaningful as in the calculation of gas consumption rate, the flow rate of hydrogen gas (as a fluidizing medium and a reductant) was varied from 3 to 6 L/min, otherwise it was fixed at 6 L/min, which corresponded to a superficial gas velocity of 0.32 Nm/s, about 4 times the minimum fluidizing velocity. The schematic diagram of experimental apparatus is shown in Fig. 2, and the fluidized bed reactor, of which inner diameter is 0.02 m, is made of SUS 310. Ar gas is used as an inert gas until the NiO powder was preheated to a targeted temperature. After every experiment, the specimen was taken out of the fluidized bed reactor and then was characterized by X-ray diffraction (XRD, D/Max 2500, Rigaku, Japan) and scanning electron microscopy (SEM, S-2400, HITACHI, Japan). The fractional conversion was identified by EDAX. The composition of Green NiO powder was analyzed by X-Ray Fluorescence Spectrometer (XRF- 1700, SHIMADZU, Japan).
3. Results and Discussion
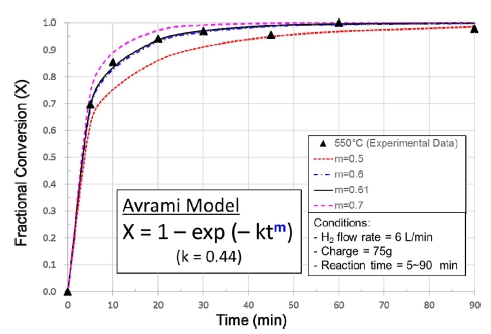
The experimental results obtained from the hydrogen reduction using the fluidized-bed reactor are shown in Fig. 3 together with Avrami models to which the experimental data are most closely fitted. According to previous investigations,7,13) it has been reported that the reaction rate increases with temperature at a low temperature range but decreases at a high temperature range due to particle agglomeration(pore blocking or sintering). This phenomenon was also observed in this study, and the boundary temperature (i.e. the maximum temperature in which NiO particles are completely reduced without agglomeration) seems to be about 550 °C, as can be confirmed from in Figs. 3 and 4. Fig. 4 shows the reduced Green NiO powders after the hydrogen reduction in the fluidized-bed reactor at various temperatures, and as can be seen in Fig. 4, the agglomeration was observed at 600 °C but not at 550 °C. Fig. 5 shows SEM images of Green NiO powder before and after hydrogen reduction in the fluidized-bed reactor at 500 °C, 550 °C and 600 °C. The NiO particles are irregular in shape with particles sizes below 10 μm. The raw NiO particles are not porous. After being reduced at 500 °C~600 °C, a small amount of micro-pores was observed on the surface of the powder. When the temperature increased to 550 °C, the size and the number of micro-pores greatly increased, indicating a substantive increase in the reduction reactivity. However, when the temperature increased further from 550 °C to 600 °C, the amount of micro-pores rather decreased as shown in Fig. 5(c)-(d). This decrease is ascribable to the sintering of the ultrafine Ni particles as reported in the previous investigation.13) Figs. 6 and 7 show the XRD patterns of the product after hydrogen reduction in the fluidized-bed reactor. As can be seen in Fig. 6, NiO could be completely reduced to Ni after more than 60 min at 550 °C. This was also confirmed from the results of the hydrogen reduction at 500 °C and 600 °C as can be seen in Fig. 7.
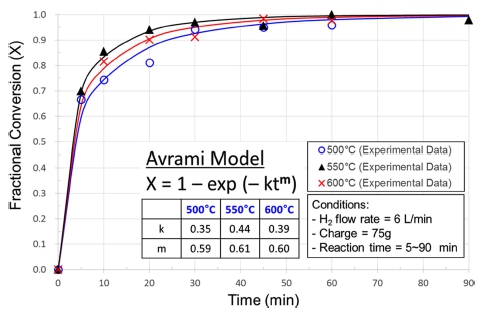
Fig. 3.
Variation of fractional conversion with time for the hydrogen reduction of Green NiO in the fluidized-bed reactor at various temperatures.

Fig. 4.
Reduced Green NiO powders after the hydrogen reduction in the fluidized-bed reactor at various temperatures.

Fig. 5.
SEM images of the Green NiO before and after hydrogen reduction in the fluidized- bed reactor at various temperatures.
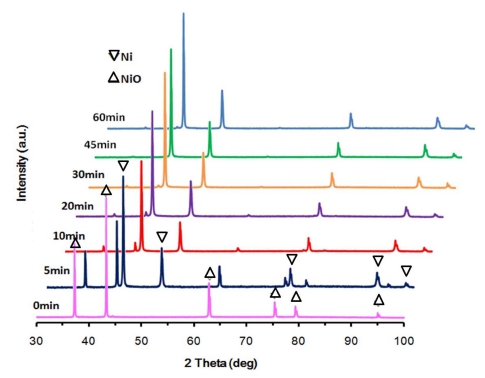
Fig. 6.
Variation of XRD patterns for the products with time of the hydrogen reduction in a fluidized-bed reactor at 550 °C.
