1. Introduction
Thermoelectric devices can convert electricity into thermal energy and vice versa with the variation of temperature.1) The efficiency of thermoelectric materials is determined by the dimensionless figure of merit ZT. The ZT value can be calculated by following equation.
Here, S is the Seebeck coefficient, σ is the electrical conductivity, κ is the thermal conductivity and T is the temperature. As shown in equation (1), high ZT value requires high power factor (PF), S2σ, and l ow t hermal conductivity (κ). However, the relationship among these thermoelectric properties are quite complex. The electrical conductivity is directly proportional to the thermal conductivity according to Widemann-Franz law (κE = σLT, where κE is electronic thermal conductivity and L is Lorenz number).2) Moreover, as the carrier concentration increases, Seebeck coefficient reduces while electrical conductivity increases. Therefore, an optimum value of S, σ and κ are required to get high dimensionless figure of merit (ZT).
The complex relationship among the Seebeck coefficient, electrical conductivity and thermal conductivity makes difficulty to get high ZT value.3) However, zintl compounds are one of appropriate thermoelectric material to minimize this complexity.4) Typically, zintl compounds have two ionic sites. One is electropositive cation and other is electronegative anion. The electropositive cation can donate electron to the electronegative anion which makes the compound ionic in nature. Moreover, if the number of electrons of cation is not sufficient to donate anions, the valance shell of anion will remain incomplete. Therefore, anion will make covalent bond with other anion. The existence of both ionic and covalent bond makes the structure of zintl compound more complex and lead to a characteristics electron-crystal phonon-glass properties.4) Zintl anions formed from heavier, softer atoms will have lower lattice thermal conductivity by definition. Furthermore, these heavy atoms produce smaller band gap semiconductors, which are more amenable to the high dopant concentrations necessary for thermoelectrics.5)
Zintl compound Mg3Sb2 shows two structures, one is cubic bixbytie and other is hexagonal.6) The cubic α- Mg3Sb2 is a high temperature phase consisting of 48 Mg and 32 Sb atoms which is similar to structure of α-La2O3. On the other hand β-Mg3Sb2 is a low temperature phase which is analogous to Mn2O3 and it contains two nonequivalent Mg site.7) The ionic Mg2+ cation has the ability to donate electron to the [Mg2Sb2]2- frame-work which is a characteristics behavior of a zintl compound.8)
The single phase Mg3Sb2 showed p-type properties, but the efficient thermoelectric properties were displayed by n-type Mg3Sb2.6,8-13) However, several attempts were taken to improve the thermoelectric performance of ptype Mg3Sb2 but still it remains below the unity. By enhancing the carrier concentration and carrier mobility, Song et al showed the improved p-type properties in the alloy system in which Mg was doped by Ag. This alloys were synthesized by a spark plasma sintering (SPS) and showed the highest ZT value of 0.51.14) Furthermore, codoping of Na and Zn was showed to increase thermoelectric performance with a peak ZT value of 0.8.15) By doping Li in Mg sites fabricated by ball milling followed by SPS, a high thermoelectric performance with a peak ZT value of 0.59 was exhibited where electrical conductivity increased significantly.16) Moreover, enhanced p-type thermoelectric properties was achieved with a peak ZT value of 0.94 by doping Bi in Sb sites where ball milling followed by SPS synthetic procedure was applied.17) In this study, Sn doping on Sb sites in p-type Mg3Sb2 was considered to enhance thermoelectric performance with very low thermal conductivity. Previously, Sn doped Mg3Sb2 which was synthesized by induction melting, ball milling followed by spark plasma sintering, showed an optimal thermoelectric performance of p-type Mg3Sb2.18) In this study, alloying with variable Sn composition (0 ≤ x ≤ 0.008) in the form of Mg3Sb2 was synthesized by a controlled melting method where elemental shots were taken followed by pulverizing and vacuum hot pressing (VHP). Herein, in order to produce single phase Mg3Sb2, excessive amount of Mg was considered to add in the initial powder mixture, because the composition of the nominally designed Mg3Sb2 usually resulted in excessive Sb contents after hot consolidation process, due to evaporation of Mg, as reported in other studies.10-11) In this study, thermoelectric properties of Mg3Sb2, in terms of Seebeck coefficient, electrical conductivity and thermal conductivity, as a function of Sn contents was investigated and compared with those of pristine sample. In order to elucidate carrier transport mechanism in the thermoelectric properties for this alloy system, electronic flow properties were also evaluated and discussed.
