1. Introduction
2. Structural and Electronic Effects of Rare-Earth Doping in Ferrites
2.1. Spinel Structure and Cation Distribution
2.2. Oxygen Vacancies and Defect Chemistry
2.3. Electronic and Magnetic Modifications
3. Gas Sensing Mechanism of Ferrite-Based Sensors
3.1. Fundamentals of Gas Sensing
3.2. Effect of Rare-Earth Dopants on Sensing Performance
4. Synthesis Strategies
4.1. Sol-Gel Synthesis
4.2. Co-precipitation Method
4.3. Hydrothermal and Solvothermal Synthesis
4.4. Auto-Combustion and Solid-State Reactions
4.5. Spray Pyrolysis and Microwave-Assisted Synthesis
4.6. Thin Film Deposition Techniques
5. Sensing Performance of La- and Dy-Doped Ferrites
5.1. Lanthanum-Doped Ferrites
5.2. Dysprosium-Doped Ferrites
5.3. Comparative Analysis and Synergistic Effects
6. Practical Considerations and Future Perspectives
7. Conclusion
1. Introduction
The increasing demand for reliable detection of toxic and hazardous gases has driven extensive research into sensitive, selective, and cost-effective gas sensing materials. Metal oxide semiconductors (MOS) are widely employed for this purpose due to their high sensitivity and ease of fabrication. Among them, magnetic ferrites with spinel structures have attracted growing attention because of their chemical stability, tunable electrical and magnetic properties, and strong surface reactivity, making them suitable for gas sensing applications under both ambient and elevated temperatures.1) Spinel ferrites, with the general formula MFe2O4 (M = Ni, Zn, Co, Cu), possess a cubic crystal structure in which cations occupy tetrahedral and octahedral sites. The distribution of cations across these sites governs the electronic structure, magnetic ordering, and charge transport behavior, all of which directly influence gas sensing performance. Modifying this cationic arrangement through controlled doping is therefore an effective strategy to enhance sensor functionality.
Rare-earth (RE) doping, particularly with lanthanum (La3+) and dysprosium (Dy3+), has emerged as a powerful approach to tailor the structural and functional properties of ferrites. Owing to their larger ionic radii and unique electronic configurations, RE ions introduce lattice distortion, alter cation distribution, and promote oxygen vacancy formation. These effects enhance gas adsorption, facilitate charge carrier transport, and improve surface redox activity, leading to superior sensing performance.2,3) La3+ substitution primarily induces lattice expansion and increases defect density, enhancing surface chemical activity and electron hopping, whereas Dy3+ incorporation introduces magnetic anisotropy and modifies electronic transport through spin-related interactions. As a result, La- and Dy-doped ferrites have demonstrated improved sensitivity, selectivity, and reduced operating temperatures for a wide range of gases, including CO, NO2, H2, CH4, NH3, and volatile organic compounds. Reported studies highlight the critical roles of dopant concentration, synthesis route, particle morphology, and surface area in determining sensing efficiency. In addition, ferrites can be synthesized using scalable and low-cost methods such as sol-gel, co-precipitation, hydrothermal, and combustion techniques, facilitating their integration into conventional sensor platforms and micro-electro-mechanical systems (MEMS)-based devices. Despite significant progress, challenges related to humidity interference, long-term stability, and dopant distribution remain. Addressing these issues requires a deeper understanding of the interplay between structural, electronic, and magnetic effects induced by RE doping. Recent advances in characterization techniques, including X-ray photoelectron spectroscopy (XPS) and transmission electron microscopy (TEM), have provided valuable insights into these relationships. In this review, recent developments in gas sensors based on La- and Dy-doped magnetic ferrites are critically examined. Emphasis is placed on structure-property-performance correlations, synthesis strategies, sensing mechanisms, and practical challenges, with the aim of guiding future research and development of high-performance ferrite-based gas sensors for environmental monitoring, industrial safety, and healthcare applications.4) While previous reviews primarily address ferrite materials in a general context, the present work critically correlates RE-induced defect chemistry, magnetic anisotropy, and spin-dependent charge transport with key sensing parameters such as sensitivity, selectivity, and response dynamics. In addition, this review highlights emerging co-doping strategies and the potential of magnetically coupled sensing mechanisms, thereby offering a unified framework that bridges fundamental materials design with practical device-level considerations for next-generation gas sensors.
