1. Introduction
A perfect margin is the most important factor among the requirements of fixed dental prosthodontics, such as crowns and bridges.1,2) A loosened margin can cause secondary caries, which can adversely affect the longevity and the durability of fixed crowns and bridges.3) To obtain accurate impressions before undertaking the procedure for fixed prosthodontics, temporary gingival retraction should be achieved by creating sufficient space in the gingival sulcular area. A thorough, comprehensive understanding of the interdependence between periodontal tissues and crowns is crucial for successful prosthetics in dentistry.
Elastomers such as polyvinyl siloxane (PVS) are excellent impression materials for fixed prostheses.4,5) PVS is known for its high accuracy in dental impression-taking. However, the hydrophobic nature of PVS can pose challenges to achieving accurate impressions. Furthermore, the quality of the impression can be significantly influenced by various clinical parameters, including the location of the finish line and the presence of sulcus bleeding during impression-taking. Gingival retraction cords are used to obtain accurate and fitting margin impressions, specifically in cases where the finish line is located subgingivally.6,7) There are several retraction cord techniques available, including mechanical and chemical displacement, or a combination of both.7) Generally, the gingival retraction cord is placed between the junctional epithelium and cemento-enamel junction and is primarily used for tissue management, such as displacing gingival tissues away from the preparation margins.3,8,9)
During cord placement, damage to the junctional epithelium and bleeding may occur due to its anatomical structure. In PVS, bleeding control is essential to obtaining an accurate impression. For bleeding control, hemostatic agents are commonly used in clinical practices, either separately or as coatings on retraction cords. However, the safety and potential risks associated with these agents have been a topic of debate since their inception.10,11) Among the various hemostasis agents, aluminum chloride hexahydrate is preferred for its gentle astringent and hemostatic properties, and its safety with no associated cardiovascular problems. However, there is a lack of research on impregnated gingival retraction cords. In addition to the disadvantages associated with chemical methods, placement of a retraction cord under gingival tissue irritates and induces an inflammatory reaction.12)
In previous studies, an impregnated gingival cord prepared according to ISO 10993-12 for biocompatibility testing showed severe cytotoxicity at its original dose.13) However, the placement time of the gingival retraction cord is short and varies depending on the clinical situation, as the cord is removed soon after taking impressions, with placement time ranging from over 10 min to less than 1 h.14) Therefore, we considered the clinical exposure time required for a 50 % reduction in cell or tissue viability (ET50) following exposure when compared to a negative control using immortalized human gingival fibroblasts (hTERT-hNOF) and immortalized human oral keratinocytes (IHOK). In addition, Kwon et al. analyzed cytokine release in the gingival tissue caused by dental luting cement and compared cell viability and cytokine release after exposure to various luting cements.15) Their study suggested a correlation between cell viability and cytokine release. Considering all the aspects mentioned previously, this study aimed to evaluate the cytotoxicity and release of interleukin (IL)-1α and IL-8 induced by impregnated gingival cords in hTERT-hNOF and IHOK.
2. Experimental Procedure
The material tested was a gingival retraction cord impregnated with aluminum chloride hexahydrate (Sure Cord Plus #00 size, Sure Dent, Seongnam, Korea). The gingival retraction cords were weighed and placed in sterile glass vials. To prepare the extracts, the test material was immersed in a cell culture medium DMEM/F12 (Gibco, Grand Island, NY, USA) for 24 h at 37 °C based on ISO 10993-12 (2012). Before using the extracts, the supernatant was removed, added to a new sterilized vial, and the pH was measured using a pH meter (Orion 4 Star; Thermo Fisher Scientific, Inc., Singapore). A serum-free cell culture medium without the sample was also incubated under the same conditions as the tested material extracts and used as a negative control.
