1 Introduction
High purity silica (SiO2) is an essential raw material for silicon and silicon compounds used in diverse applications such as EMC, LCD, and optical glass, etc.1-4) Main impurities of the silica minerals are iron and aluminum with a form of oxide and trace impurities such as Ca, Mg, K, Na, Ti are distributed with forms of complex oxides.5,6) Purification process of the silica by removal of the impurities is composed of physical and chemical treatment by utilizing the differences in physical/ chemical properties of minerals, which means the importance of its mineralogical characteristics in order to enhance purity grade of the silica. Generally, crushing/ grinding, gravity and magnetic separation, and chemical washing are sequentially used for purification and upgrade of the silica minerals.7-9)
In this study, aiming to develop a purification process from natural silica ore to the least 4N grade high purity silica powder, the mineralogical characteristics of a natural silica ore, such as distributions and forms of existing impurities, was analyzed. And its effect on the purification process with acid washing was discussed with respect to particle size of as milled silica ore.
2 Experimental procedure
2.1 Raw materials
A natural silica mineral used in this study was Chinese ore. Its chemical composition is summarized in Table 1.
2.2 Sample preparation
For mineralogical and chemical analysis of the silica ore, its powder and thin-plate were prepared. In order to investigate relationship between impurity liberation and particle size of the mineral, the natural silica ore was pulverized by crushing(jaw crusher) and milling(attrition mill). With 50~325 mesh screening, the powder was classified by 4 size groups.
For purifying relatively high quality silica having a purity of at least 99 %, fine milling and washing of whole ore is inefficient process. The information about mineralogical and chemical characteristics of impurity is helpful to choose processing parameters in the purifi-cation process such as degree of milling, washing solution. The silica ore was sliced and polished to prepare a mirror surface. Possibly existing sites(grain boundary, crack, or fracture surface) of the impurity in the ore should be analyzed by optical and electron microscope and its phase and distribution were confirmed by XRD and EDS.
2.3 Acid Washing
The removal behaviors of impurities were investigated according to different kinds of acidic washing solution.
① Acid washing after dry milling
Effect of the acid solution on washing of pulverized silica ore was investigated using nitric acid (HNO3), hydrochloric acid (HCl), sulfuric acid (H2SO4), acidic mixture (H2SO4 + H2O2). Content of impurity in the solution was measured by ICP-AES after washing and filtering.
2.4 Analysis
The particle morphology and impurities distribution of pulverized silica ore were analyzed using a Field Emission Scanning Electron Microscope(FE-SEM, JEOL JSM-6500F) with an energy dispersive spectrometer (EDS). The elemental composition and its content were measured by ICP-AES(Inductively Coupled Plasma Atomic Emission Spectroscopy, ICAP6500DVO) and the crystal phase was determined by X-Ray Diffraction(XRD, D/ MAX-2500) measurement.
3 Results and Discussion
3.1 Effect of particle size on impurity liberation
Generally, purification method is composed of physical process such as size reduction, magnetic and gravity separation and chemical purification like acidic washing. To completely separate the impurities from the silica, liberation of impurity is required by appropriate size reduction.
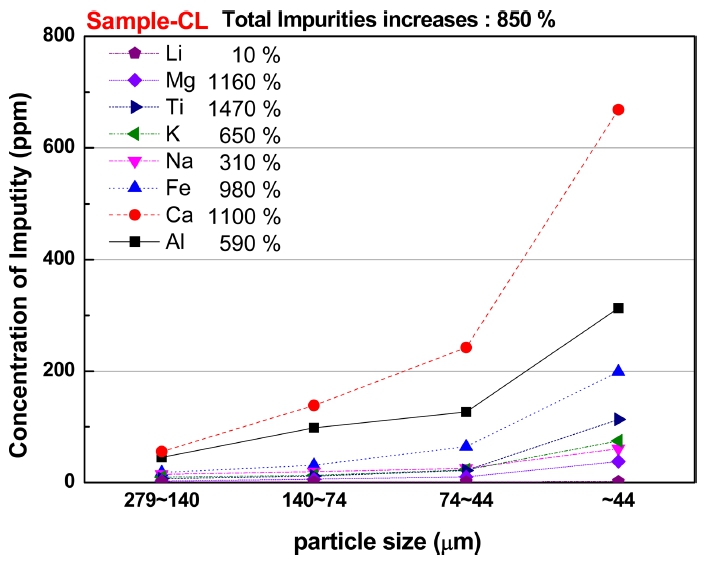
The silica ore was pulverized by autogenous milling to eliminate the contamination from milling process. The milled silica ore powder was classified by 50~325 mesh (aperture size 279~44 μm) screening and thereafter chemically analyzed by the ICP-AES. Fig. 1 shows the content of impurities according to the as milled particle size.