2. Structural and Electronic Effects of Rare-Earth Doping in Ferrites
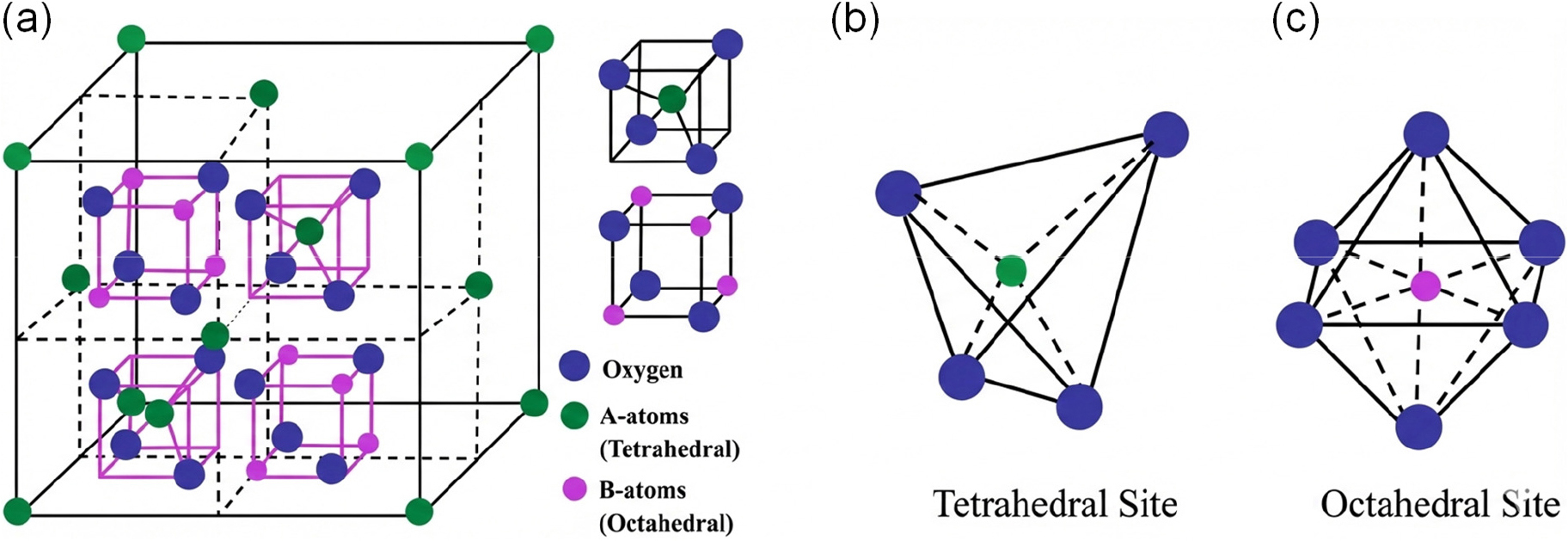
RE doping has proven to be an effective strategy for tuning the structural, electronic, and gas sensing properties of spinel ferrite materials. Spinel ferrites with the general formula AB2O4 (A = divalent metal ions such as Ni2+, Co2+, or Zn2+; B = Fe3+) possess a cubic crystal structure whose functional characteristics are highly sensitive to compositional modification. Incorporation of RE ions such as lanthanum (La3+) and dysprosium (Dy3+) significantly alters cation distribution, lattice parameters, defect chemistry, and magnetic interactions. These modifications directly influence charge transport and surface reactivity, which are critical for gas sensing performance. This section summarizes the key structural and electronic changes induced by RE doping and their implications for enhanced sensing behavior (Fig. 1).5)
2.1. Spinel Structure and Cation Distribution
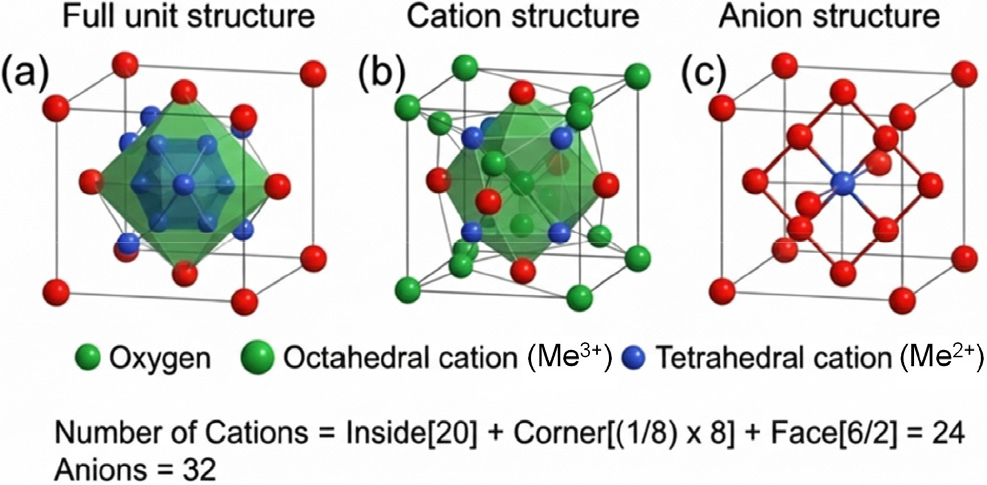
Spinel ferrites possess a face-centered cubic oxygen lattice in which cations occupy tetrahedral (A) and octahedral (B) sites. Depending on cation distribution, these materials adopt normal or inverse spinel structures, where divalent and trivalent cations occupy A and B sites in different proportions.6) This distribution is highly flexible and depends on ionic size, valence state, synthesis conditions, and dopant incorporation, enabling tunable structural and electronic properties. Incorporation of RE ions such as La3+ and Dy3+ preferentially occurs at octahedral sites through substitution of Fe3+ ions due to their identical oxidation state.7) The larger ionic radii of these RE ions induce lattice expansion and local structural strain, modifying metal-oxygen-metal bond angles and interionic distances. These distortions alter Fe2+/Fe3+ electron hopping pathways that govern charge transport, thereby directly influencing the electrical response of ferrites during gas exposure (Fig. 2). Furthermore, RE doping can disturb the equilibrium cation distribution, leading to partial inversion of the spinel structure. Such rearrangement of cations between tetrahedral and octahedral sites modifies electronic structure and magnetic ordering by altering super-exchange interactions and charge carrier mobility.8) These RE-induced changes in cation distribution and lattice symmetry play a critical role in controlling sensor sensitivity and selectivity, highlighting their importance in the design of high-performance ferrite-based gas sensing devices.
2.2. Oxygen Vacancies and Defect Chemistry
