1. Introduction
Lithium and lithium compounds have widely drawn attention as representative materials for the 21st century, due to their variety of applications in many fields such as lithium batteries, refrigerant adsorbents, computers, mobile phones, pharmaceuticals, alloying elements, catalysts, etc. Lithium is primarily recovered from ore, mines and salt lakes. Its land reserves have been reported to be about 4 million tons, but geographically highly-concentrated.1) On the other hand, the total amount of lithium in seawater is estimated to be about 2,300 hundred million tons, 60,000 times greater than its land reserves,2) and thus many researches have been in progress for the lithium recovery from seawater.
The well-known techniques to separate and recover lithium from seawater are co-precipitation,3) solvent extraction, 4) and ion exchange.5-7) However, co-precipitation is limited on the selectivity of metal ions and solvent extraction requires a large quantity of organic solvents, which leads to environmental pollution, offensive odor, and fire.8) On the other hand, ion exchange process is favored on its low sludge production and low operation cost along with the regeneration of its chemically stable resins.
Many researches for removal and/or recovery of metal ions in aqueous solution by ion exchange have been carried out. Wang et al. reported the removal of Cd(II) by using a strong-acid cation exchange resin (001x7) with HCl and NaOH treated.5) The removal of Cr(VI) by using a Na+ type ion exchange resin (Lewatit FO36) was investigated by Rafati et al.,7) and then, Alyuz and Veli removed Zn2+ and Ni2+ by another Na+ type ion exchange resin (Dowex HCR S/S).8) These researches have shown that various types of ion exchange resins are available for efficient removal and/or recovery of heavy metal ions in aqueous solution. However, the recovery of light metal ions such as lithium ion (Li+) has not been done until a recent report by Navarrete-Guijosa et al.6) that a strongacid cation exchange resin (Amberlite IR120) exchanged with Na+ and H+ is capable of lithium recovery in aqueous solution and Na+ exchanged one has higher ion exchange capacity.
The capacity of ion exchange resin is generally related to its functional group, cation form, and porous structure. In the present work, the characteristics of lithium recovery by ion exchange was thus reviewed by changing function group (-SO3H and -COOH), porosity(porous and gel types), and cation form (K+, Na+, and H+) of ion exchange resins. Moreover, equilibrium isotherm and kinetics as well as the effect of pH were also investigated by batch type experiments.
2. Experimental
Three commercial ion exchange resins were bought from a domestic company, a porous type strong acid cation exchange resin(CMP28) having -SO3H as functional group, a gel type strong acid cation exchange resin(SCRB) with the same -SO3H functional group, and a porous type weak acid cation exchange resin(WK60L) having -COOH as functional group. Properties of those ion exchange resins are summarized in Table 1.
Table 1.
Properties of ion exchange resins used in this study.
To change the cation form of resins into K+, Na+ and H+, 1 M aqueous solutions were prepared from KCl (Junsei, EP), NaCl (Shinyo, GR), and HCl (Samchun, EP), respectively. Then, 200 mL of each prepared solution was mixed with 2 g of each resin and stirred at a speed of 250 rpm in a gyratory shaker (Johnsam, JS-FS-2500) for 24 hr. This step was repeated three times with new solution, and finally reacted resins were washed with distilled water several times before ion exchange experiments. The lithium chloride (LiCl, Shinyo, EP) was diluted with ultrapure water (Milli-Q Millipore, 18.2 MΩ·cm conductivity) to obtain a stock solution of 1,000 mg/L lithium ion concentration.
Adsorption experiments were carried out batch wise. A 0.2 g ion exchange resin and 200 mL aqueous lithium solution were put into 500 mL Erlenmeyer flask and stirred at 200 rpm for 12 hr at 80 °C. The initial lithium ion concentration was varied between 10 and 100 mg/L by using the stock solution. Samples were taken by a certain period of time and centrifuged by a centrifuge (Centrifuge 5415C, Germany) at a speed of 10,000 rpm for 5 min. The lithium ion concentration of supernatant liquid was then measured by ion chromatography (DX- 120, Dionex). The pH of aqueous lithium solution was controlled by 0.1 M NaOH and 0.1 M HCl solutions and measured by a pH meter (Orion, Model 420A).
The ion exchange capacity, qt(mg/g), was calculated by the following equation:
where C0 and Ct(mg/L) are initial concentration and the concentration of lithium ion at any time t, respectively. V(L) is the volume of aqueous lithium solution and m(g) is the amount of ion exchange resin.