The smaller particle size showed the more content of impurities, which implies that the more impurities were liberated with size reduction by pulverization process. In addition, the fraction of smaller particle size appeared to contain the more impurity content, which means that the relatively smaller impurity particles liberated from grain boundaries and within grains were passed through the aperture of lower mesh sieves and were accumulated to the fraction of smaller particle size.
3.2 Distribution and phase of impurity
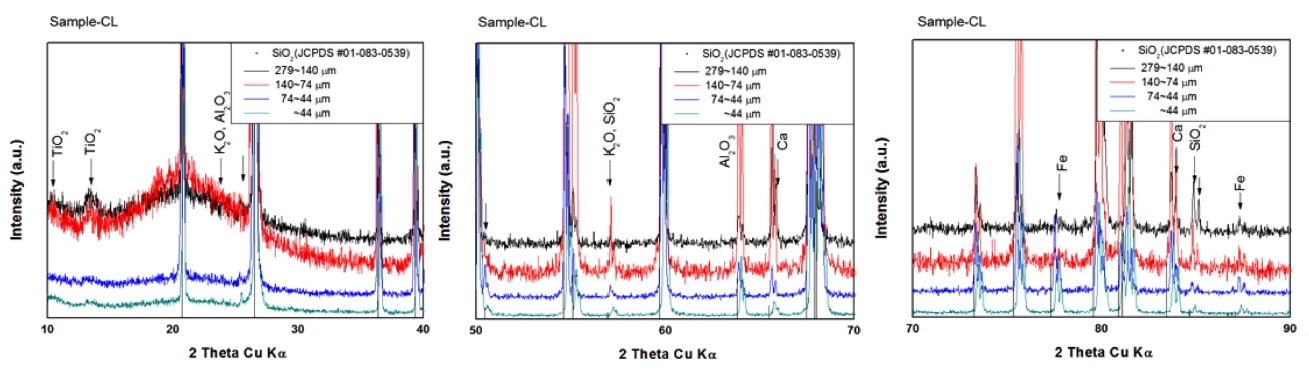
Grain boundaries and cracks are expected to have the higher content of the impurities, therefore the chemical composition of crack plane was analyzed and shown in Fig. 2. It was shown that particles of needle shape (a) or agglomerated particles (b) have a large content of the impurities. From EDS analysis in Fig. 2 and 3 XRD results with diffraction angle range in Fig. 3, impurity elements were identified as Al, Fe Ca K, and Na, confirming silicate minerals such as feldspar, wollastonite, albite. In addition, it was also confirmed in Fig. 3 that the Fe was present as single oxide and impurities of Al, Ca, Mg, K etc. existed as complex oxides.
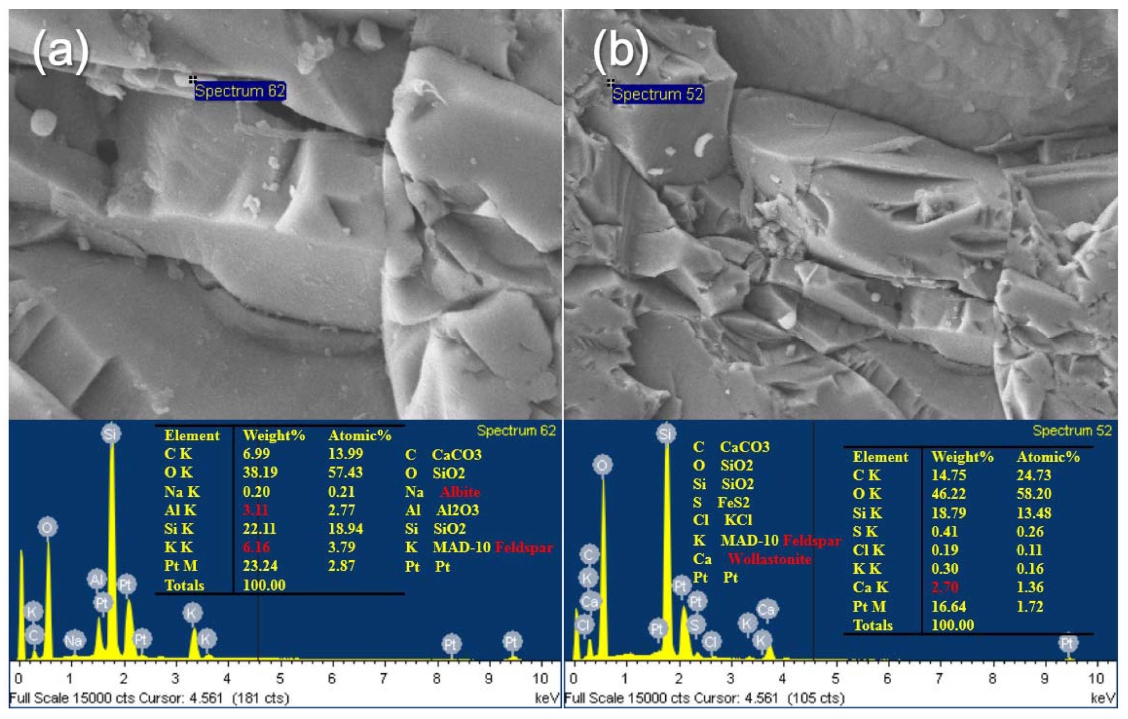
Fig. 2.