RE doping plays a crucial role in defect engineering of spinel ferrites, primarily through the generation of oxygen vacancies and related point defects. Although RE ions typically substitute Fe3+ in an iso-valent manner, differences in ionic radii and the resulting lattice distortion disrupt local electrostatic equilibrium. When dopant concentration or strain exceeds the lattice tolerance, charge compensation occurs through the formation of oxygen vacancies, which strongly influence gas sensing behavior. Oxygen vacancies act as active sites for oxygen adsorption and surface redox reactions. In ambient conditions, adsorbed oxygen species extract electrons from the conduction band, forming ionized oxygen species whose concentration depends on operating temperature.9) The presence of oxygen vacancies enhances chemisorption by providing high-energy, unoccupied lattice sites, thereby increasing surface reactivity toward target gases. In addition, these vacancies function as electron donors, increasing free carrier concentration and modifying the space charge layer near the surface. The resulting changes in carrier density and depletion layer width directly affect resistance modulation during exposure to reducing or oxidizing gases. Consequently, higher oxygen vacancy densities are commonly associated with enhanced sensitivity, lower detection limits, and faster response-recovery characteristics. RE doping can also induce secondary defects such as cation vacancies or interstitials, depending on synthesis conditions, further influencing charge transport and baseline resistance. Overall, controlled defect engineering via RE doping provides an effective strategy to optimize the sensing performance of ferrite-based gas sensors.10)
2.3. Electronic and Magnetic Modifications
RE doping induces pronounced modifications in the electronic structure and magnetic behavior of spinel ferrites, arising from the interaction between RE electronic configurations, transition-metal cations, and the local crystal field environment. From an electronic perspective, RE incorporation alters the density of states near the conduction and valence bands by introducing localized f-orbital contributions that interact with the d-orbitals of Fe3+, Ni2+, and Co2+ ions. In particular, Dy3+, with partially filled 4f orbitals, can generate mid-gap or shallow donor states that facilitate charge carrier excitation and hopping, thereby enhancing electrical conductivity.11) In contrast, La3+, possessing an empty 4f shell, does not directly contribute to magnetic ordering but influences the electronic structure through lattice distortion and dielectric polarization effects. Substitution of magnetic Fe3+ ions with non-magnetic La3+ weakens super-exchange interactions, resulting in reduced net magnetization. This controlled suppression