3. Results and Discussion
3.1. Effect of Resin Type
Fig. 1 illustrates ion exchange capacities of aqueous lithium recovery for three commercial ion exchange resins (CMP28, SCR-B, and WK60L) and their derivatives with cations exchanged with K+, Na+, and H+. As clearly shown in Fig. 1, strong acid cation exchange resins (CMP28 and SCR-B) have higher ion exchange capacities of 3.71-4.02 mg/g and 3.60-3.88 mg/g, respectively, while the weak acid cation exchange resin (WK60L) only has 1.251-0.52 mg/g. At first glance, this result seems contradictory to the trend of their standard exchange capacities indicated in Table 1. It was expected that WK60L would have higher ion exchange capacity because it has a standard exchange capacity of 4.4 eq/L, much higher than 2.2 eq/L of CMP28 and 2.0 eq/L of SCR-B. However, it was reported in our previous study that strong acid cation exchange resins produce more cations and thus have higher ion exchange capacities against weak acid cation exchange resins, even when the absolute values of standard ion exchange capacities of weak acid cation exchange resins are greater than the ones of strong acid cation exchange resins.9)
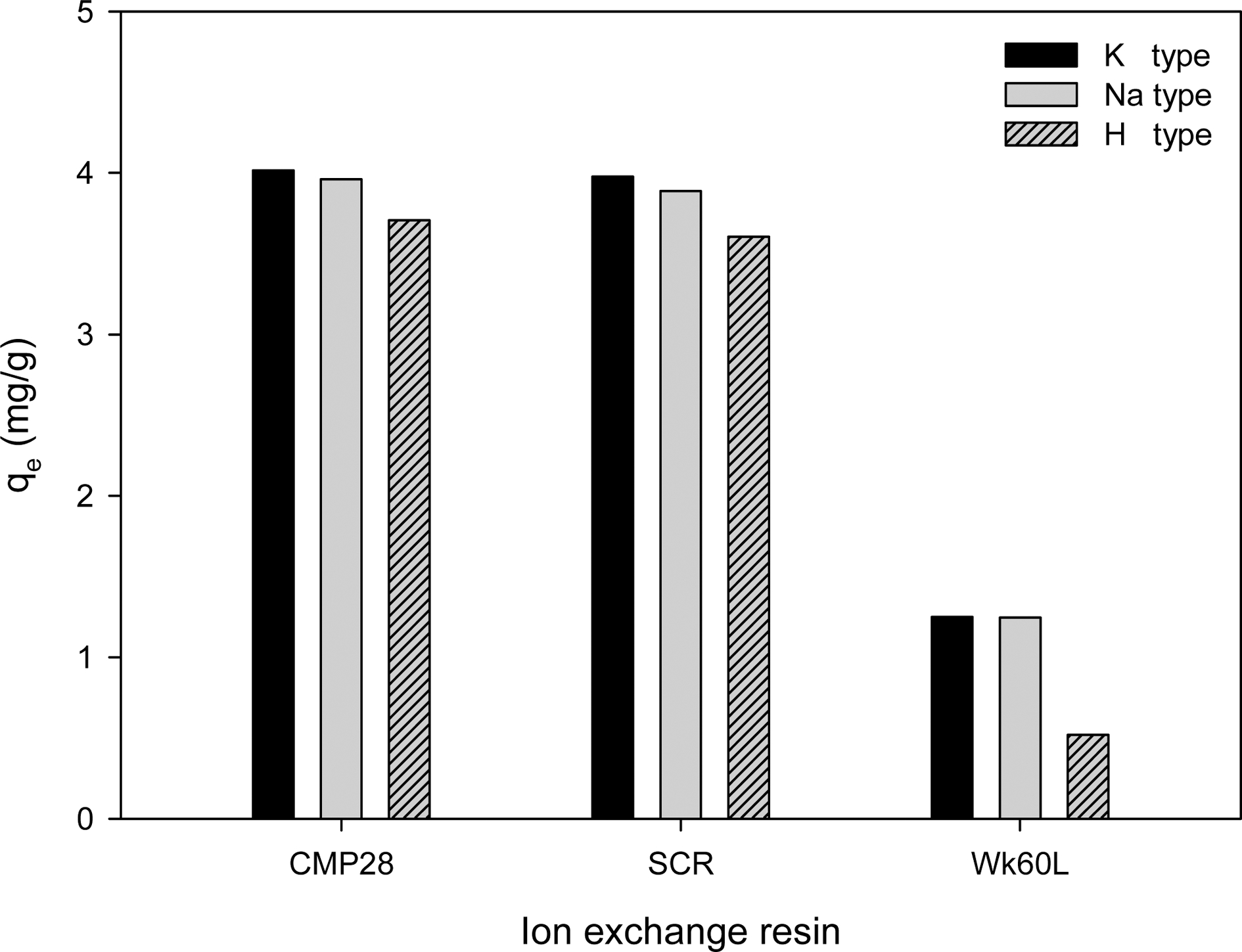
Fig. 1.
