1. Introduction
In recent years, the organometal halide perovskite materials have attracted considerable attention due to the tunability of the physical properties arising from their chemical diversity.1-6) AMX3 general formula is commonly used to present the family of these materials, where A is an organic ammonium cation(methyl ammonium, ethyl ammonium, etc.),1,2) M is a metal cation(Pb, Sn, etc.),3,4) X is a halogen atom(Cl, Br, and I).5,6) Owing to its vast variety of chemical composition, those materials exhibit a wide range of unique electronic and optical properties that are highly required in novel electronic and optical applications. The other powerful tool to tune the properties of these material is their size5-8) and shape diversity. For example, CH3NH3PbBr3 shows high photoluminescence efficiency and color tunability as the particle size goes to nano-scale range,6,7) thus enabling its application in light emitting devices,8-14) solar cells,7,15) sensors,16,17) piezoelectric devices,18-20) etc.
For lighting, photovoltaic, and piezoelectric devices with a planar structure, where perovskite particle - polymer composite is required as an active layer,8,12-15,21-23) a precise control of particle dispersion and their concentration in polymer matrix, and film uniformity are essential to prevent their aggregation and achieve a uniform coating.8,12,15) Typically, perovskite - polymer composites have been prepared by combining a polymer with the precursors of the desired perovskite material in a good solvent such as N,N-dimethylformamide(DMF) and spin-coating on a substrate. For example, Wang et al. demonstrated perovskite nanoparticle-based composite can be used as an active layer in light emitting diode, however, it was limited to very low particle concentration.22) Therefore, this method would result in micro-scale large aggregates when high density precursor solution is used to achieve dense particle coverage.8,14)
Here, we report on a simple method to fabricate a thin film of poly(methyl methacrylate) (PMMA)-CH3NH3PbBr3 perovskite nanoparticle composites. We showed that the presence of PMMA during the formation of perovskite NPs suppresses their aggregation, and thus results in a uniform film with higher luminescence, compared to the conventional perovskite composites.
2. Experimental Section
Lead (II) bromide (PbBr2, 99 %), n-octylamine (≥ 99 %), and oleic acid (≥ 90 %) were received from Aldrich. Methylamine water solution (CH3NH2, 33%), hydrobromic acid (HBr, 48 %), toluene (99 %), and N,N-dimethylformamide (99.5 %) were received from Daechung Chemicals & Metals. CH3NH3Br was synthesized by the previously reported method.6,24,25) The reaction was carried out in an ice bath (0 °C, 2 h) to effectively remove the heat generated during the reaction and to prevent reagent evaporation. The water solvent was evaporated by a rotary evaporator at 40~45 °C. Finally, the obtained precipitate was washed with diethyl ether for three times and dried under vacuum (60 °C, 6 h) condition. The precursor solution for CH3- NH3PbBr3 was prepared by dissolving CH3NH3Br (0.16 mmol) and PbBr2 (0.2 mmol) dissolved in 5 mL of DMF that contains 20 μL of n-octylamine and 0.5 mL of oleic acid as a surfactant. PMMA-CH3NH3PbBr3 nanocomposite solution was fabricated by the precipitation method in the presence of PMMA (MW = 120,000 g/mol).6) Precursor solution (2 mL) was dropwise added into the PMMA (200 mg) in toluene (10 mL) under vigorous stirring. For the reference sample, the NPs were synthesized in toluene, and PMMA (200 mg) was subsequently added. PMMAperovskite composite was obtained by centrifugation at 7000 rpm for 30 min and dispersing in toluene (0.5 mL). Thin film is fabricated by spin-coating on an ozonetreated slide glass. The phase formation is measured by X-ray diffraction using a Bruker AXS D8 diffractometer with Cu-Kα radiation at λ = 1.54 Å. The scanning electron microscope(SEM) and the transmission electron microscope( TEM) was performed on a Hitachi S4800 and JEM 2100, respectively. UV-vis absorption spectra(OA) were obtained using Shimadzu UV-2600 spectrophotometer. Photoluminescence( PL) spectra were recorded using Hitachi F-7000 fluorescence spectrometer at room temperature.
3. Results and Discussion
Fig. 2a shows the typical appearance of as-prepared PMMA-CH3NH3PbBr3 perovskite nanocomposite solution under daylight that emits bright blue-green light under ultra violet(UV) illumination (365 nm). It should be noted that the reference sample shows the identical appearance. XRD pattern(Fig. 2b) confirms a formation of CH3NH3- PbBr3 nanocrystals belonging to a space group of Pm-3m with a lattice spacing, a = 5.9896 Å. Electron micrographs show that the solution of the CH3NH3PbBr3 perovskite formed in the presence of PMMA consists of ~5 nm perovskite NPs(Fig. 2(c-e)).