of magnetic ordering can be advantageous in sensing environments where minimized magnetic interference is desirable. Dy3+ doping introduces strong magnetic anisotropy and modifies exchange interactions, particularly at grain boundaries. Enhanced spin polarization and increased coercivity and remanence have been reported in Dy-doped ferrites, reflecting improved magnetic stability. These magnetic effects influence electron transport through spin-dependent scattering mechanisms, which can enhance charge carrier mobility, especially at lower temperatures. Such coupling between magnetic and electronic properties can affect surface charge transfer processes and, in certain cases, enable magneto-resistive or dual-mode gas sensing behavior. Additionally, RE doping disrupts long-range magnetic ordering and introduces spin disorder, reducing the activation energy for charge transport and improving conductivity under low-temperature operating conditions. Collectively, the combined tuning of electronic structure, magnetic ordering, and defect chemistry enhances the gas sensing performance of ferrites. Careful optimization of dopant type and concentration through controlled synthesis is therefore essential to fully exploit these electronic and magnetic modifications for practical sensing applications (Fig. 3).12)
3. Gas Sensing Mechanism of Ferrite-Based Sensors
3.1. Fundamentals of Gas Sensing
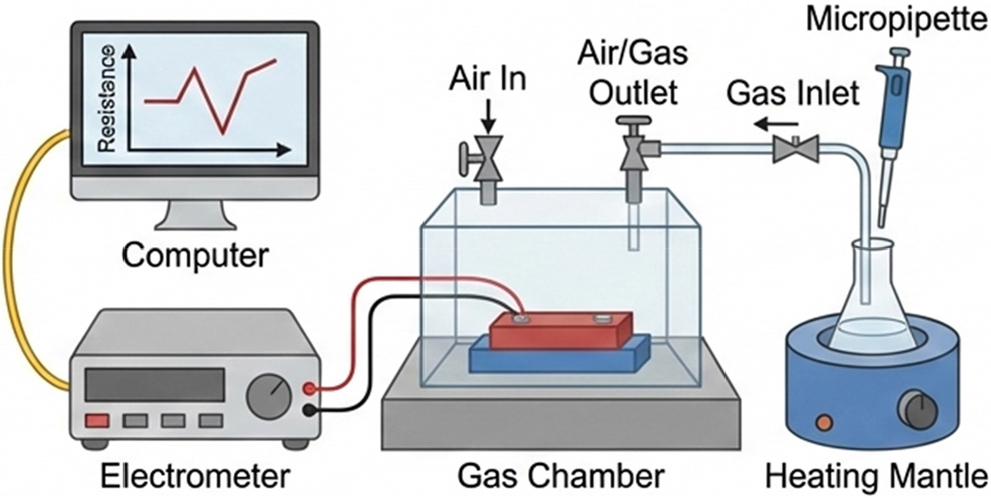
Gas sensing (Fig. 4) in n-type semiconducting ferrites is governed by resistance modulation caused by surface-mediated redox reactions between the sensing layer (Fig. 5) and surrounding gas molecules, where oxygen adsorption in air forms ionized oxygen species and an electron depletion layer that increases resistance, reducing gases such as CO, H2, or CH4 return trapped electrons to the conduction band and decrease resistance, oxidizing gases such as NO2 or O3 further extract electrons and increase resistance, and the overall sensing behavior is strongly influenced by surface area, crystallite size, defect density, operating temperature, and RE doping, which enhances oxygen vacancy concentration, modifies band structure, and improves low-temperature sensitivity.13,14,15) The gas sensor response (S) is defined consistently throughout this review to ensure clarity in performance comparison. For n-type semiconducting ferrites, the response is typically expressed as S = Rg/Ra for oxidizing gases and S = Ra/Rg for reducing gases, where Ra is the electrical resistance in air and Rg is the resistance in the presence of the target gas. This definition reflects the direction of resistance modulation arising from electron withdrawal or donation during surface redox reactions.