Ion exchange capacities of aqueous lithium recovery vs. various resins. The amount of resin, the initial lithium ion concentration, and the operating temperature were 0.2 g, 10 mg/L, and 20 °C, respectively.
Meanwhile, the comparison of CMP28 and SCR-B, which are the same strong acid cation exchange resins but have different pore structures, porous and gel types, respectively, demonstrates that CMP28 has a higher ion exchange capacity of 3.71-4.02 mg/g than 3.60-3.88 mg/g for SCR-B. It is probably due to the higher porosity of porous type ion exchange resins and also consistent with our previous study that porous type resins are more effective in the removal of ammonia nitrogen.10)
Ion exchange capacities according to the cation form have changed with the following order; K+ > Na+ > H+. In general, the cation exchange is affected by electronegativity under Coulomb’s law.11,12) The electron density inside the resins shifts from functional group (COO- or SO-) to cation by the interaction between them and the interaction becomes stronger in proportion to the electronegativity of cation.13) Considering that the electronegativities of K+, Na+, and H+ are 0.82, 0.93, and 2.1, respectively, it is reasonable that K+ form resins have the highest ion exchange capacity for aqueous lithium recovery and H+ form resins do the lowest value. Moreover, H+ form resins have additional hydrogen bonding inside.6) We also reported in their study on the lithium ion adsorption by using zeolites modified with K+, Na+, Ca2+, Mg2+, and Al3+ cations that K+ form zeolite had the highest adsorption capacity.14)
The following kinetic and equilibrium isotherm studies were performed with the K+ form and porous type strong acid cation exchange resin (CMP28-K) of the best performance.
3.2. Effect of Lithium Ion Concentration
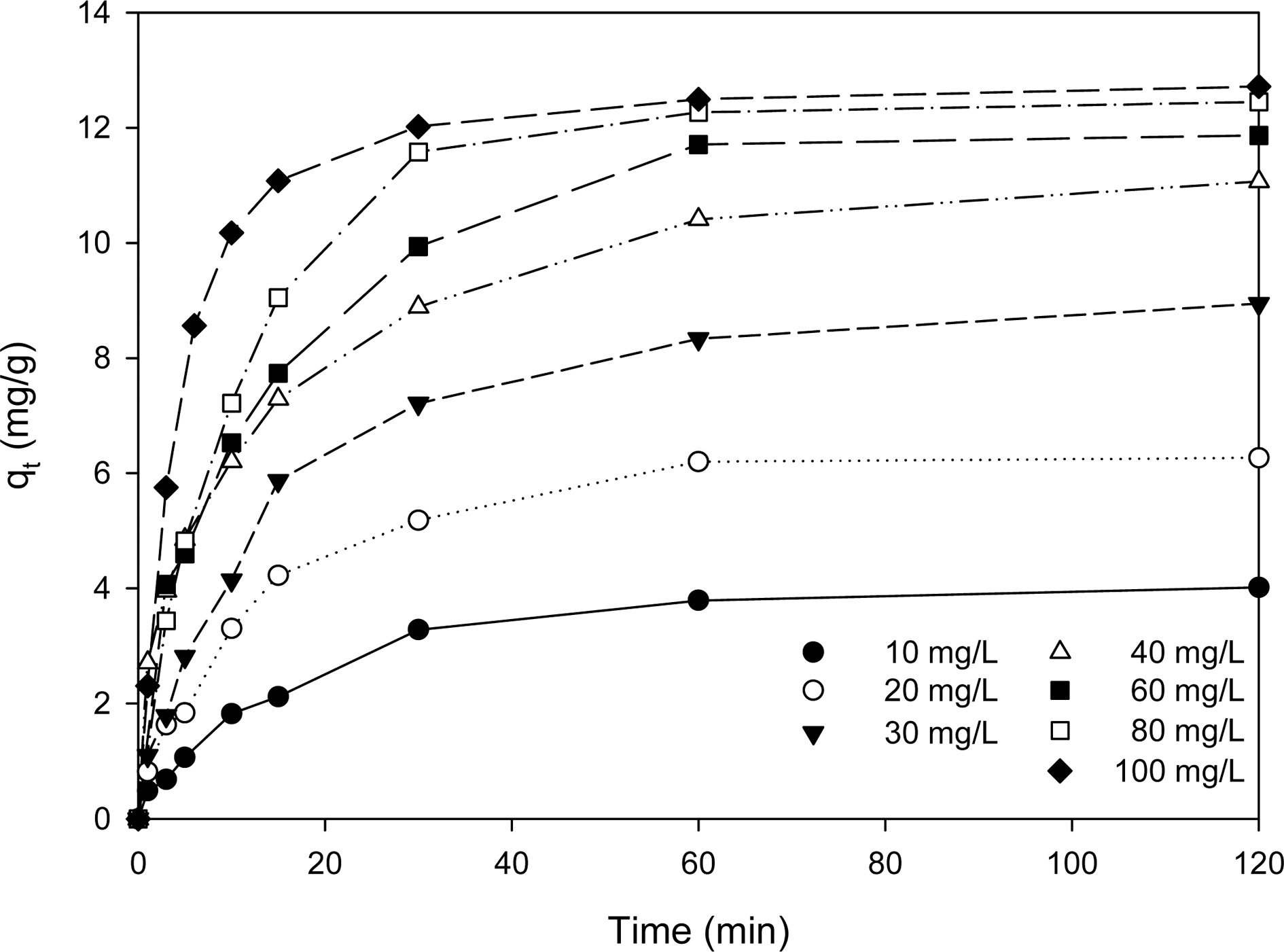
Fig. 2 shows the change of ion exchange capacities of aqueous lithium recovery by CMP28-K with time at different initial lithium ion concentrations between 10 and 100 mg/L. As shown in Fig. 2, aqueous lithium recovery by ion exchange is very fast in the first 15 min and reaches equilibrium after 40 min at all concentrations. It is explained by the drastic decrease of active sites for ion exchange.15)