Chemical composition of impurities on crack plane. (a) needle shape particles, (b) agglomerated particles.
The presence and the distribution of impurities by EDS mapping is shown in Fig. 4. A lot of particles less than 10 μm were found on the fracture surface. Most of them were identified as silica, although some particles of unique shapes or agglomerated particles, as in Fig. 2, were impurities. Plane mapping results in Fig. 4 confirmed that large content of the impurities was distributed on the fracture surface, which was not the case at grain boundaries. Ca, Ti and K were distributed on a wide surface range, otherwise Fe and Al were observed as concentrated forms.
3.3 Effect of acidic washing solution
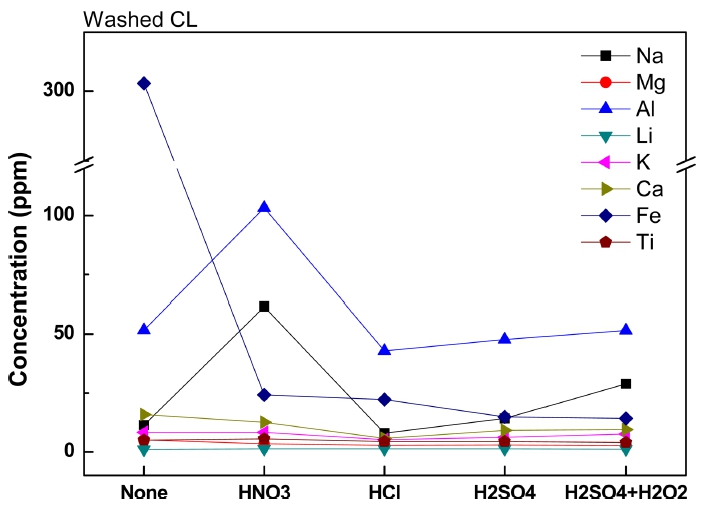
The ground silica ore powder of 3~10 μm (d50 = 8.937 μm) after crushing and milling was applied to acidic washing, which showed the most effective liberation of impurities. Typically, mono oxides can be easily removed in the acid solution.10-12) Fig. 5 shows the content of the impurities according to acidic solutions. 11~31 % of impurities were removed depending on the kind of applied solutions and impurities, and the highest removal of Fe was confirmed. The relatively high content of Fe was attributed to the metallic Fe contamination during crushing with Jaw crusher. HCl was the most excellent acidic solution for impurity removal, and of HNO3 appeared the worst.
3.4 Mechano-chemical acidic washing
Conventional process for the removal of Fe and Al, the main impurities in silica ore, is the repetitive washing with acidic solution after physical separation process such as the magnetic separation and gravity separation.13,14) Physical separation process is also applied between each step of the washing process.8) This study determined the particle size of the ground silica ore for the efficient liberation of the impurities, by which the 1step purification process for producing a high purity silica powder was proposed, i.e., the wet milling in acidic washing solution.
The Mechano-Chemical Washing(MCW) is expected to enhance the efficiency of impurity removal by physical energy generated from mechanical milling in acidic solution, and that is also expected to be more economical than the conventional multi step process. The effect of milling time and milling speed during the mechanochemical acidic washing is shown in Fig. 6. It is clearly evident in Fig. 6(a) that the content of impurities decreased with the increasing milling speed, and that the main impurities of Fe and Al showed the great removal efficiency. This mechano-chemical process showed the improved removal efficiency of impurities than conventional process by more than 2 times. The milling speed of 400 rpm was chosen to reveal the effect of milling time because the milling speed over that showed no changes in impurity content.