Grain boundaries in polycrystalline ferrites act as potential barriers to charge transport, and gas adsorption at these interfaces modifies barrier height, producing measurable resistance changes and improved response dynamics, particularly in nanostructured materials with high grain boundary density. Sensor selectivity and response-recovery behavior are additionally influenced by surface chemistry, morphology, and environmental factors such as humidity, which can compete with target gases for active sites. Overall, gas sensing in ferrite-based materials arises from dynamic surface-gas interactions coupled with charge transport modulation across grains and grain boundaries. Advances in nano-structuring and RE doping have significantly improved the sensitivity, selectivity, and operational stability of ferrite gas sensors, broadening their applicability in environmental monitoring, industrial safety, and healthcare diagnostics.16)
3.2. Effect of Rare-Earth Dopants on Sensing Performance
RE doping is an effective strategy for tailoring the physicochemical and electronic properties of spinel ferrites, thereby significantly enhancing their gas sensing performance. Among RE elements, lanthanum (La3+) and dysprosium (Dy3+) have attracted particular interest due to their distinct ionic sizes and electronic structures, which modify lattice dynamics, surface chemistry, and charge transport behavior critical to sensing processes.17) Substitution of Fe3+ ions with La3+ or Dy3+ induces lattice distortion arising from ionic radius mismatch, leading to the formation of oxygen vacancies. These vacancies act as active adsorption sites for oxygen and target gas molecules, promoting surface redox reactions and enhancing modulation of the electron depletion layer during gas exposure. Increased oxygen vacancy density improves electron exchange at the surface, resulting in higher sensitivity and faster response-recovery characteristics. In particular, La3+ doping modifies the space-charge region and increases electron donor density, thereby enhancing sensor response toward reducing gases such as H2, CH4, and CO while enabling lower operating temperatures. In addition, La3+ incorporation can suppress grain growth during synthesis, increasing surface area and strengthening gas-solid interactions.18)
In contrast, Dy3+ doping introduces magnetic and electronic effects associated with its partially filled 4f orbitals. Dy3+ ions modify magnetic exchange interactions and induce magnetic anisotropy, which influences grain boundary transport and spin-dependent charge carrier mobility. These effects enhance electrical conductivity and accelerate signal transduction, leading to improved response speed and thermal stability, particularly under fluctuating operating conditions. As a result, Dy-doped ferrites exhibit superior sensitivity toward gases such as CO and LPG and demonstrate stable performance in high-temperature environments.19) RE dopants also tend to segregate at grain boundaries and surfaces, forming RE-enriched regions that act as catalytically active sites. This surface enrichment improves gas selectivity, with La3+ favoring interactions with acidic gases such as NO2 and Dy3+ enhancing responses toward reducing gases. Furthermore, RE doping influences particle morphology and porosity by controlling grain size and pore distribution through synthesis routes such as sol-gel, hydrothermal, and co-precipitation methods. Reduced grain size and interconnected porosity facilitate rapid gas diffusion and improve adsorption-desorption kinetics, directly enhancing sensor dynamics.20) Experimental studies confirm these effects, with La-doped ZnFe2O4 and NiFe2O4 showing enhanced sensitivity and selectivity toward ethanol, methanol, and NH3, while Dy-doped CoFe2O4 and NiFe2O4 exhibit improved responses to CO and LPG. Co-doping strategies combining La and Dy further demonstrate synergistic improvements in sensitivity, linearity, and long-term stability. Moreover, RE doping generally lowers optimal operating temperatures, enabling energy-efficient operation and compatibility with flexible and portable sensing platforms. However, excessive dopant concentrations may lead to secondary phase formation or dopant agglomeration, underscoring the need for precise compositional control. Overall, La3+ and Dy3+ doping provides a powerful pathway to enhance sensitivity, selectivity, response speed, and thermal stability in ferrite-based gas sensors through combined structural, electronic, and magnetic modifications.21)
4. Synthesis Strategies