3.3. Kinetics
Navarrete-Guijosa et al. proposed the following equation for the kinetics of ion exchange with lithium ion.6)
where ka(1/min) and kd(1/min) are the ion exchange rate constant between resin and lithium ion and the desorption rate constant of lithium ion, respectively. The θ and 1-θ are fractions of anionic sites on resin where lithium ions already occupies and where lithium ions may occupy by ion exchange, respectively.
In equilibrium, θ is defined as follows:
where C0, Ct, Ce are the initial lithium ion concentration, the lithium ion concentration at any time t, and the equilibrium lithium ion concentration, respectively.
By inserting Eqn. (3) into Eqn. (2) and integrating Eqn. (2), and then applying Eqn. (1), it reaches the following equation:
where qt(mg/g) and qe(mg/g) is the ion exchange capacities at any time t and equilibrium, respectively.
The pseudo-first-order rate equation is defined as follows:16)
where K1(1/min) is the pseudo-first-order rate constant.
The pseudo-second-order rate equation is defined as follows:16)
where K2(g/mg·min) is the rate constant of pseudo-secondorder rate constant.
To evaluate the ion exchange rate equations, Marquardt’s percent standard deviation (MPSD, Δqe) was used as defined below:
where N is the number of experimental data. The qt,exp (mg/g) and qt,cal(mg/g) are ion exchange capacities from experiments and calculations, respectively.
The kinetic parameters for aqueous lithium recovery by ion exchange with CMP28-K are summarized in Table 2. Those values were obtained by applying Eqns. (4-6) into the experimental data shown in Fig. 2. The pseudosecond- order rate equation shows the best fit with experimental data with Δqe = 2.98-17.68 %, while the rate equation proposed by Navarrete-Guijosa et al.6) and the pseudofirst- order rate equation have Δqe = 4.70-32.02 % and Δqe = 5.90-32.84 %, respectively. As shown in Table 2, the aqueous lithium recovery increased from 4.42 to 12.71 mg/g as the initial lithium ion concentration increased.
Table 2.