In this study, immortalized human gingival fibroblasts (hTERT-hNOF) and immortalized human oral keratinocytes (IHOK) were used. hTERT-hNOFs cells were derived from gingival fibroblasts, primarily cultured from healthy human adults, and transfected with the puromycin-resistant retroviral vector plpc-hTERT (Clontech Laboratories, Palo Alto, CA, USA). IHOK cells were obtained by immortalizing human gingival epithelial cells by transfection of the pLXN vector containing the E6/E7 open reading frame of HPV type 16, as previously described.16,17)
The hTERT-hNOF and IHOK cells were cultured in a mixture of Dulbecco’s Modified Eagle’s Medium and Ham’s Nutrient Mixture F-12, with a 3:1 ratio (DMEM/F12 3:1; Gibco, Grand Island, NY, USA), supplemented with 10 % fetal bovine serum (Gibco), and 1 % penicillin/streptomycin (Invitrogen, Grand Island, NY, USA), at 37 °C in a fully humidified atmosphere with 5 % CO2.18)
After 24 h, the cell culture medium was removed and extracts of the material were added. After each endpoint, the supernatants were collected, and cell viability was evaluated using an MTT (3-[4,5-dimethylthiazol-2-yl]-2,5 diphenyl tetrazolium bromide) assay based on ISO 10993-5.
To evaluate cytokine release, 50 µL of the supernatant from each cell was allowed to react with 50 µL of either mouse anti-human IL-1α or IL-8 (R&D System, Minneapolis, MN, USA) in a 96-well plate for 2 h. After the reaction, the cells were washed three times with 0.05 % Tween 20 in phosphate-buffered saline (PBS) and treated with 50 µL of biotinylated goat anti-human IL-1α or IL-8. After washing with 0.05 % Tween 20, 50 µL of streptavidin conjugated to horseradish peroxidase was added. After a final wash with Tween 20, the color was allowed developed using tetramethylbenzidine, and the reaction was stopped using 2N H2SO4. Absorbance was measured at 450 nm using an ELISA (Epoch, BioTek Instruments, VT, USA). Cell viability and cytokine release were analyzed using a one-way ANOVA (PASW 18.0, IBM Co., NY, USA), and the Tukey’s test was performed as a post-hoc test (p = 0.05).
3. Results and Discussion
For a well-fitted restoration, the influence of the retraction and impression techniques is critical to the displacement of tissues. Clinically, gingival retraction is used in a wide range of applications in dentistry to expose the subgingival crown margin.19) Although various materials have been introduced, retraction cords are commonly used as they are cost-effective and provide adequate gingival displacement.
The retraction cords were divided into non-medicated and medicated groups. Non-medicated cords are safe but have limited potential to control bleeding and fluid seepage. Medicated cords are more effective, but their systemic toxicity and deleterious influences on periodontal tissue are a concern.20) To the best of our knowledge, both types of cords can stimulate and irritate the gingival tissue physically or chemically.7)
Medicated cords are impregnated with chemicals such as aluminum potassium sulfate, aluminum chloride, epinephrine, zinc chloride, ferric sulfate, and sympathomimetic amines.9) The retraction agents, which are solutions with highly acidic properties, possess pH values ranging from 0.8 to 3.0. This acidity level poses a potential risk to the periodontal tissue.21) For the cytotoxicity test, the cords are extracted according to international standards for the biological evaluation of medical devices, and the extracted cords have low acidity.13)
During the placement of the impregnated cord, the sulcus, especially the junctional epithelium, is irritated, causing lysis of fibroblasts.11,22) The cytotoxicity of various chemical retraction agents has been tested in fibroblasts in vitro, and most retraction agents produced strong toxicity, with 25 % aluminum chloride being the strongest.10,11) Some side effects have been investigated, including tissue inflammation and cell viability with impregnated cords.23) There is a direct relationship between placement time and damage to sulcus tissue, and cord placement with 4 % epinephrine and 25 % aluminum chloride for up to 3 min in the gingival sulcus does not cause gingival recession. However, prolonged cord placement for three minutes or more causes gingival recession in dogs.24)
The junctional epithelium is a type of stratified squamous epithelium that is non-keratinized. It is located directly beneath the sulcular epithelium, which lines the gingival sulcus from its base to the free gingival margin.25) Thus, the toxicity of various hemostatic agents has been reported in fibroblasts,26) and toxicity tests have mainly been conducted on fibroblasts. Although these fibroblasts were the primary cell lines, we used two immortalized human oral cell lines, hTERT-hNOF and IHOK, to mimic the oral environment.