The fabrication of RE-doped ferrite nanomaterials for gas sensing applications requires careful control over both the chemical synthesis route and the resulting microstructural and surface properties. A variety of synthesis techniques have been developed and optimized to produce ferrites with controlled stoichiometry, morphology, crystallinity, and dopant distribution. The selection of a suitable synthesis method significantly influences the material’s gas sensing performance, as parameters such as surface area, particle size, porosity, and defect states are highly sensitive to the preparation route. In this section, we detail the most commonly employed synthesis strategies for La- and Dy-doped ferrites and the corresponding characterization techniques used to analyse their structure and properties.22)
4.1. Sol-Gel Synthesis
The sol-gel method is a versatile and widely adopted technique for synthesizing doped ferrite materials at relatively low temperatures. This method involves the hydrolysis and condensation of metal alkoxides or metal salts in solution to form a homogeneous sol, which eventually evolves into a gel network. The gel is then dried and calcined to produce crystalline ferrite powders. For RE-doped ferrites, this method enables uniform mixing of precursor ions at the molecular level, thereby ensuring homogeneous incorporation of dopants such as La3+ and Dy3+ into the spinel lattice.23) The sol-gel method offers the advantage of precise stoichiometric control and typically yields fine-grained, high-surface-area powders, which are desirable for gas sensing due to enhanced surface reactivity.
4.2. Co-precipitation Method
The co-precipitation technique involves the simultaneous precipitation of metal ions from a homogeneous aqueous solution by adjusting the pH with a base such as NaOH or NH4OH. This method is simple, cost-effective, and suitable for producing large quantities of ferrite nanoparticles. During this process, RE ions can be effectively doped by co-precipitating them with other transition metal ions, leading to the formation of mixed hydroxide precursors. These precursors are subsequently washed, dried, and calcined to yield the final doped ferrite materials.24) Co-precipitation provides good control over particle composition and allows tuning of the material properties by modifying synthesis parameters such as pH, temperature, and precursor concentration.
4.3. Hydrothermal and Solvothermal Synthesis
Hydrothermal synthesis involves treating aqueous solutions of metal salts under elevated temperature and pressure in a sealed autoclave. This method allows the growth of highly crystalline nanostructures with well-defined morphologies and tunable particle sizes. When doped with RE elements, ferrite materials synthesized via hydrothermal methods often exhibit improved crystallinity and reduced grain boundaries, which are beneficial for stable and reproducible gas sensing.25) Solvothermal methods are similar in principle but use organic solvents instead of water, enabling the synthesis of unique nanostructures and complex morphologies.
4.4. Auto-Combustion and Solid-State Reactions
Auto-combustion synthesis, also known as the self-propagating high-temperature synthesis (SHS) method, involves the exothermic reaction of a homogeneous precursor mixture containing metal nitrates and a fuel such as urea, glycine, or citric acid. Upon ignition, the fuel undergoes rapid combustion, releasing heat that facilitates the formation of the ferrite phase. This method is particularly advantageous for producing highly porous, low-density powders with high surface activity.26) Solid-state reactions, although involving high temperatures and extended calcination times, are still employed for bulk production of ferrites due to their simplicity and scalability. However, achieving uniform dopant distribution is more challenging with this method.
4.5. Spray Pyrolysis and Microwave-Assisted Synthesis
Spray pyrolysis is a technique wherein a precursor solution is atomized into fine droplets and passed through a high-temperature zone, resulting in the formation of spherical ferrite particles. This method offers excellent control over particle size and morphology. Microwave-assisted synthesis, on the other hand, provides rapid heating and short reaction times, enabling the synthesis of nanocrystalline ferrites with high purity and controlled size distribution. When combined with doping strategies, these techniques can enhance the material’s surface area and defect concentration, both of which are critical for gas sensor sensitivity.27)
4.6. Thin Film Deposition Techniques
For the development of ferrite-based gas sensors integrated into electronic devices, thin film fabrication methods such as pulsed laser deposition (PLD), sputtering, and chemical vapor deposition (CVD) are employed. These techniques allow the formation of high-quality thin films with controlled thickness, surface uniformity, and dopant incorporation. The films can be directly deposited onto sensor substrates, eliminating the need for additional binder or sintering steps.28,29,30) The precise control over microstructure in these techniques leads to improved device reproducibility and performance.