Kinetics parameters for aqueous lithium recovery by ion exchange with CMP28-K.
| C0 (mg/L) | qe,exp (mg/g) | Eq (4) | Pseudo-first-order, Eq. (5) | Pseudo-second-order, Eq. (6) | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| qe (mg/g) | ka (l/min) | Δqe(%) | qe (mg/g) | k1 (l/min) | Δqe(%) | qe (mg/g) | k2(g/mg•min) | Δqe(%) | ||
| 10 | 4.42 | 5.04 | 0.0758 | 17.61 | 4.32 | 0.0669 | 18.49 | 5.04 | 0.015 | 17.56 |
| 20 | 6.27 | 7.07 | 0.0904 | 13.51 | 6.15 | 0.0767 | 19.15 | 7.07 | 0.013 | 16.51 |
| 30 | 8.94 | 9.99 | 0.0816 | 13.08 | 8.60 | 0.0715 | 18.19 | 9.99 | 0.0082 | 12.17 |
| 40 | 11.07 | 11.25 | 0.1505 | 18.45 | 10.12 | 0.1110 | 26.72 | 11.25 | 0.0134 | 17.68 |
| 60 | 11.86 | 12.895 | 0.1135 | 8.67 | 11.93 | 0.0612 | 15.61 | 12.89 | 0.0088 | 7.64 |
| 80 | 12.44 | 13.94 | 0.1154 | 4.71 | 12.32 | 0.0929 | 5.89 | 13.94 | 0.0083 | 2.98 |
| 100 | 12.71 | 13.43 | 0.2714 | 9.59 | 12.25 | 0.1946 | 32.84 | 13.43 | 0.020 | 9.58 |
3.4. Equilibrium Isotherm
The relationship between ion exchange capacity and equilibrium concentration plays an important role in explaining the interaction between resin and solute, and optimizing the ion exchange process.17) Freundlich, Langmuir, and Dubinin-Radushkevich(D-R) isotherms were applied to experimental data for the aqueous lithium recovery by ion exchange with CMP28-K.
The Fruendlich isotherm is given by:
where KF(L/mg) is the Freundlich constant, Ce(mg/L) is the equilibrium ion exchange capacity, and 1/n is the adsorption strength constant.