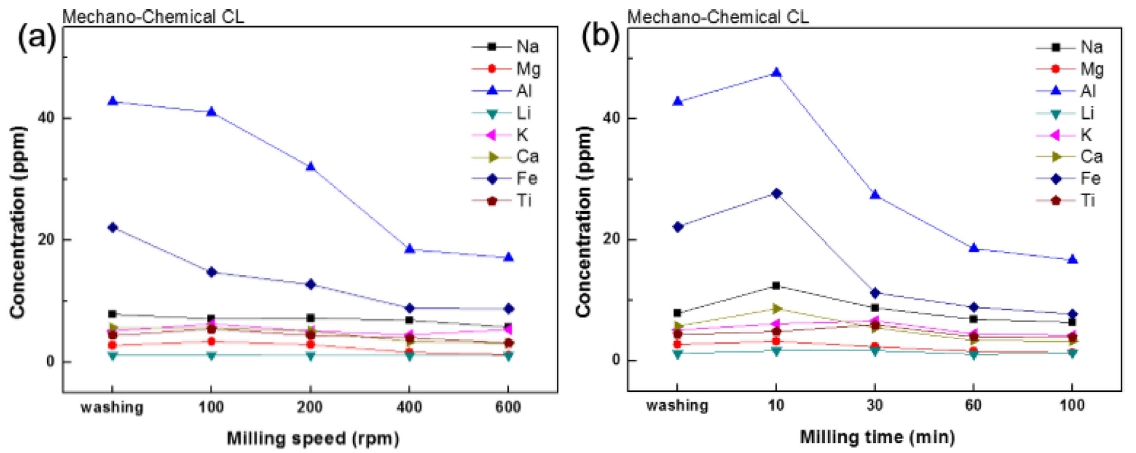
Fig. 6.
Effect of wet milling condition on impurity removal; (a) milling speed (1 M HCl, 60 min), (b) milling time (1 M HCl, 400 rpm).
The effect of milling time, Fig. 6(b), showed effective impurity removal after 30 min by MCW. This time corresponded to the reduced processing time by ~30 % comparing with the process without MCW. The residual amounts of the impurities were mostly less than 10 ppm except for Al.
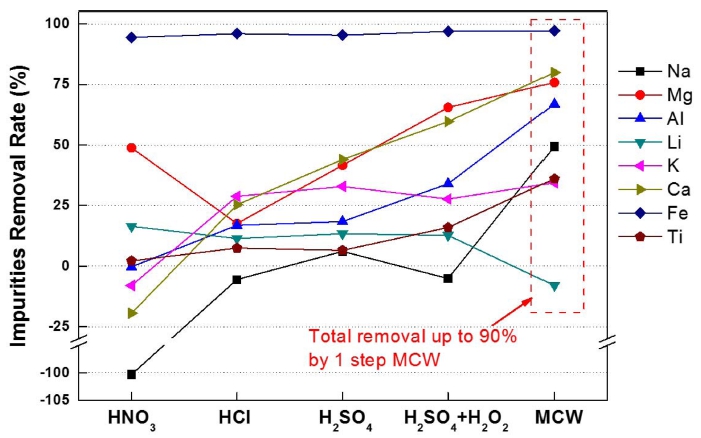
The removal efficiency of impurities in silica ore with the conventional multi step process and the one step MCW process is compared and in Fig. 7. The major impurities were removed by ~90 % when applying the MCW process at 400 rpm for 1 hour in 1 M HCl solution, and resulted in a 4N grade silica powder of 99.995 %. A ~90 % of the impurities were removed while applying the MCW process, 3N grade starting silica ore was converted to the 4N grade silica powder, and ~15 % of silica ore was lost possibly due to the finer powder less than ~2.5 μm by water washing and filtering after the MCW process.
4 Conclusions
In this study, the characteristics of impurities in natural silica ore were investigated by mineralogical analysis for preparing a highly pure silica powder. The content of impurities increased with decreasing particle size of silica ore after pulverization and screening. The main impurities Al, Fe were found to be liberated and detected on the particle surface. The impurities in silica ore existed either as single oxide or as complex oxides like feldspar. HCl was the most effective acidic solution for impurity removal. The MCW process was proposed and applied to enhance the efficiency of impurity removal through the economical single step washing process. The MCW process appeared more effective in purification owing to shorter process time and more impurity removal than the conventional multi step process. With the MCW process, 99.995 % of highly pure silica powder was able to be produced from a natural silica ore.