5. Sensing Performance of La- and Dy-Doped Ferrites
RE dopants play a pivotal role in fine-tuning the structural and electronic characteristics of ferrite-based gas sensors. Among the various dopants, lanthanum (La) and dysprosium (Dy) have garnered considerable attention due to their distinctive influence on the gas sensing behavior of ferrite materials. This section comprehensively explores the individual and comparative sensing performance of La- and Dy-doped ferrites, highlighting key mechanisms, gas selectivity, temperature dependence, and sensing efficiency.
5.1. Lanthanum-Doped Ferrites
La3+ doping in ferrite systems such as NiFe2O4, ZnFe2O4, and CoFe2O4 significantly enhances gas sensing performance through lattice expansion, oxygen vacancy generation, and electronic structure modification.31) The larger ionic radius of La3+ promotes defect formation and surface activation, leading to improved chemisorption of gas molecules and enhanced charge transport. As a result, La-doped ferrites commonly exhibit increased sensitivity and reduced operating temperatures. La-doped ZnFe2O4 has shown particularly strong responses toward reducing gases such as ethanol, methanol, and ammonia, attributed to increased surface basicity and a higher density of oxygen vacancies that facilitate rapid surface redox reactions.32) La incorporation also suppresses grain growth during synthesis, producing smaller crystallite sizes and increased porosity, which improve gas diffusion and adsorption kinetics. However, sensing performance strongly depends on dopant concentration; excessive La content can lead to dopant clustering or secondary phase formation, reducing sensitivity due to diminished electron mobility and surface activity. Optimal La concentrations typically lie in the range of 2-3 mol%, where maximum response and selectivity are achieved.
5.2. Dysprosium-Doped Ferrites
Dy3+ doping introduces additional magnetic and electronic effects that distinctly influence gas sensing behavior. Owing to its partially filled 4f orbitals, Dy3+ modifies magnetic exchange interactions, spin polarization, and grain boundary transport, thereby affecting charge carrier mobility and resistance modulation.33) Dy-doped ferrites such as NiFe2O4, CoFe2O4, and MgFe2O4 have demonstrated enhanced sensitivity toward gases including CO, LPG, and H2. In Dy-doped NiFe2O4, performance enhancement arises from the combined effects of increased oxygen vacancy concentration and improved magnetic ordering, which facilitate efficient surface redox reactions and stable electron transport. Experimental studies report significantly improved sensitivity, shortened response-recovery times, and excellent repeatability over extended testing cycles.34) Similar trends are observed in Dy-CoFe2O4 systems, where enhanced magnetic interactions contribute to stronger resistance modulation upon gas exposure. Additionally, Dy doping improves thermal and chemical stability, making these materials suitable for high-temperature sensing environments. Particle size refinement and improved crystallinity induced by Dy incorporation further support enhanced gas adsorption and sensing reliability.