The Langmuir isotherm is defined by:
where KL(L/mg) is the Langmuir constant, and qm(mg/g) is the maximum ion exchange capacity.
Finally, the Dubinin-Radushkevich(D-R) isotherm is as follows:
where Xm(mol/g) is the maximum ion exchange capacity, β(mol2/kJ2) is the ion exchange energy constant, and ε is the Polanyi potential which is calculated by:
where R(kJ/mol·K) is the ideal gas constant, and T(K) is the absolute temperature.
E(kJ/mol) is the average ion exchange energy and defined as follows:
Experimental ion exchange isotherms of the aqueous lithium recovery with CMP28-K and numerical fits of Freundlich, Langmuir, and D-R models are compared in Fig. 3. The experimental data in Fig. 3 were applied to Eqns. (8-10) and the parameters calculated by regression method are summarized in Table 3. As shown in Fig. 3 and Table 3, Langmuir isotherm gives the best fit with r2 = 0.9932. The maximum ion exchange capacity from Langmuir isotherm was 13.88 mg/g, which is greater by a factor of about 1.4 when compared to 10.5 mg/g of Na+ form and gel type cation exchange Amberlite IR 120 resin in the lithium ion adsorption study by Navarret- Guijosa et al.6) For references, this value(13.88 mg/g) is higher than 4.92 mg/g with activated carbon modified with nitric acid in the work of Kam et al.,18) but is lower than 27.21 mg/g with lithium manganese oxide synthesized by wet mixing in the work of You et al.19) and 16.9 mg/g with nanocrystal MnO2 synthesized by hydrothermal method in the work of Zhang et al.20)