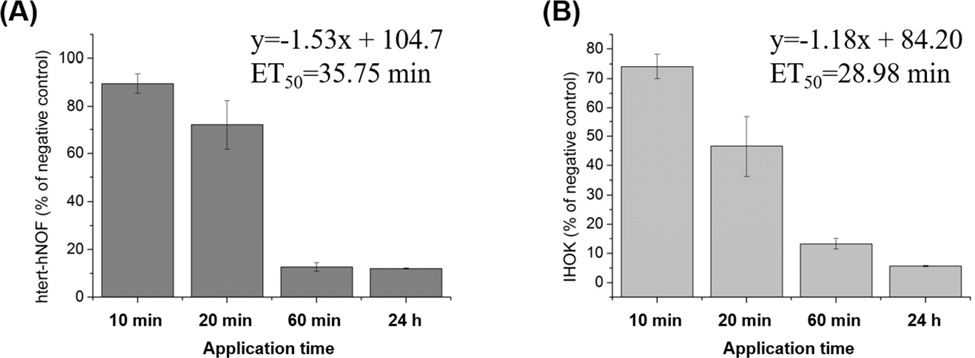
The results of the cell viability test measured using the MTT assay are shown in Fig. 1. The cell viability after exposure to the extract solution for 10 min exceeded 70 % in both cell types. The ET50 values for hTERT-hNOF and IHOK were 35.75 and 28.98 min, respectively.
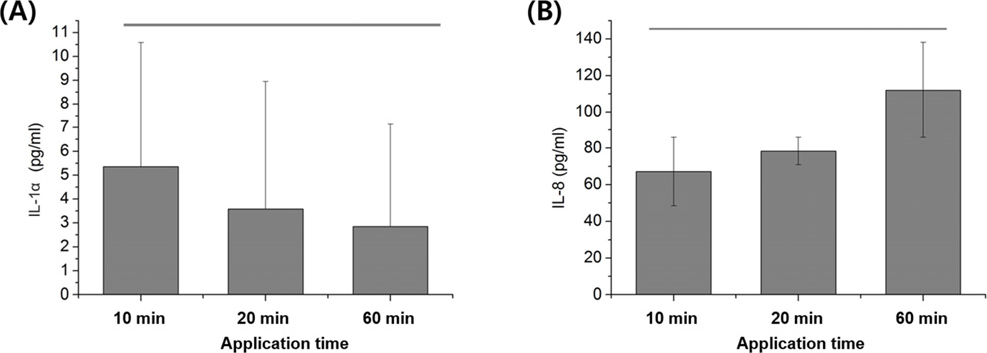
The results of the cytokine release test for IL-1α and IL-8 from IHOK following exposure to the gingival retraction cord are shown in Fig. 2, respectively. For IHOK, the IL-1α levels were 5.35 ± 5.22 pg/mL at 10 min, 3.58 ± 5.38 pg/mL at 20 min and 2.85 ± 4.28 pg/mL at 60 min of exposure. Meanwhile, the IL-8 levels were 67.16 ± 18.70 pg/mL at 10 min, 78.36 ± 7.50 pg/mL at 20 min, and 111.9 ± 26.10 pg/mL at 60 min of exposure. However, no significant differences were observed between the cells exposed for different periods (p > 0.05). Moreover, no cytokine release was detected in hTERT-hNOFs. Interestingly, no or very low levels of interleukin were released from hTERT-hNOF cells in previous studies.15,27)
Following the placement of the retraction cord, the gingival tissue sustained severe injury, and at the same time, an inflammatory reaction with interleukin release could be detected. This indicates the toxicity of the medicated cord and its chemical reaction (Fig. 3). Additionally, it can be used to describe clinical tissue reactions.