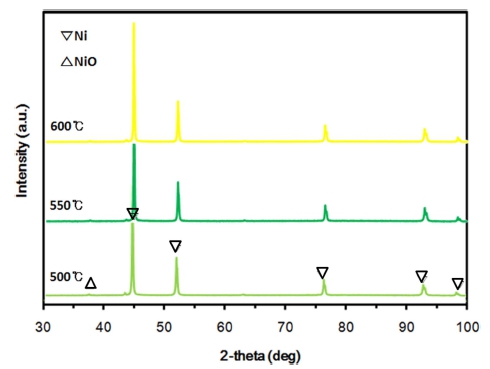
Fig. 7.
XRD patterns of the products after 60 min of hydrogen reduction in a fluidized-bed reactor in the temperature range of 500 °C - 600 °C.
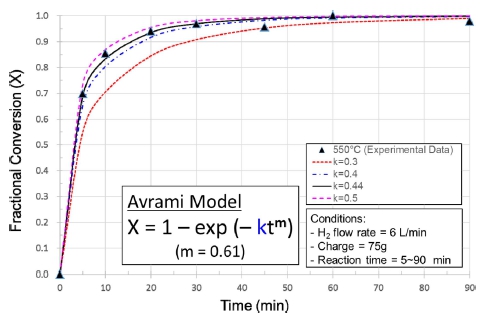
Several attempts of curve fitting were made to obtain a rate expression suitable for the hydrogen reduction of NiO in the fluidized bed. Unlike the previous investigations which were carried out at temperatures lower than 450 °C, the first order Avrami2) and the chemical reaction control models14) didn’t give satisfactory and consistent results over the temperature range from 500 °C to 600 °C. The model to which the experimental data are most closely fitted, was found to be the Avrami model, however, of very low value of the exponent “m” of 0.6 ± 0.01(low than 1), as shown in Fig. 3. Although there is no clear physical interpretation of the Avrami exponent “m”, it was known to be originally held to have a value between 1 and 4 which reflected the nature of the transformation in question.17) It has become, however, customary to regard it as an adjustable parameter that may be non-integral, and can be less than 1 in certain cases such as rapid super-cooling18) and crystallization just on a film.19) In the fluidized bed, particles move heavily in the reactor, jumping up and falling down. This mixing of concurrent and countercurrent flows can affect the kinetics of the hydrogen reduction behavior of NiO particles. In this study, as can be seen in Figs. 3 and 8, an Avrami exponent value of about 0.6 was found be best fitted for all the three temperatures, and it seems that this value may represent a characteristic of the reaction in this fluidized bed reactor. For the rate constant “k”, it was found to be a value in the range of 0.35~0.45 depending upon the temperature, as shown in Figs. 3 and 9. These values are somewhat lower than what were reported in the previous investigations,2,5,20) even if the difference in temperature range is considered. The reaction rate constant is a function of temperature and can be described by the Arrhenius relation. In principle, the rate constant increases with the temperature, however, in this study the value decreased when the temperature increased from 550 °C to 600 °C. This decrease can be ascribed to the sintering phenomenon which leads to a decrease in the area of reactive site in particles. The measurement of the rate constant at 600 °C was affected by this sintering effect, therefore, the value 0.39 cannot be taken as a true one.
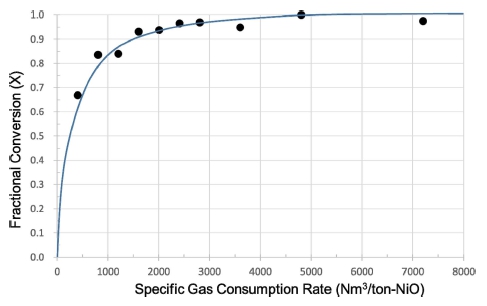
Main objective of this research is to obtain optimal operational conditions which could be effectively used in a commercial operation of a fluidized-bed reactor for the hydrogen reduction of nickel oxides. According to the experimental results of this study, it is concluded that 550 °C is the high limit of temperature and 60 min is the lowest limit of resident time. Another important condition is the specific gas consumption rate, i.e. the volume amount(Nm3) of hydrogen gas used to reduce 1 ton of Green NiO ore powder. The optimum gas consumption rate was found to be 5,000 Nm3/ton-NiO, as can be shown in Fig. 10. The data used in this figure were obtained at all the three temperatures and at different sample amounts and gas flows.
4. Conclusion
The objective of this study is to obtain experimental data which could be effectively used in a commercial operation of a fluidized-bed reactor for the hydrogen reduction of nickel oxides. In this study, it is concluded that 550 °C is the high limit of temperature and 60 min is the lowest limit of resident time. In addition, the specific gas consumption rate needed at minimum for the complete reduction was found to be 5,000 Nm3/ton-NiO.
Avrami model was applied to this study, and the experimental data are most closely fitted with an exponent ‘m’ of 0.6 ± 0.01 and with an overall rate constant “k” in the range of 0.35~0.45, depending upon the temperature. In the fluidized bed, particles are move heavily in the reactor, jumping up and falling down. This mixing of concurrent and countercurrent flows can affect the kinetics of the hydrogen reduction behavior of NiO particles. This aspect was observed in this study through the kinetic analysis using Avrami model, although the clear physical interpretation was not possible. Although the usage of the Avrami may be erroneous since “k” and “E(activation energy) are both influenced by “m”, nevertheless in this study it did give a good model to express the hydrogen reduction of NiO powder in the fluidized bed.