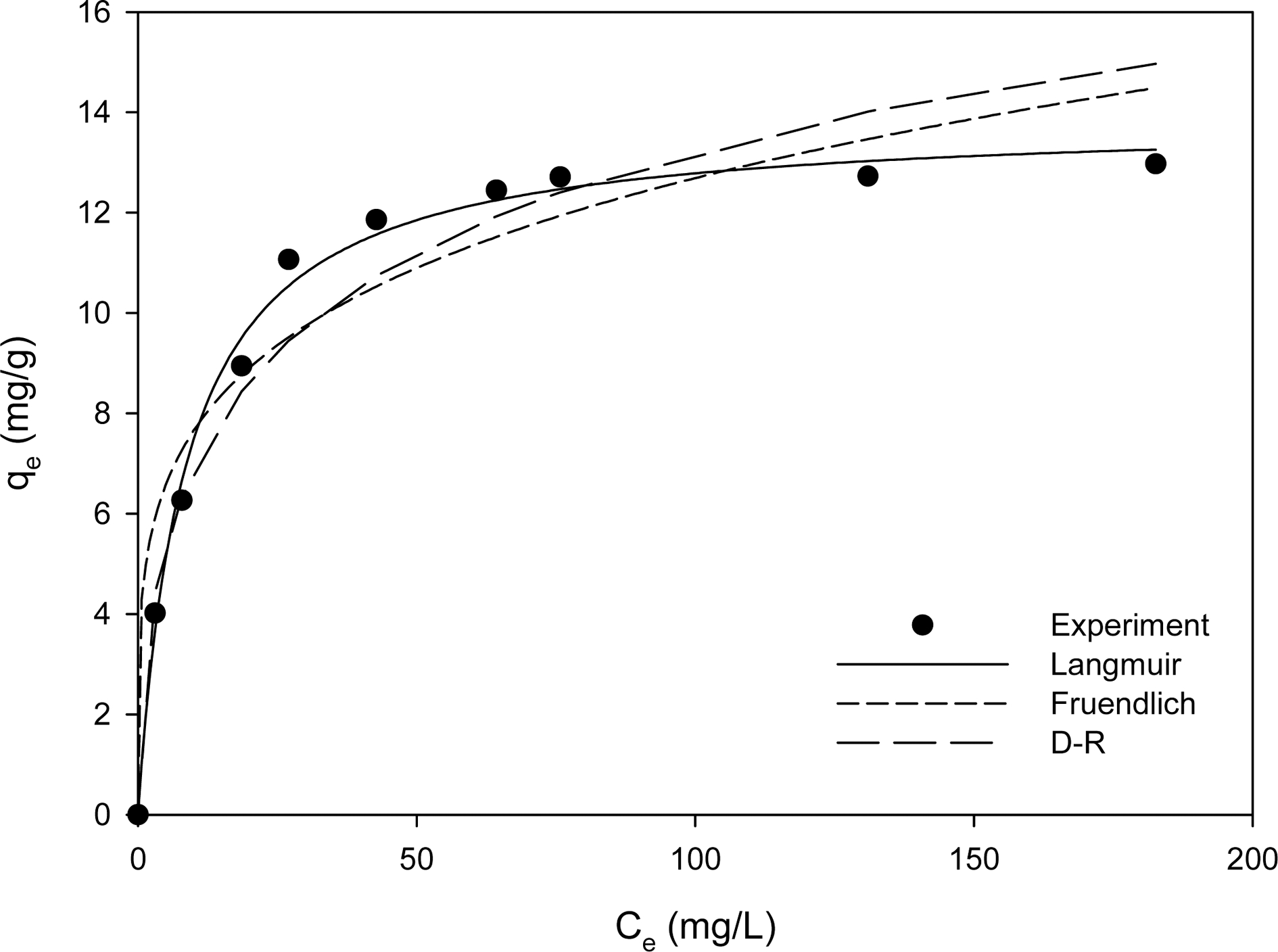
Fig. 3.
Ion exchange isotherm of aqueous lithium recovery with CMP28-K and numerical fits of Freundlich, Langmuir, and D-R models.
Table 3.
Parameters of equilibrium isotherm models for the aqueous lithium recovery with CMP28-K ion exchange resin.
| Freundlich | Langmuir | D-R | |||||||
|---|---|---|---|---|---|---|---|---|---|
| KF (L/mg) | n ((mg/g)(1/mg)1/n) | r2 | KL (L/mg) | qm (mg/g) | r2 | E (kJ/mol) | Xm (mol/g) | β (mol2/kJ2) | r2 |
| 4.60 | 4.54 | 0.9275 | 0.11368 | 13.88 | 0.9932 | 10.78 | 0.0031 | 0.0043 | 0.9404 |
Meanwhile, the calculated average ion exchange energy from D-R isotherm was 10.78 kJ/mol. Now that the value is placed between 8 and 16 kJ/mol, the aqueous lithium recovery with CMP28-K is confirmed by the mechanism of ion exchange.21)
3.5. Effect of pH
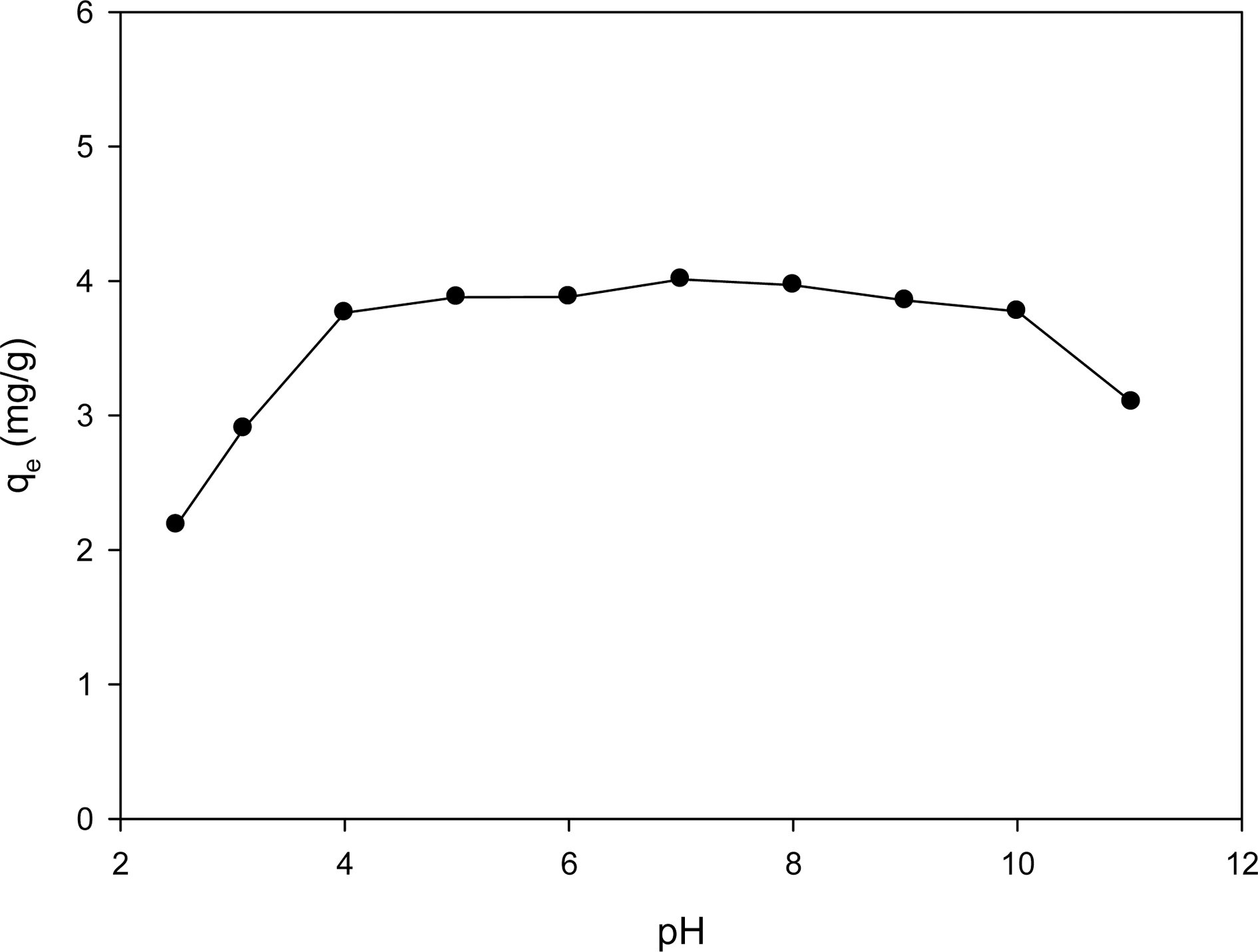
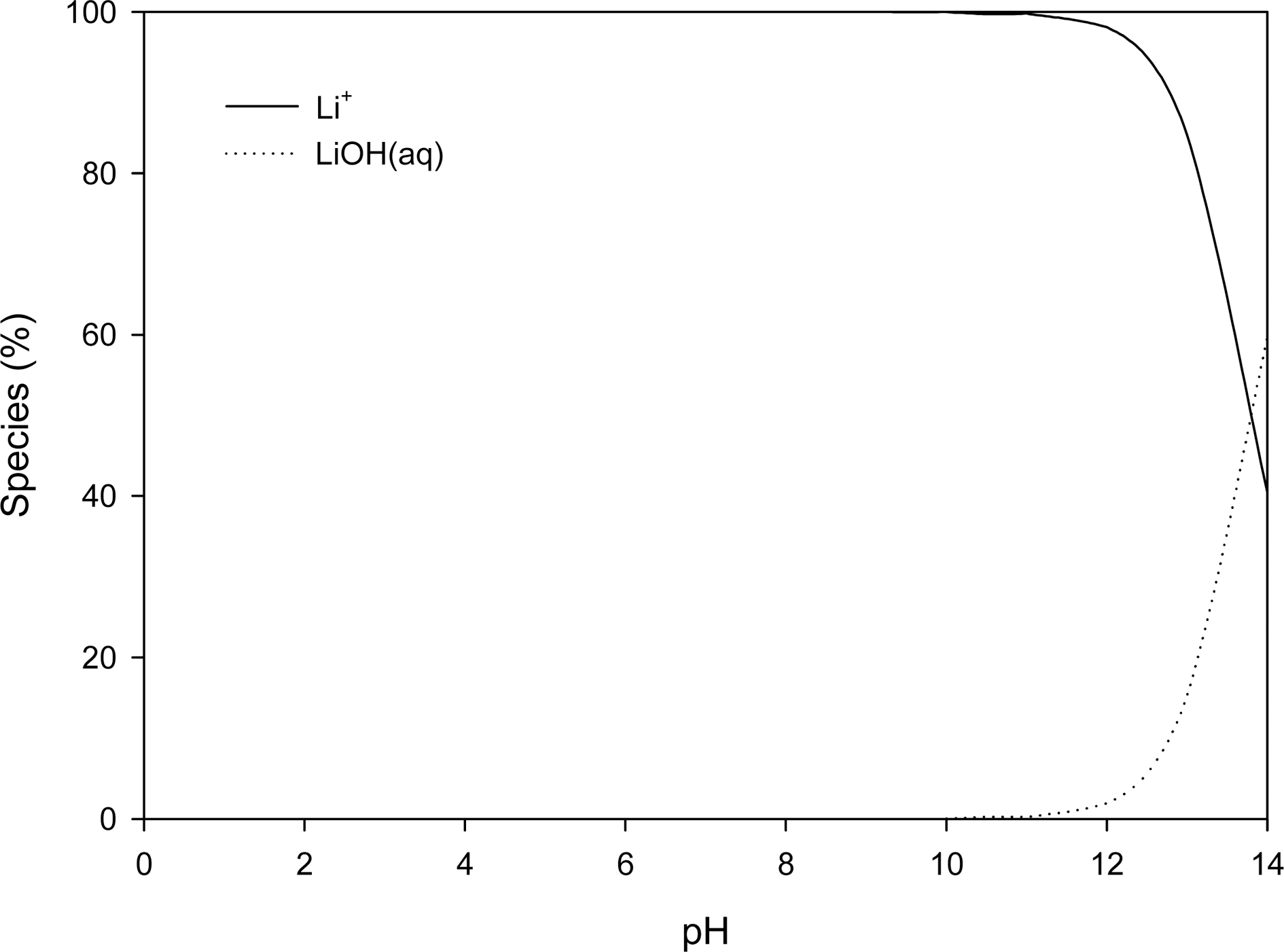
The effect of pH on the ion exchange capacity of aqueous lithium recovery by ion exchange with CMP28- K is illustrated in Fig. 4. The lithium ion concentration in aqueous solution was maintained constantly at 10 mg/L. As shown in Fig. 4, the ion exchange capacity is greatly decreased at below pH 4. It is attributed to the competition of lithium ion and hydrogen ion for the ion exchange with CMP28-K in