2. Materials and Methods
All of sample with nominal composition Mg3.8Sb2-xSnx were synthesized by using the elemental shots of Mg (99.9 % purity, Kojundo), Sb (99.999 % purity, Kojundo) and Sn (99.9 % purity, Kojundo) under an inert atmosphere (Ar gas). The measured sample was then put into a graphite crucible with stopper which inner walls were coated by a thick layer of boron nitride (BN) for controlled melting. After that the crucible was kept into the vacuum furnace. Sample then heated at 1,173 K for 1 hour. After the completion of heating process, a mortar pestle was used to pulverize the cooled ingot. A 325 mesh sieve was also used to sieve the powders. The whole pulverizing to sieving process was maintained under Ar gas atmosphere. Vacuum hot press (VHP) was carried out for the consolidation of sample at 70 MPa and at 873 K for 4 hours. To characterize the phases, X-ray diffract meter (XRD, BRUKER AXS Advance D-8, Germany) with Cu-Kα radiation source was taken. Surface morphology was investigated by scanning electron microscopy (SEM, Quanta-400, Netherland). A tube shape sample (3 × 3 × 10 mm3) was used to calculate the Seebeck coefficient and electrical conductivity by using ZEM-3 (ULVACRIKO, Japan). Thermal diffusivity (d) was obtained by laser flash method of TC-9000H (ULVAC-RIKO, Japan). Density (r) was measured according to Archimedes principle. Thermal conductivity (k) was evaluated by following an equation, k = d CP × r. Here, CP is specific heat capacity of Mg3Sb2 and it was taken from literature.19) Transport properties were determined by Van der Pauw method (modified keithley-7065, USA) at a constant magnetic field (1T) and constant electric current (50 mA).
3. Results and Discussion
XRD patterns of pulverized powders are displayed in Fig 1(a). Single phase Mg3Sb2 was achieved by adding excess amount of Mg with a nominal formula Mg3.8Sb2. In Fig. 1(a), it was seen that small amount of the high temperature phase α-Mg3Sb2 and Mg2Sn were formed during the process for x = 0.006 and x = 0.008, respectively. The secondary phase Mg2Sn is known to be thermodynamically stable and typically shows n-type conduction properties.20) Thus, it may hamper the transport properties in a p-type Mg3Sb2.
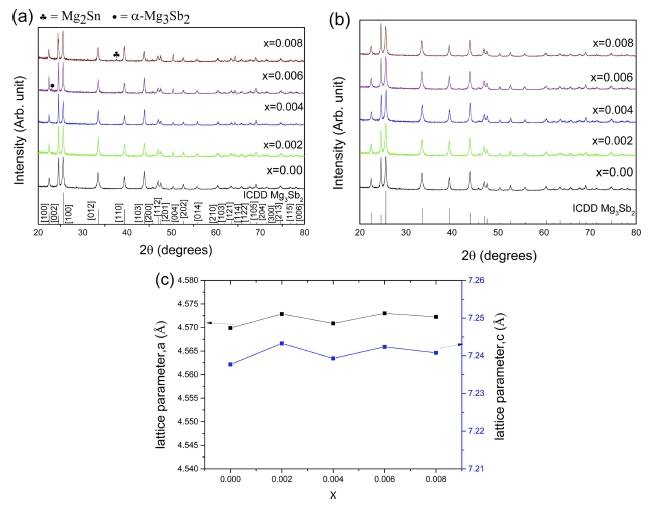
Fig 1
XRD diffraction patterns of Mg3.8Sb2-xSnx (0 ≤ x ≤ 0.008) for (a) powder samples, (b) VHPed samples, and (c) lattice parameters with respect to Sn content (x).
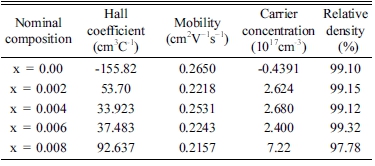
Fig. 1(b) shows XRD patterns of VHPed samples. No secondary peaks are observed for all samples, implying that alloying might proceed further during the hot consolidation process.21) The lattice parameter of VHPed Mg3.8Sb2-xSnx were plotted against the doping amounts and shown in Fig. 1(c). The addition of Sn showed a negligible effect on the lattice constant in this case, possibly due to similar size of Sb and Sn each other. The theoretical density of Mg3Sb2 was taken from literature22) and all VHPed samples showed relatively greater than 96% of theoretical density as shown in Table 1.