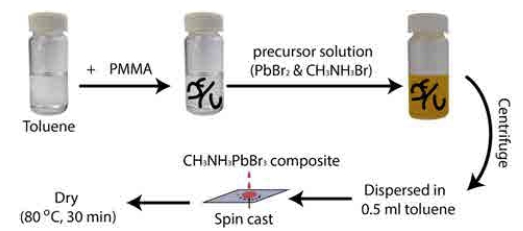
Fig. 1
Schematic illustration of the experimental flow to prepare PMMA-CH3NH3PbBr3 nanoparticle composites.
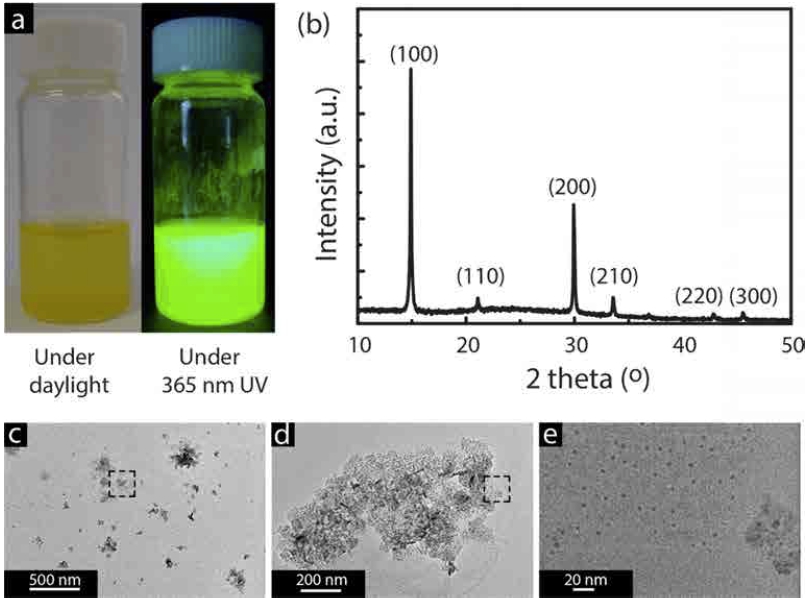
Fig. 2
(a) As-prepared PMMA-CH3NH3PbBr3 perovskite nanocomposite solution under day light and under UV illumination (365 nm) and (b) corresponding XRD pattern. (c-e) Low and high magnification of TEM images of the PMMA-CH3NH3PbBr3 perovskite nanocomposites prepared by drop-casting at low concentration where the squares denote the magnified area.
Fig. 3 shows a comparison of UV-vis absorbance and photoluminescence emission spectra of PMMA - CH3NH3- PbBr3 nanocomposites. The UV-vis absorption spectra of all samples exhibit a band edge at 492 nm (~2.52 eV). The band edge value is blue-shifted by ~55 nm (250 meV) from that of the bulk CH3NH3PbBr326-27) due to quantum confinement effect.6,24,25) In the solution, PL emission spectra are peaked at 496 nm (2.54 eV), and the full width at half maximum(FWHM) value of the composite and the reference is 22 nm, respectively. PL emission is also blue-shifted by ~40 nm compared to the bulk CH3NH3PbBr3.26-27) Small Stokes shift (~10 meV) implies that the PL emission originates from excitonic recombination. 27) In the film, the PL emission of the PMMA - CH3NH3PbBr3 is red-shifted by 10~15 nm compared to that of the solution while the band edge on absorbance spectra does not show a significant change. The red-shift of PL emission for film could be originated from the reabsorption of the shorter wavelength PL emission between CH3NH3PbBr3 particles in the proximity of the CH3NH3PbBr3 nanoparticles is dominant in the film case. In contrast, the absorption edge of film remained the same suggesting that there is no particle growth and thus the particles size remains the same. In addition, we calculated the ratio(IPL/OA) of the integral PL emission (underline area of the PL emission spectra, IPL) to optical absorbance(OA) at 365 nm. This parameter relatively characterizes the power conversion efficiency. In the solution, the IPL/OAreference is 1.4 times higher than IPL/OAcomposite, whereas the IPL/ OAcomposite is 1.8 times higher than IPL/ OAreference in the film.
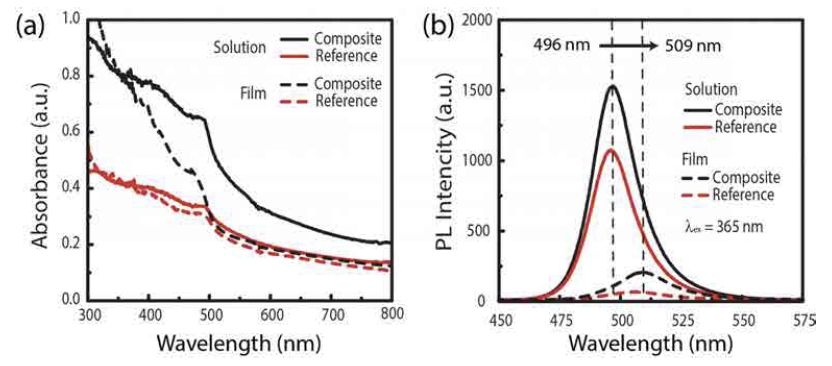
Fig. 3
(a) UV-vis absorption and (b) PL emission spectra of the PMMA-CH3NH3PbBr3 perovskite nanocomposite and reference. The solid and dashed lines denote PMMA-CH3NH3PbBr3 perovskite nanocomposite solution and film, respectively. PL was recorded with an excitation source (365 nm).