this pH region. Meanwhile, the ion exchange capacity was also reduced at above pH 10. The distribution of lithium species by pH is simulated by Visual MINTEQ software and shown in Fig. 5. In high pH region (pH > 10), lithium is likely to exist as a form of hydroxide and thus lithium ions are not sufficient for ion exchange. In the region between pH 4 and pH 10, the ion exchange capacity was maintained at about 4 mg/g regardless of pH change.
4. Conclusion
In this study, it has been investigated how the ion exchange capacity of aqueous lithium recovery changes according to the characteristics of resins; functional group, cation form, porous structure. First, strong acid cation exchange resins were favored for the aqueous lithium recovery against weak acid cation exchange resins. Second, a porous type CMP28 resin was better than a gel type SCR-B. Thirdly, K+ form resins had the highest ion exchange capacity for aqueous lithium recovery among ion exchange resins with cation modified with K+, Na+, and H+.
Kinetic and equilibrium isotherm studies were carried out for the K+ form and porous type strong acid cation exchange resin (CMP28-K) showing the best performance. The pseudo-second-order rate equation and Langmuir isotherm showed the best fits, and the maximum ion exchange capacity of aqueous lithium recovery by CMP28- K was 13.88 mg/g. Meanwhile, the ion exchange capacity of CMP28-K was stably maintained in the pH region of 4-10.