Table 1
Transport properties and relative density of vacuum hot pressed Mg3.8Sb2-xSnx (0≤x≤0.008) samples at room temperature.
 |
SEM observation was carried out in order to investigate the microstructural development and homogeneities in the microstructure. Fig. 2(a) shows a microstructure of pulverized powder sample for x = 0.006. The particles were typically angular shape under 45 μm in diameter since samples were sieved under 325 meshes after pulverization. The surface morphology of a fractured sample for VHPed x = 0.006 is shown in Fig. 2(b). The fractured sample shows quite dense microstructure with no obvious cracks. The typical lamellar structure can be identified in the fractured surface as in the similar crystallites.18) Energy dispersive X-ray spectroscopy (EDS) elemental mapping with a line profile of Sn for the VHPed sample of x = 0.006 are shown in Fig. 3. Mg, Sb, and Sn are homogenously distributed throughout the spotting area without any other impurities. From EDS line profile, it is also shown that Sn is homogenously distributed within the matrix, indicating that doping was performed adequately.

Fig. 2
Scanning electron microscopy for x = 0.006 sample after (a) as-sieved, and (b) VHP (fractured).
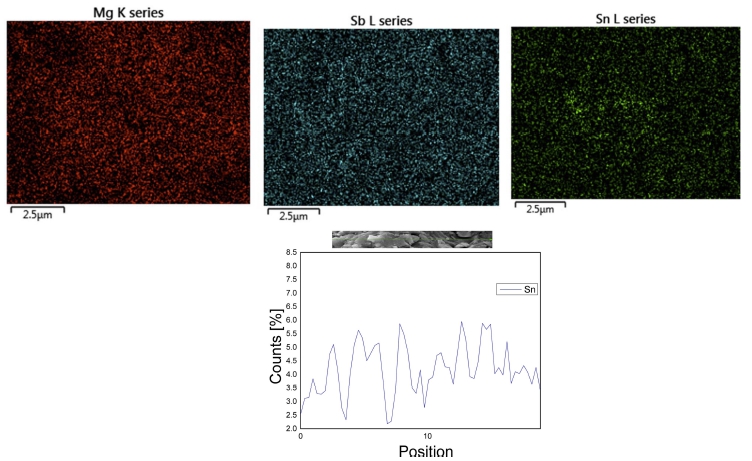
Fig. 3
EDS mapping for x = 0.006 shows elemental distribution of Mg, Sb and Sn and EDX line mapping shows that of Sn for x = 0.006.
As a function of temperature, the electrical conductivity (σ), and Seebeck coefficient (S), are displayed in Fig. 4(a) and 4(b) respectively. The temperature dependence of electrical conductivity is seen to increase with temperature. It is observed that all compositions show very high resistivity at room temperature. These semiconductors show an increase of electrical conductivity above 473 K. This is believed that the majority carrier (hole) gains sufficient energy to cross the band gap region at 473 K. Except x= 0.008, all of samples showed higher electrical conductivity than that of un-doped one. This is possibly due to the increase of carrier concentration in Sn doped samples. The electrical conductivity for x = 0.008 is found to be lower than that of pristine one, which could be due to the formation of n-type Mg2Sn during the process. With respect to magnitude, the highest electrical conductivity was found for two compositions with nominal formula Mg3.8Sb1.996Sn0.004 and Mg3.8Sb1.994Sn0.006 at 873 K.
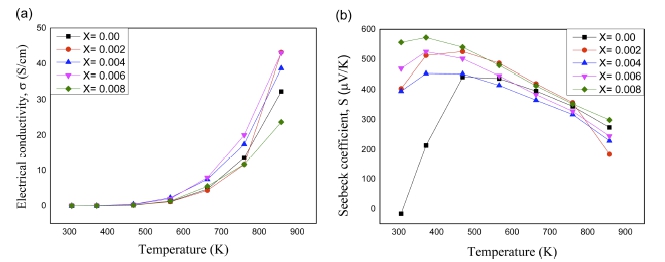
Fig. 4
Temperature dependence (a) electrical conductivity, (b) Seebeck coefficient of Mg3.8Sb2-xSnx (0 ≤ x ≤ 0 . 008).
In Fig. 4(b), it is seen that the Seebeck coefficient of all Mg3Sb2 with variable Sn composition is positive, implying p-type conduction. A low negative Seebeck coefficient was observed for un-doped Mg3.8Sb2 specimen at room temperature which is already reported in previous study, and then turn to p-type afterwards.23) This might be due to the presence of excess Mg. The excess Mg within the Mg3Sb2 cluster moves the position of Fermi energy level close to the conduction band and showed ntype property which was explained by a theoretical study.8) This small amount of excessive Mg in Mg3.8Sb2 sample might not be resolved in a conventional XRD analysis. Similarly, conventional XRD did not resolve excess Zn in Zn-Sb system due to the resolution limit, but it was observed in a transmission electron microscopy analysis.24) With respect to temperature, the Seebeck coefficients of all Sn doped samples increased up to 350 K, and then decreased afterward. Except for x = 0.008, the Seebeck coefficient increases with increasing Sn content (x) at 873 K. For example, x = 0.006 has a higher Seebeck coefficient than x = 0.004 and 0.002 at 873 K. Seebeck coefficient variation behavior along with electrical conductivity in the test temperature range seemed to fit well each other as expected.