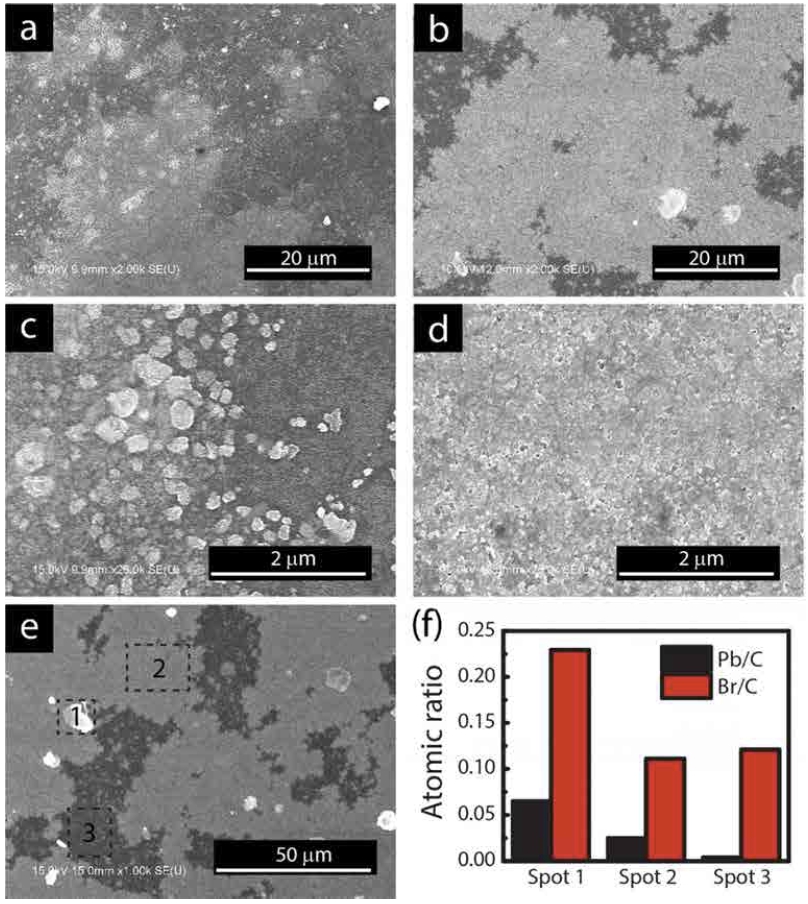
To understand the origin of enhanced PL performance in the film, we further examined the surface morphology of the film for the reference and the composite(Fig. 4). The composite sample shows larger coverage compared to the reference sample where micro-scale aggregates are observed in both cases(Fig. 4(a-b)). Furthermore, higher magnification images(Fig. 4(c-d)) exhibit large amounts of such aggregates in the reference sample compared to the composite sample where the individual CH3NH3Pb- Br3 perovskite particles form a uniform film. We performed energy disperse x-ray spectroscopy(EDS) to reveal the compositional variation in Fig. 4e. In spot 1, a large- size particle shows about 1:3 ratio of Pb to Br contents, confirming the atomic ratio of CH3NH3PbBr3 perovskite materials. Similarly, the uniform film region in spot 2 also shows the same atomic ratio, indicating the presence of CH3NH3PbBr3 perovskite nanoparticles in PMMA. In contrast, negligible amounts of Pb is observed in the uncovered area (spot 3), indicating the absence of CH3- NH3PbBr3 perovskite nanoparticles. Br/C ratio in spot 3 results from the presence of PMMA along with methylammonium bromide and octylammonium bromide. Synthesis of perovskite particles in the presence of PMMA is shown to suppress their aggregation into coarse clusters, and thus form a uniform film with homogeneous particle distribution, smooth coverage, and enhanced PL performance compared to the reference.
4. Conclusions
To summarize, we report PMMA-CH3NH3PbBr3 perovskite nanoparticle-based composite film. We introduced PMMA during perovskite particle synthesis process that prevents the particle aggregation, thus enables uniform film formation. PMMA-CH3NH3PbBr3 perovskite nanocomposite showed improved PL performance and higher PL emission to absorption ratio over a PMMA-CH3NH3- PbBr3 perovskite mixture. This approach opens doors toward to CH3NH3PbBr3 perovskite NP-polymer composite with controlled NP concentration.