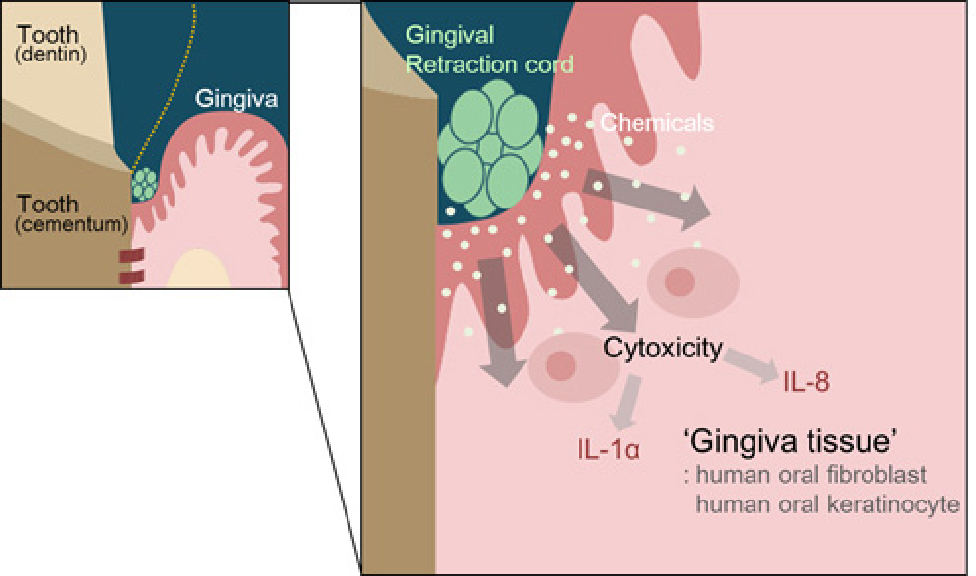
Fig. 3
The green gingival retraction cord was placed for retraction gingival tissue around the prepared tooth. The chemicals from the gingival retraction cord may influence the fibroblasts and keratinocytes and the viability of cells may be affected. The affected cells may secrete cytokines such as interleukins 1 alpha and 8.
Medical devices undergo toxicity tests, which are usually conducted in two dimensions. However, these tests tend to maximize the results compared to clinical or physiological reactions. Although some dental materials have been used for several decades in dentistry, they have been shown to exhibit high toxicity in vitro. Therefore, we decided that a new definition of toxicity was necessary. The ET50, which is the amount of time required for onset of destruction, is a novel way for us to quantify toxicity. The shorter the ET50, the more toxic a compound is considered.14,28)
In our study, we performed the extraction according to the recommended standard, and the ratio was 0.1 g of cord immersed in 1 mL of cell culture medium. In addition, the tested material contained 0.05 g of aluminum chloride hexahydrate in 10 inches of the impregnated cord, according to the manufacturer’s specifications. The cell viability test revealed high acidity and its toxicity to the two cell types. In our study, the MTT assay with a 24 h exposure revealed severe toxicity, but with a 10 min exposure, cell viability was over 70 %. Similar results have been reported in a previous study.11) However, cytokine production did not significantly differ between exposure times. It is well known that oral mucosa cells can produce both IL-1α and IL-8 even without stimulation, and both cytokines were released even in the negative control. IHOK are known as major producers of IL-1α, which regulates the release of other cytokines.29-31) IL-8 then promotes the release of chemicals for wound healing through controlling the expression of other cytokines and various growth factors.32,33) The roles and interactions between each cytokine are still unclear. Although Kwon et al. tried to explain the relationship between cytotoxicity and cytokine release, their explanations were inadequate. Currently, molecular investigations, such as real-time quantitative reverse transcription polymerase chain reaction, are being conducted on cytokines like IL-1α, IL-8, and TNF-α to explain this relationship.15) However, these further investigations are outside the scope of our paper.
However, studies on gingival retraction and medicated cords is still limited. In addition, more studies on ET50 are needed to compare prior toxicity scales and concepts. A better understanding of cytokine release is required for its application in biocompatibility evaluations for clinical applications. Although an impregnated dental gingival retraction cord is a useful dental material, various tests should be conducted.
4. Conclusion
In this study, cell viability and cytokine release in two human oral cell lines were measured after the application of extracts of dental impregnated gingival retraction cords to investigate the possibility of using them for biocompatibility testing. These results confirmed the cell viability and cytokine release in cells exposed to the impregnated gingival retraction cord. In addition, the application of extracts to hTERT-hNOF and IHOK during the actual contact period and determination of ET50 may be beneficial for evaluating the biocompatibility of dental-impregnated gingival retraction cords.