The Hall coefficient, mobility and carrier concentration at room temperature are shown in Table 1. The carrier concentration is inversely proportional to the Seebeck coefficient and directly proportional to the electrical conductivity. Due to the higher carrier concentration of VHPed sample x = 0.004, it shows the lowest Seebeck coefficient at room temperature.
With the variation of temperature, total thermal conductivity and lattice thermal conductivity (κL) are provided in Fig 5(a) and 5(b), respectively. The total thermal conductivity is seen to decrease with increasing temperature up to 773 K and slightly to increase afterwards for all compositions. This behavior can be explained in terms of lattice thermal conductivity (κL) and electronic thermal conductivity (κE). It is known that total thermal conductivity is the summation of κL and κE and according to the Wiedemann-Franz law, electronic thermal conductivity can be calculated via κE = LσT (the Lorenz number L = 2.38 × 10-8 WΩK-2). From Figs. 5(a) and 5(b), it is clearly observed that the difference between total thermal conductivity and lattice thermal conductivity is very low, which means the main donor of total thermal conductivity is κL. Lattice thermal conductivity is also seen to decrease with increasing temperature for all composition as in Fig. 5(b). This behavior can be explained by an acoustic phonon scattering which can reduce the phonon mean free path at high temperature.25) The differences in atomic mass, size and lattice force can deform the crystal structure and then make to increase the phonon scattering which is responsible to decrease the lattice thermal conductivity. It is also observed in Fig. 5(a) that total thermal conductivity is slightly increased at 873 K for all composition. This is may be due to increase in electrical conductivity at that temperature. For all Sn doped specimens, it is seen that the value of total thermal conductivity is much lower than that of pristine one. This reflects that doping is performed effectively as anticipated. Specimen x = 0.006 shows the lowest thermal conductivity at 873 K.
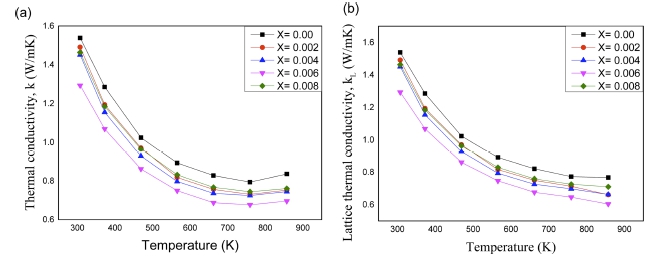
Fig 5
Temperature dependence of (a) thermal conductivity and (b) lattice thermal conductivity of VHPed Mg3.8Sb2-xSnx sample.
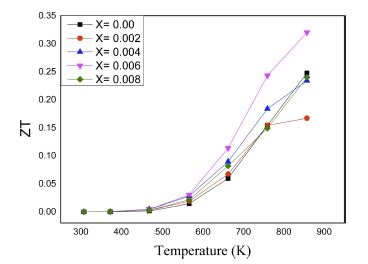
The temperature dependence of the dimensionless figure of merit (ZT) is evaluated in Fig. 6. The ZT value for all composition is seen to increase with temperature in this temperature range. Due to the best combination of electronic transport properties and lowest thermal conductivity, specimen x = 0.006 showed the maximum ZT value with about 0.32 at 873 K. This value is of 77 % improvement compared with intrinsic one.
4. Conclusions
Sn content p-type Mg3Sb2 have been successfully synthesized by controlled melting, pulverizing followed by VHP. Thermoelectric properties in this study are seen to be improved by chemical doping with Sn in Sb sites. The anionic substitution induced by doping in Mg3.8Sb2-xSnx showed a reduction in total thermal conductivity, and it showed the optimized value of Seebeck coefficient as well. Sample with x = 0.006 shows the lowest thermal conductivity which is about 83 % lower than that of intrinsic Mg3Sb2. The maximum ZT was obtained to be about 0.32 in the specimen Mg3.8Sb1.994Sn0.006 at 873 K, showing comprehensive improvement compared to the pristine one. In spite of low thermal conductivity was found in this study, the acquired electrical conductivity seems to be quite low due to the lower value of carrier concentration. Thus, it is suggested that selective doping might increase carrier concentration more, possibly enhancing ZT value further in p-type Mg3Sb2.