5.3. Comparative Analysis and Synergistic Effects
When comparing the sensing behaviors of La- and Dy- doped ferrites, several distinctions and complementary characteristics emerge. La3+ doping predominantly contributes to chemical activity enhancements, especially toward polar and acidic gases like ethanol and ammonia. The introduction of La3+ improves selectivity and reduces the activation energy required for gas adsorption and reaction. This makes La-doped ferrites particularly suitable for low-temperature gas sensing applications where operational energy efficiency is a priority. On the other hand, Dy3+ doping primarily enhances the magnetic and electronic transport properties, contributing to improved sensitivity and faster electron exchange mechanisms. The magnetic nature of Dy3+ ions leads to alterations in the spin-polarized transport and promotes grain boundary scattering effects, which can be exploited for detecting gases like CO and LPG. Interestingly, recent research has explored the concept of co-doping ferrites with both La3+ and Dy3+ ions to achieve synergistic improvements in sensing performance. The rationale behind this approach is to combine the surface activity enhancements of La with the magnetic tuning effects of Dy. In one such study, La-Dy co-doped NiFe2O4 exhibited a broader gas detection range, superior linear response characteristics, and faster recovery times compared to singly doped counterparts. This co-doping strategy opens up new possibilities for designing multi-functional sensors with tailored selectivity and performance.35) Another important aspect of comparative analysis is the dopant concentration. Both La and Dy have optimal doping levels (typically 1-3 mol%), beyond which negative effects such as lattice distortion, phase segregation, and reduced electron mobility can occur. Thus, precise control over dopant concentration and synthesis parameters is essential for achieving the desired sensing characteristics. Moreover, the influence of dopants on other physical properties such as surface area, porosity, grain size, and thermal conductivity should also be considered in sensor design. For example, La-doped ferrites tend to exhibit higher porosity and surface area, which are advantageous for gas adsorption, whereas Dy-doped materials often display enhanced thermal and magnetic stability. Both La and Dy dopants offer distinct yet complementary pathways for enhancing the gas sensing performance of ferrite materials. Through careful selection and optimization of dopant type, concentration, and synthesis methodology, it is possible to tailor the sensing properties of ferrite-based sensors to meet specific application requirements. Further research into co-doping strategies and the integration of these materials into sensor devices will be key to realizing their full potential in practical environmental and industrial monitoring systems (Table 1).
Table 1.
The influence of dopant type, synthesis route, and operating conditions on sensing performance.
6. Practical Considerations and Future Perspectives
RE-doped ferrite gas sensors, particularly La- and Dy-based systems, show strong potential for high-performance gas detection; however, several practical challenges remain for real-world deployment. Sensitivity to environmental humidity is a major limitation, as water molecules compete with target gases for active sites, causing signal drift and reduced selectivity. Hydrophobic surface modification, composite sensing layers, and humidity-compensated signal processing are therefore essential. Dopant inhomogeneity, segregation, and long-term drift arising from grain growth, surface contamination, and oxygen vacancy annihilation can compromise stability during prolonged operation. Advanced synthesis methods with improved dopant control, combined with nanostructure engineering and surface passivation, are required to enhance durability and reproducibility. Nanostructuring and catalytic functionalization further improve sensitivity and selectivity by increasing surface area and reducing reaction energy barriers.
The integration of RE-doped ferrites into flexible and wearable platforms, along with data-driven sensing using artificial intelligence and machine learning, represents a promising direction for next-generation sensor systems. Future studies should also explore alternative RE dopants, co-doping strategies, and sustainable synthesis routes to minimize environmental impact. Overall, overcoming challenges related to stability, selectivity, and scalability will be key to translating RE-doped ferrite gas sensors from laboratory research to practical applications in environmental monitoring, industrial safety, and healthcare diagnostics.
7. Conclusion
RE-doped spinel ferrites have emerged as highly promising materials for gas sensing owing to their tunable structural, electronic, and surface properties, with La3+ and Dy3+ doping offering complementary benefits through lattice distortion, modified charge transport, oxygen vacancy formation, and improved sensing performance; La3+ primarily enhances surface chemical activity, lowers operating temperature, and improves response-recovery behavior, whereas Dy3+ promotes magnetic anisotropy and spin-related transport, thereby improving sensitivity and stability, while co-doping further strengthens synergistic effects for broader detection capability and long-term reliability, although challenges such as humidity interference, dopant segregation, and signal drift still require attention through optimized composition, nanostructuring, surface functionalization, flexible low-power integration, and data-driven sensing strategies. Future research in RE-doped ferrite gas sensors should focus on several critical directions to advance both fundamental understanding and practical applicability, including operando characterization techniques such as in situ X-ray photoelectron spectroscopy and impedance spectroscopy under gas exposure to directly probe dynamic surface reactions and charge transfer processes, exploration of magneto-resistive and spin-dependent sensing mechanisms particularly in Dy-doped systems for multifunctional sensor development, device-level validation under real-world conditions including variable humidity, temperature fluctuations, and long-term operational stability to bridge laboratory-scale studies and commercial deployment, and integration with flexible electronics and machine learning-assisted sensing for next-generation sensor technologies.